Abstract
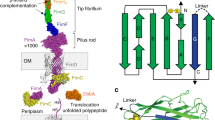
Many bacteria are characterized by nanoscale ultrastructures, for example S-layers, flagella, fimbriae, or pili. The last two are especially important for attachment to different abiotic and biotic surfaces and for host–pathogen interactions. In this study, we investigated the geometric and elastic properties of pili of different Corynebacterium diphtheriae strains by atomic force microscopy (AFM). We performed quantitative contour-length analysis of bacterial pili and found that the visible contour length of the pili can be described by a log-normal distribution. Our data revealed significant strain-specific variations in the mean visible contour length of the pili, ranging from 260 to 1,590 nm. To estimate their full contour length, which is not directly accessible from the AFM images, we developed a simple correction model. Using this model, we determined the mean full contour length as 510–2,060 nm. To obtain the persistence length we used two different methods of analysis, one based on the end-to-end distance of the pili and one based on the bending angles of short segments. In comparison, the bending angle analysis proved to be more precise and resulted in persistence lengths in the narrow range of 220–280 nm, with no significant strain-specific variations. This is small compared with some other bacterial polymers, for example type IV pili, F-pili, or flagella.





Similar content being viewed by others
References
Abels JA, Moreno-Herrero F, van der Heijden T, Dekker C, Dekker NH (2005) Single-molecule measurements of the persistence length of double-stranded RNA. Biophys J 88(4):2737–2744. doi:10.1529/biophysj.104.052811
Anselmetti D, Hansmeier N, Kalinowski J, Martini J, Merkle T, Palmisano R, Ros R, Schmied K, Sischka A, Toensing K (2007) Analysis of subcellular surface structure, function and dynamics. Anal Bioanal Chem 387(1):83–89. doi:10.1007/s00216-006-0789-3
Arce FT, Carlson R, Monds J, Veeh R, Hu FZ, Stewart PS, Lal R, Ehrlich GD, Avci R (2009) Nanoscale structural and mechanical properties of nontypeable Haemophilus influenzae biofilms. J Bacteriol 191(8):2512–2520. doi:10.1128/jb.01596-08
Baumann CG, Smith SB, Bloomfield VA, Bustamante C (1997) Ionic effects on the elasticity of single DNA molecules. Proc Natl Acad Sci USA 94(12):6185–6190
Bednar J, Furrer P, Katritch V, Stasiak A, Dubochet J, Stasiak A (1995) Determination of DNA persistence length by cryo-electron microscopy. Separation of the static and dynamic contributions to the apparent persistence length of DNA. J Mol Biol 254(4):579–594. doi:10.1006/jmbi.1995.0640
Bertuccini L, Baldassarri L, von Hunolstein C (2004) Internalization of non-toxigenic Corynebacterium diphtheriae by cultured human respiratory epithelial cells. Microb Pathog 37(3):111–118. doi:10.1016/j.micpath.2004.06.002
Bezanilla M, Manne S, Laney DE, Lyubchenko YL, Hansma HG (1995) Adsorption of DNA to mica, silylated mica, and minerals: characterization by atomic force microscopy. Langmuir 11(2):655–659. doi:10.1021/la00002a050
Colombo AV, Hirata R Jr, de Souza CM, Monteiro-Leal LH, Previato JO, Formiga LC, Andrade AF, Mattos-Guaraldi AL (2001) Corynebacterium diphtheriae surface proteins as adhesins to human erythrocytes. FEMS Microbiol Lett 197(2):235–239. doi:10.1111/j.1574-6968.2001.tb10609.x
Dorst L, Smeulders AWM (1987) Length estimators for digitized contours. Comput Vis Graph Image Process 40(3):311–333
Dupres V, Alsteens D, Pauwels K, Dufrêne YF (2009) In vivo imaging of S-layer nanoarrays on Corynebacterium glutamicum. Langmuir 25(17):9653–9655. doi:10.1021/la902238q
Fälker S, Nelson AL, Morfeldt E, Jonas K, Hultenby K, Ries J, Melefors Ö, Normark S, Henriques-Normark B (2008) Sortase-mediated assembly and surface topology of adhesive pneumococcal pili. Mol Microbiol 70(3):595–607. doi:10.1111/j.1365-2958.2008.06396.x
