Abstract
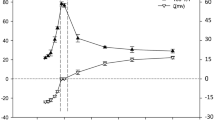
We studied the complex between ovalbumin and long flexible poly-(sodium 4-styrene sulfonate) as a function of pH and temperature. We used various techniques [turbidimetry, conductometry, dynamic light scattering, viscosimetry, and ultra-small-angle light scattering (USALS)] to fully characterize the coacervate complex. Different phases of complexation versus temperature were determined by turbidimetric analysis (pHc, pHϕ1, and pHϕ2). The optimal protein/polyelectrolyte interaction occurred at pHopt 4. An increase in temperature made the hydrophobic interactions more favorable in the case of the soluble complex and complex coacervation phases (pH > pHϕ2). We systematically determined the activation energy to follow the conformational changes of the complex at different temperatures. At pHopt, the size of the formed complex showed a remarkable decrease with temperature increase. USALS was used to determine simultaneously the radius of gyration (Rg) and fractal dimension Df of the coacervate.








Similar content being viewed by others
References
Abbes F, Masmoudi M, Kchaou W, Danthine S, Blecker C, Attia H, Besbes S (2015) Effect of enzymatic treatment on rheological properties, glass temperature transition and microstructure of date syrup. LWT Food Sci Technol 60(1):339–345. https://doi.org/10.1016/j.lwt.2014.08.027
Aubert C, Cannell DS (1986) Restructuring of colloidal silica aggregates. Physl Rev Lett. https://doi.org/10.1103/PhysRevLett.56.738
Ayadi M, Leuliet J, Chopard F, Berthou M, Lebouche M (2004) Electrical conductivity of whey protein deposit: Xanthan gum effect on temperature dependency. Food Bioprod Process 82:320–325. https://doi.org/10.1205/fbio.82.4.320.56398
Benoit P, Deransart E (1976) La conductivité-définition: loi de la conductivite´, les mesures physico-chimiques dans l’industrie, pH, potentiel d’oxydo-réduction, conductivité, ions spécifiques. In: Benoit P, Deransart E (eds) Techniques et Documentation, Entreprise Moderne d’Edition, Paris, France, pp 225–231, 265–286
Berne B, Pecora R (2000) Dynamic light scattering with applications to chemistry, biology, and physics. Dover Publications, New York
Bordi F, Colby RH, Cametti C, De Lorenzo L, Gili T (2002) Electrical conductivity of polyelectrolyte solutions in the semidilute and concentrated regime: the role of counterion condensation. J Phys Chem B 106:6887–6893. https://doi.org/10.1021/jp020262i
Bowman W, Rubinstein M, Tan J (1997) Polyelectrolyte−gelatin complexation: light-scattering study. Macromolecules 30:3262–3270. https://doi.org/10.1021/ma961915u
Brown W (1993) Dynamic light scattering: the method and some applications. Clarendon Press, Oxford
Cao Y, Fang Y, Nishinari K, Phillips GO (2016) Effects of conformational ordering on protein/polyelectrolyte electrostatic complexation: ionic binding and chain stiffening. Sci Rep 6(23739):1–11. https://doi.org/10.1038/srep23739
Chen SH, Teixeira J (1986) Structure and fractal dimension of protein–detergent complexes. Phys Rev Lett 57:2583. https://doi.org/10.1103/PhysRevLett.57.2583
Cipelletti L, Manley S, Ball R, Weitz D (2000) Universal aging features in the restructuring of fractal colloidal gels. Phys Rev Lett 84:2275. https://doi.org/10.1103/PhysRevLett.84.2275
Cohen MH, Turnbull D (1959) Molecular transport in liquids and glasses. J Chem Phys 31:1164–1169. https://doi.org/10.1063/1.1730566
