Abstract
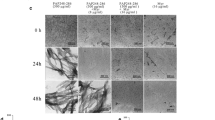
Semen-derived enhancer of virus infection (SEVI), a naturally occurring peptide fragment of prostatic acid phosphatase, enhances HIV infectivity by forming cationic amyloid fibrils that aid the fusion of negatively charged virion and target cell membranes. Cu(II) and Zn(II) inhibit fibrillization of SEVI in a kinetic assay using the fibril-specific dye ThT. TEM suggests that the metals do not affect fibril morphology. NMR shows that the metals bind to histidines 3 and 23 in the SEVI sequence. ITC experiments indicate that SEVI forms oligomeric complexes with the metals. Dissociation constants are micromolar for Cu(II) and millimolar for Zn(II). Because the Cu(II) and Zn(II) concentrations that inhibit fibrillization are comparable with those found in seminal fluid the metals may modulate SEVI fibrillization under physiological conditions.





Similar content being viewed by others
Abbreviations
- DMSO:
-
Dimethyl sulfoxide
- EDTA:
-
Ethylenediaminetetraacetic acid
- ITC:
-
Isothermal titration calorimetry
- HIV:
-
Human immunodeficiency virus
- HSQC:
-
Heteronuclear single-quantum coherence
- NMR:
-
Nuclear magnetic resonance
- NOESY:
-
Nuclear Overhauser enhancement spectroscopy
- PBS:
-
Phosphate-buffered saline
- TBS:
-
Tris-buffered saline
- TEM:
-
Transmission electron microscopy
- ThT:
-
Thioflavin T
- TOCSY:
-
Total correlation spectroscopy
- SEVI:
-
Semen-derived enhancer of virus infection—in this case, specifically the 248–286 fragment of human prostatic acid phosphatase: PAP248–286
References
Alexandrescu AT, Kammerer RA (2003) Structure and disorder in the ribonuclease S-peptide probed by NMR residual dipolar couplings. Protein Sci 12:2132–2140
Andreu JM, Timasheff SN (1986) The measurement of cooperative protein self-assembly by turbidity and other techniques. Methods Enzymol 130:47–59
Aslamkhan AG, Aslamkhan A, Ahearn GA (2002) Preparation of metal ion buffers for biological experimentation: a methods approach with emphasis on iron and zinc. J Exp Zool 292:507–522
Bai Y, Milne JS, Mayne L, Englander SW (1993) Primary structure effects on peptide group hydrogen exchange. Proteins 17:75–86
Bedwal RS, Bahuguna A (1994) Zinc, copper and selenium in reproduction. Experientia 50:626–640
Bohler K, Meisinger V, Klade H, Reinthaller A (1994) Zinc levels of serum and cervicovaginal secretion in recurrent vulvovaginal candidiasis. Genitourin Med 70:308–310
Brender JR, Hartman K, Nanga RP, Popovych N, de la Salud Bea R, Vivekanandan S, Marsh EN, Ramamoorthy A (2010) Role of zinc in human islet amyloid polypeptide aggregation. J Am Chem Soc 132:8973–8983
Brender JR, Nanga RP, Popovych N, Soong R, Macdonald PM, Ramamoorthy A (2011) The amyloidogenic SEVI precursor, PAP248-286, is highly unfolded in solution despite an underlying helical tendency. Biochim Biophys Acta 1808:1161–1169
Camejo MI, Abdala L, Vivas-Acevedo G, Lozano-Hernandez R, Angeli-Greaves M, Greaves ED (2011) Selenium, Copper and Zinc in seminal plasma of men with varicocele, relationship with seminal parameters. Biol Trace Elem Res 143:1247–1254. doi:10.1007/s12011-011-8957-5
Cohlberg JA, Li J, Uversky VN, Fink AL (2002) Heparin and other glycosaminoglycans stimulate the formation of amyloid fibrils from alpha-synuclein in vitro. Biochemistry 41:1502–1511
Collier HB (1979) Binding of Zn2+ by buffers. Clin Chem 25:495–496
Creighton TE (1993) Proteins: structures and molecular properties, 2nd edn. W. H. Freeman and Company, New York