Gaspar AH, Ton-That H (2006) Assembly of distinct pilus structures on the surface of Corynebacterium diphtheriae. J Bacteriol 188(4):1526–1533. doi:10.1128/jb.188.4.1526-1533.2006
Gittes F, Mickey B, Nettleton J, Howard J (1993) Flexural rigidity of microtubules and actin filaments measured from thermal fluctuations in shape. J Cell Biol 120(4):923–934. doi:10.1083/jcb.120.4.923
Guttilla IK, Gaspar AH, Swierczynski A, Swaminathan A, Dwivedi P, Das A, Ton-That H (2009) Acyl enzyme intermediates in sortase-catalyzed pilus morphogenesis in gram-positive bacteria. J Bacteriol 191(18):5603–5612. doi:10.1128/jb.00627-09
Hirata R Jr, Souza SMS, Rocha de Souza CM, Andrade AF, Monteiro-Leal LH, Formiga LCD, Mattos-Guaraldi AL (2004) Patterns of adherence to HEp-2 cells and actin polymerisation by toxigenic Corynebacterium diphtheriae strains. Microb Pathog 36(3):125–130. doi:10.1016/j.micpath.2003.10.002
Hirata R Jr, Napoleao F, Monteiro-Leal LH, Andrade AFB, Nagao PE, Formiga LCD, Fonseca LS, Mattos-Guaraldi AL (2002) Intracellular viability of toxigenic Corynebacterium diphtheriae strains in Hep-2 cells. FEMS Microbiol Lett 215(1):115–119. doi:10.1111/j.1574-6968.2002.tb11379.x
Hogan M, LeGrange J, Austin B (1983) Dependence of DNA helix flexibility on base composition. Nature 304(5928):752–754. doi:10.1038/304752a0
Huxley HE, Stewart A, Sosa H, Irving T (1994) X-ray diffraction measurements of the extensibility of actin and myosin filaments in contracting muscle. Biophys J 67(6):2411–2421. doi:10.1016/s0006-3495(94)80728-3
Jiao Y, Schäffer TE (2004) Accurate height and volume measurements on soft samples with the atomic force microscope. Langmuir 20(23):10038–10045. doi:10.1021/la048650u
Kang HJ, Paterson NG, Gaspar AH, Ton-That H, Baker EN (2009) The Corynebacterium diphtheriae shaft pilin SpaA is built of tandem Ig-like modules with stabilizing isopeptide and disulfide bonds. Proc Natl Acad Sci USA 106(40):16967–16971. doi:10.1073/pnas.0906826106
Koch AL (1966) The logarithm in biology 1. Mechanisms generating the log-normal distribution exactly. J Theoret Biol 12(2):276–290. doi:10.1016/0022-5193(66)90119-6
Kojima H, Ishijima A, Yanagida T (1994) Direct measurement of stiffness of single actin filaments with and without tropomyosin by in vitro nanomanipulation. Proc Natl Acad Sci USA 91(26):12962–12966
Landau LD, Lifshits EM, Pitaevskiĭ LP (1980) Statistical physics. Course of theoretical physics, 3rd edn. Butterworth-Heinemann, Oxford
Landau LD, Lifshitz EM, Kosevich AM, Pitaevskiĭ LP (1986) Theory of elasticity. Course of theoretical physics, 3rd edn. Butterworth-Heinemann, Oxford
Leung KM, Wanger G, Guo Q, Gorby Y, Southam G, Lau WM, Yang J (2011) Bacterial nanowires: conductive as silicon, soft as polymer. Soft Matter 7(14):6617–6621. doi:10.1039/c1sm05611e
Limpert E, Stahel WA (2011) Problems with using the normal distribution—and ways to improve quality and efficiency of data analysis. PLoS ONE 6(7):e21403. doi:10.1371/journal.pone.0021403
Limpert E, Stahel WA, Abbt M (2001) Log-normal distributions across the sciences: keys and clues. Bioscience 51(5):341–352. doi:10.1641/0006-3568(2001)051[0341:LNDATS]2.0.CO;2
Mandlik A, Swierczynski A, Das A, Ton-That H (2007) Corynebacterium diphtheriae employs specific minor pilins to target human pharyngeal epithelial cells. Mol Microbiol 64(1):111–124. doi:10.1111/j.1365-2958.2007.05630.x
Mandlik A, Das A, Ton-That H (2008) The molecular switch that activates the cell wall anchoring step of pilus assembly in gram-positive bacteria. Proc Natl Acad Sci USA 105(37):14147–14152. doi:10.1073/pnas.0806350105
Mantelli S, Muller P, Harlepp S, Maaloum M (2011) Conformational analysis and estimation of the persistence length of DNA using atomic force microscopy in solution. Soft Matter 7(7):3412–3416. doi:10.1039/c0sm01160f
Miller E, Garcia T, Hultgren S, Oberhauser AF (2006) The mechanical properties of E. coli type 1 pili measured by atomic force microscopy techniques. Biophys J 91(10):3848–3856. doi:10.1529/biophysj.106.088989