Cousin F, Gummel J, Combet S, Boué F (2011) The model Lysozyme-PSSNa system for electrostatic complexation: Similarities and differences with complex coacervation. Adv Coll Interface Sci 167:71–84. https://doi.org/10.1016/j.cis.2011.05.007
De Alwis AAP, Fryer PJ (1992) Operability of the ohmic heating process: electrical conductivity effects. J Food Eng 15:21–48. https://doi.org/10.1016/0260-8774(92)90038-8
Espinosa-Andrews H, Baez-Gonzalez JG, Cruz-Sosa F, Vernon-Carter EJ (2007) Gum arabic–chitosan complex coacervation. Biomacromol 8:1313–1318. https://doi.org/10.1021/bm0611634
Ferri F (1997) Use of a charge coupled device camera for low-angle elastic light scattering. Rev Sci Instrum 68(6):2265–2274. https://doi.org/10.1063/1.1148135
Girard M, Turgeon SL, Gauthier SF (2002) Inter biopolymer complexing between β lactoglobulin and low- and high-methylated pectin measured by potentiometric titration and ultrafiltration. Food Hydrocoll 16:585–591. https://doi.org/10.1016/S0268-005X(02)00020-6
Gold B (2014)Stereoelectronic control of cycloadditions and fragmentationsFlorida State University College of Arts and Science. Thesis November 6
Gtari W, Bey H, Aschi A, Othman T (2017) Impact of macromolecular crowding on structure and properties of pepsin and trypsin. Mater Sci Eng C 72:98–105. https://doi.org/10.1016/j.msec.2016.11.046
Guerin R, Delplace G, Dieulot JY, Leuliet JC, Le Bouche MA (2004) Method for detecting in real time structure changes of food products during a heat transfer process. J Food Eng 64:289–296. https://doi.org/10.1016/j.jfoodeng.2003.10.011
Guinier A, Fournet G, Walker C (1955) Small angle scattering of X-rays. Wiley, New York, DOI
Gummel J, Boué F, Clemens D, Cousin F (2008) Finite size and inner structure controlled by electrostatic screening in globular complexes of proteins and polyelectrolytes. Soft Matter 4:1653–1664. https://doi.org/10.1039/B803773F
Kaibara K, Okazaki T, Bohidar H, Dubin P (2000) pH-induced coacervation in complexes of bovine serum albumin and cationic polyelectrolytes. Biomacromol 1:100–107. https://doi.org/10.1021/bm990006k
Kantor Y, Witten T (1984) Mechanical stability of tenuous objects. Journal de physique Lettres 45:675–679. https://doi.org/10.1051/jphyslet:019840045013067500
Li X, Fang Y, Al-Assaf S, Phillips GO, Yao X, Zhang Y, Zhao M, Zhang K, Jiang F (2012) Complexation of bovine serum albumin and sugar beet pectin: structural transitions and phase diagram. Langmuir 28:10164–10176. https://doi.org/10.1021/la302063u
Liberatore MW, Wyatt NB, Henry M, Dubin PL, Foun E (2009) Shear-induced phase separation in polyelectrolyte/mixed micelle coacervates. Langmuir 25(23):13376–13383. https://doi.org/10.1021/la903260r
Liu SH, Low NH, Nickerson MT (2009) Effect of pH, salt, and biopolymer ratio on the formation of pea protein isolate–gum Arabic complexes. J Agric Food Chem 57:1521–1526. https://doi.org/10.1021/jf802643n
Liu S, Cao YL, Ghosh S, Rousseau DR, Low NH, Nickerson MT (2010) Intermolecular interactions during complex coacervation of pea protein isolate and gum Arabic. J Agric Food Chem 58:552–556. https://doi.org/10.1021/jf902768v
Mine Y (1995) Recent advances in the understanding of egg white protein functionality. Trends Food Sci Technol 6:225–232. https://doi.org/10.1016/S0924-2244(00)89083-4