Easterhoff D, DiMaio JT, Doran TM, Dewhurst S, Nilsson BL (2011) Enhancement of HIV-1 infectivity by simple, self-assembling modular peptides. Biophys J 100:1325–1334
Giehm L, Otzen DE (2010) Strategies to increase the reproducibility of protein fibrillization in plate reader assays. Anal Biochem 400:270–281
Good NE, Winget GD, Winter W, Connolly TN, Izawa S, Singh RM (1966) Hydrogen ion buffers for biological research. Biochemistry 5:467–477
Hagenfeldt K (1972) Intrauterine contraception with the copper-T device. 4. Influence on protein and copper concentrations and enzyme activities in uterine washings. Contraception 6:219–230
Harper JD, Lansbury PT Jr (1997) Models of amyloid seeding in Alzheimer’s disease and scrapie: mechanistic truths and physiological consequences of the time-dependent solubility of amyloid proteins. Annu Rev Biochem 66:385–407
Hauber I, Hohenberg H, Holstermann B, Hunstein W, Hauber J (2009) The main green tea polyphenol epigallocatechin-3-gallate counteracts semen-mediated enhancement of HIV infection. Proc Natl Acad Sci USA 106:9033–9038
Hong S, Klein EA, Das Gupta J, Hanke K, Weight CJ, Nguyen C, Gaughan C, Kim KA, Bannert N, Kirchhoff F, Munch J, Silverman RH (2009) Fibrils of prostatic acid phosphatase fragments boost infections with XMRV (xenotropic murine leukemia virus-related virus), a human retrovirus associated with prostate cancer. J Virol 83:6995–7003
Huang YL, Tseng WC, Cheng SY, Lin TH (2000) Trace elements and lipid peroxidation in human seminal plasma. Biol Trace Elem Res 76:207–215
Kim KA, Yolamanova M, Zirafi O, Roan NR, Staendker L, Forssmann WG, Burgener A, Dejucq-Rainsford N, Hahn BH, Shaw GM, Greene WC, Kirchhoff F, Munch J (2010) Semen-mediated enhancement of HIV infection is donor-dependent and correlates with the levels of SEVI. Retrovirology 7:55
LeVine H 3rd (1993) Thioflavine T interaction with synthetic Alzheimer’s disease beta-amyloid peptides: detection of amyloid aggregation in solution. Protein Sci 2:404–410
Martellini JA, Cole AL, Svoboda P, Stuchlik O, Chen LM, Chai KX, Gangrade BK, Sorensen OE, Pohl J, Cole AM (2011) HIV-1 enhancing effect of prostatic acid phosphatase peptides is reduced in human seminal plasma. PLoS ONE 6:e16285
Morgan CJ, Gelfand M, Atreya C, Miranker AD (2001) Kidney dialysis-associated amyloidosis: a molecular role for copper in fiber formation. J Mol Biol 309:339–345
Morris AM, Watzky MA, Finke RG (2009) Protein aggregation kinetics, mechanism, and curve-fitting: a review of the literature. Biochim Biophys Acta 1794:375–397
Munch J, Rucker E, Standker L, Adermann K, Goffinet C, Schindler M, Wildum S, Chinnadurai R, Rajan D, Specht A, Gimenez-Gallego G, Sanchez PC, Fowler DM, Koulov A, Kelly JW, Mothes W, Grivel JC, Margolis L, Keppler OT, Forssmann WG, Kirchhoff F (2007) Semen-derived amyloid fibrils drastically enhance HIV infection. Cell 131:1059–1071
Noormagi A, Gavrilova J, Smirnova J, Tougu V, Palumaa P (2010) Zn(II) ions co-secreted with insulin suppress inherent amyloidogenic properties of monomeric insulin. Biochem J 430:511–518
Olsen JS, Brown C, Capule CC, Rubinshtein M, Doran TM, Srivastava RK, Feng C, Nilsson BL, Yang J, Dewhurst S (2010) Amyloid-binding small molecules efficiently block SEVI (semen-derived enhancer of virus infection)- and semen-mediated enhancement of HIV-1 infection. J Biol Chem 285:35488–35496