Nagashima H, Asakura S (1980) Dark-field light microscopic study of the flexibility of F-actin complexes. J Mol Biol 136(2):169–182
Ott L, Höller M, Rheinlaender J, Schäffer TE, Hensel M, Burkovski A (2010) Strain-specific differences in pili formation and the interaction of Corynebacterium diphtheriae with host cells. BMC Microbiol 10:257. doi:10.1186/1471-2180-10-257
Reguera G, McCarthy KD, Mehta T, Nicoll JS, Tuominen MT, Lovley DR (2005) Extracellular electron transfer via microbial nanowires. Nature 435(7045):1098–1101. doi:10.1038/nature03661
Rivetti C, Codeluppi S (2000) Accurate length determination of DNA molecules visualized by atomic force microscopy: evidence for a partial B- to A-form transition on mica. Ultramicroscopy 87(1–2):55–66. doi:10.1016/s0304-3991(00)00064-4
Rivetti C, Guthold M, Bustamante C (1996) Scanning force microscopy of DNA deposited onto mica: Equilibration versus kinetic trapping studied by statistical polymer chain analysis. J Mol Biol 264(5):919–932. doi:10.1006/jmbi.1996.0687
Rogers EA, Das A, Ton-That H (2011) Adhesion by pathogenic corynebacteria. In: Linke D, Goldman A (eds) Bacterial adhesion, vol 715. Advances in experimental medicine and biology. Springer, the Netherlands, pp 91–103. doi:10.1007/978-94-007-0940-9_6
Silverman PM, Clarke MB (2010) New insights into F-pilus structure, dynamics, and function. Integr Biol 2(1):25–31. doi:10.1039/b917761b
Skerker JM, Berg HC (2001) Direct observation of extension and retraction of type IV pili. Proc Natl Acad Sci USA 98(12):6901–6904. doi:10.1073/pnas.121171698
Swierczynski A, Ton-That H (2006) Type III pilus of corynebacteria: pilus length is determined by the level of its major pilin subunit. J Bacteriol 188(17):6318–6325. doi:10.1128/jb.00606-06
Taylor WH, Hagerman PJ (1990) Application of the method of phage T4 DNA ligase-catalyzed ring-closure to the study of DNA structure: II. NaCl-dependence of DNA flexibility and helical repeat. J Mol Biol 212(2):363–376. doi:10.1016/0022-2836(90)90131-5
Touhami A, Jericho MH, Boyd JM, Beveridge TJ (2006) Nanoscale characterization and determination of adhesion forces of Pseudomonas aeruginosa pili by using atomic force microscopy. J Bacteriol 188(2):370–377. doi:10.1128/jb.188.2.370-377.2006
Trachtenberg S, Hammel I (1992) The rigidity of bacterial flagellar filaments and its relation to filament polymorphism. J Struct Biol 109(1):18–27. doi:10.1016/1047-8477(92)90063-g
Wang MD, Yin H, Landick R, Gelles J, Block SM (1997) Stretching DNA with optical tweezers. Biophys J 72(3):1335–1346. doi:10.1016/s0006-3495(97)78780-0
Wang YA, Yu X, Overman S, Tsuboi M, Thomas GJ Jr, Egelman EH (2006) The structure of a filamentous bacteriophage. J Mol Biol 361(2):209–215. doi:10.1016/j.jmb.2006.06.027
Wright CJ, Shah MK, Powell LC, Armstrong I (2010) Application of AFM from microbial cell to biofilm. Scanning 32(3):134–149. doi:10.1002/sca.20193
Yanagawa R, Honda E (1976) Presence of pili in species of human and animal parasites and pathogens of the genus corynebacterium. Infect Immun 13(4):1293–1295
Yanagida T, Nakase M, Nishiyama K, Oosawa F (1984) Direct observation of motion of single F-actin filaments in the presence of myosin. Nature 307(5946):58–60. doi:10.1038/307058a0
Acknowledgments
ISS strains were kindly provided by C. v. Hunolstein (Istituto Superiore di Sanita, Rome). LO and AB are grateful for financial support of the Deutsche Forschungsgemeinschaft (SFB796, B5). We thank Yves Muller (Biotechnology Division, University of Erlangen-Nuremberg) for assistance with the Protein Data Bank. We thank Asylum Research and Atomic Force F&E for support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rheinlaender, J., Gräbner, A., Ott, L. et al. Contour and persistence length of Corynebacterium diphtheriae pili by atomic force microscopy. Eur Biophys J 41, 561–570 (2012). https://doi.org/10.1007/s00249-012-0818-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-012-0818-4