Niu F, Su Y, Liu Y, Wang G, Zhang Y, Yang Y (2014) Ovalbumin–gum Arabic interactions: effect of pH, temperature, salt, biopolymers ratio and total concentration. Colloids Surf B 113:477–482. https://doi.org/10.1016/j.colsurfb.2013.08.012
Othman M, Aschi A, Gharbi A (2016) Polyacrylic acids–bovine serum albumin complexation: structure and dynamics. Mater Sci Eng C 58:316–323. https://doi.org/10.1016/j.msec.2015.08.057
Scharamm G (2000) A practical approach to rheology and rheometry. Gebrueder HAAKEE GMBH, Karlsruhe, Federal Republic of Germany
Singh SS, Siddhanta AK, Bandyopadhyay S, Meena R, Prasad K, Bohidar HB (2007) Intermolecular complexation and phase separation in aqueous solutions of oppositely charged biopolymers. Int J Biol Macromol 41:185–192. https://doi.org/10.1016/j.ijbiomac.2007.02.004
Sorensen C (2001) Light scattering by fractal aggregates: a review. Aerosol Sci Technol 35:648–687. https://doi.org/10.1080/02786820117868
St-Gelais B, Champagne CP, Erpmoc F, Audet P (1995) The use of electrical conductivity to follow acidification of dairy blends. Int Dairy J 5:427–438. https://doi.org/10.1016/0958-6946(95)00027-Z
Tiebackx FW (1922) Ist die Gelatine-Gummiarabikumflockung ein chemischer oder ein kolloidchemischer Proze. Colloid Polym Sci 31:102–103. https://doi.org/10.1007/BF01422395
Trabelsi S, Aschi A, Othman T, Gharbi A (2014) Complex formation between ovalbumin and strong polyanion PSSNa: study of structure and properties. Mater Sci Eng C 42:295–302. https://doi.org/10.1016/j.msec.2014.05.042
Trabelsi S, Bassalah MA, Aschi A, Othman T, Gharbi A (2016) Study the cooperative motion of long-chain polyelectrolyte in presence of small globular protein. Phys B 503:18–24. https://doi.org/10.1016/j.physb.2016.09.003
Tsouli J, Ville A, Valla H (1976) Controle de la fabrication du fromage Emmenthal par la meéthode conductimétrique. Le Lait 559–560:600–607. https://doi.org/10.1051/lait:1976559-56031
Turgeon S, Schmitt C, Sanchez C (2007) Protein–polysaccharide complexes and coacervates. Curr Opin Colloid Interface Sci 12:166–178. https://doi.org/10.1016/j.cocis.2007.07.007
Weinbreck F, Nieuwenhuijse H, Robijn GW, De Kruif CG (2004) Complexation of whey proteins with carrageenan. J Agric Food Chem 52:3550–3555. https://doi.org/10.1021/jf034969t
Won‐Woo K, Byoungseung Y (2009) Rheological behaviour of acorn starch dispersions: effects of concentration and temperature. Int J Food Sci Technol 44:503–509. https://doi.org/10.1111/j.1365-2621.2008.01760.x
Xiao Q, Tong Q, Lim LT (2012) Pullulan-sodium alginate based edible films: rheological properties of film forming solutions. Carbohyd Polym 87:1689–1695. https://doi.org/10.1016/j.carbpol.2011.09.077
Zhuang Y, Zhou W, Nguyen MH, Hourigan JA (1997) Determination of protein content of whey powder using electrical conductivity measurement. Int Dairy J 7:647–653. https://doi.org/10.1016/S0958-6946(97)00059-9
Acknowledgements
This research was supported by the Ministry of Higher Education and Scientifi Research in Tunisia (MHESRT). We thank Pr. Luca cipelletti from Laboratory of Charles Coulomb University of Montpellier, France, for the help with the USALS software.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Feddaoui, W., Aschi, A., Bey, H. et al. Study of the complex coacervation mechanism between ovalbumin and the strong polyanion PSSNa: influence of temperature and pH. Eur Biophys J 48, 803–811 (2019). https://doi.org/10.1007/s00249-019-01406-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-019-01406-y