Olsen JS, Dimaio JT, Doran TM, Brown C, Nilsson BL, Dewhurst S (2012) Seminal plasma accelerates semen-derived enhancer of viral infection (SEVI) fibril formation by the prostatic acid phosphatase (PAP248-286) peptide. J Biol Chem 287:11842–11849
Owen DH, Katz DF (2005) A review of the physical and chemical properties of human semen and the formulation of a semen simulant. J Androl 26:459–469
Raman B, Ban T, Yamaguchi K, Sakai M, Kawai T, Naiki H, Goto Y (2005) Metal ion-dependent effects of clioquinol on the fibril growth of an amyloid beta peptide. J Biol Chem 280:16157–16162
Roan NR, Munch J, Arhel N, Mothes W, Neidleman J, Kobayashi A, Smith-McCune K, Kirchhoff F, Greene WC (2009) The cationic properties of SEVI underlie its ability to enhance human immunodeficiency virus infection. J Virol 83:73–80
Sacchettini JC, Kelly JW (2002) Therapeutic strategies for human amyloid diseases. Nat Rev Drug Discov 1:267–275
Salamekh S, Brender JR, Hyung SJ, Nanga RP, Vivekanandan S, Ruotolo BT, Ramamoorthy A (2011) A two-site mechanism for the inhibition of IAPP amyloidogenesis by zinc. J Mol Biol 410:294–306
Santner A, Uversky VN (2010) Metalloproteomics and metal toxicology of alpha-synuclein. Metallomics 2:378–392
Sheftic SR, Croke RL, LaRochelle JR, Alexandrescu AT (2009) Electrostatic contributions to the stabilities of native proteins and amyloid complexes. Methods Enzymol 466:233–258
Silvennoinen L, Sandalova T, Schneider G (2009) The polyketide cyclase RemF from Streptomyces resistomycificus contains an unusual octahedral zinc binding site. FEBS Lett 583:2917–2921
Singh N, Das D, Singh A, Mohan ML (2010) Prion protein and metal interaction: physiological and pathological implications. Curr Issues Mol Biol 12:99–107
Steinmetz MO, Jelesarov I, Matousek WM, Honnappa S, Jahnke W, Missimer JH, Frank S, Alexandrescu AT, Kammerer RA (2007) Molecular basis of coiled-coil formation. Proc Natl Acad Sci USA 104:7062–7067
Tougu V, Tiiman A, Palumaa P (2011) Interactions of Zn(II) and Cu(II) ions with Alzheimer’s amyloid-beta peptide. Metal ion binding, contribution to fibrillization and toxicity. Metallomics 3:250–261
Volles MJ, Lansbury PT Jr (2007) Relationships between the sequence of alpha-synuclein and its membrane affinity, fibrillization propensity, and yeast toxicity. J Mol Biol 366:1510–1522
Wurm M, Schambach A, Lindemann D, Hanenberg H, Standker L, Forssmann WG, Blasczyk R, Horn PA (2010) The influence of semen-derived enhancer of virus infection on the efficiency of retroviral gene transfer. J Gene Med 12:137–146
Zhang Y-Z (1995) Protein and peptide structure and interactions studied by hydrogen exchange and NMR. Ph.D. Thesis, University of Pennsylvania
Acknowledgments
We thank Molly Siegel and Tyler Daman for technical assistance during the early stages of this project and Dr Olga Vinogradova for use of her ITC machine. This work was supported by an NSF Graduate Research Fellowship to SRS, a UConn Summer Undergraduate Research Fellowship to JS, and by an American Diabetes Association Basic Science Award to ATA.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors Sarah R. Sheftic and Jessica M. Snell contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sheftic, S.R., Snell, J.M., Jha, S. et al. Inhibition of semen-derived enhancer of virus infection (SEVI) fibrillogenesis by zinc and copper. Eur Biophys J 41, 695–704 (2012). https://doi.org/10.1007/s00249-012-0846-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-012-0846-0