Abstract
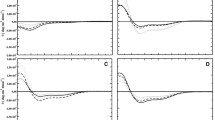
The interaction of the synthetic antimicrobial peptide P5 (KWKKLLKKPLLKKLLKKL-NH2) with model phospholipid membranes was studied using solid-state NMR and circular dichroism (CD) spectroscopy. P5 peptide had little secondary structure in buffer, but addition of large unilamellar vesicles (LUV) composed of dimyristoylphosphatidylcholine (DMPC) increased the β-sheet content to ~20%. Addition of negatively charged LUV, DMPC–dimyristoylphosphatidylglycerol (DMPG) 2:1, led to a substantial (~40%) increase of the α-helical conformation. The peptide structure did not change significantly above and below the phospholipid phase transition temperature. P5 peptide interacted differently with DMPC bilayers with deuterated acyl chains (d54-DMPC) and mixed d54-DMPC–DMPG bilayers, used to mimic eukaryotic and prokaryotic membranes, respectively. In DMPC vesicles, P5 peptide had no significant interaction apart from slightly perturbing the upper region of the lipid acyl chain with minimum effect at the terminal methyl groups. By contrast, in the DMPC–DMPG vesicles the peptide increased disorder throughout the entire acyl chain of DMPC in the mixed bilayer. P5 promoted disordering of the headgroup of neutral membranes, observed by 31P NMR. However, no perturbations in the T 1 relaxation nor the T 2- values were observed at 30°C, although a slight change in the dynamics of the headgroup at 20°C was noticeable compared with peptide-free vesicles. However, the P5 peptide caused similar perturbations of the headgroup of negatively charged vesicles at both temperatures. These data correlate with the non-haemolytic activity of the P5 peptide against red blood cells (neutral membranes) while inhibiting bacterial growth (negatively charged membranes).





Similar content being viewed by others
References
Bak M, Rasmussen JT, Nielsen NC (2000) Simpson: a General simulation program for solid-state NMR spectroscopy. J Magn Reson 147:296–330
Bechinger B (2005) Detergent-like properties of magainin antibiotic peptides: a 31P solid-state NMR spectroscopy study. Biochim Biophys Acta 1712:101–108
Bycroft BW, Shute RE (1985) The molecular basis for the mode of action of beta-lactam antibiotics and mechanisms of resistance. Pharm Res 2(1):3–14
Cornell BA, Separovic F (1983) Membrane thickness and acyl chain length. Biochim Biophys Acta 733:189–193
Cornell BA, Davenport JB, Separovic F (1982) Low frequency motion in membranes: the effect of cholesterol and proteins. Biochim Biophys Acta 689:337–345
Cornell BA, Hiller RG, Raison J, Separovic F, Smith R, Vary JC, Morris C (1983) Biological membranes are rich in low frequency motion. Biochim Biophys Acta 732:473–478
Cullis PR, de Kruijff B (1979) Lipid polymorphism and the functional roles of lipids in biological membranes. Biochim Biophys Acta 559:399–420
Davis JH (1983) The description of membrane lipid conformation, order, and dynamics by 2H NMR. Biochim Biophys Acta 737:117–171
Davis JH, Bloom M, Butler KW, Smith IC (1980) The temperature dependence of molecular order and the influence of cholesterol in Acholeplasma laidlawii membranes. Biochim Biophys Acta 597:477–491
Epand RF, Schmitt MA, Gellman SH, Epand RM (2006) Role of membrane lipids in the mechanism of bacterial species selective toxicity by two α/β-antimicrobial peptides. Biochim Biophys Acta 1758:1343–1350
Fernandez DI, Gehman JD, Separovic F (2009) Membrane interactions of antimicrobial peptides from Australian frogs. Biochim Biophys Acta 1788:1630–1638
Fung BM, Khitrin AK, Ermolaev K (2000) An improved broadband decoupling sequence of liquid crystals and solids. J Magn Reson 142:97–101
Galassi M, Davies J, Theiler J, Gough B, Jungman G, Alken P, Booth M , Rossi F (2009) GNU Scientific Library reference manual (3rd ed vol 1.12). Network Theory Ltd. http://www.gnu.org.software/gsl/. (ISBN 0-9546120-7-8)
Guiliani A, Pirri G, Nicoletto SF (2007) Antimicrobial peptides: an overview of a promising class of therapeutics. Cent Eur J Biol 2(1):1–33
Hooper DC (2001) Mechanisms of action of antimicrobials: focus on fluoroquinolones. Clin Infect Dis 32:S9–S15
Lau T-L, Gehman JD, Wade JD, Masters CL, Barnham KJ, Separovic F (2007) Cholesterol and clioquinol modulation of Aβ (1–42) interaction with phospholipid bilayers and metals. Biochim Biophys Acta 1768:3135–3144
Mor’e JJ (1978) The Levenberg–Marquardt algorithm: implementation and theory. In: Watson G (ed) Lecture notes in Mathematics, vol 630
Oliphant CM, Green GM (2002) Quinolones: a comprehensive review. Am Fam Physician 65:455–464
Oren Z, Shai Y (1999) Mode of action of linear amphipathic α-helical antimicrobial peptides. Biopolymers (Peptide Science) 47:451–463
Park Y, Lee DG, Jang S-H, Woo E-R, Jeong HG, Choi C-H, Hahm K-S (2003) A Leu–Lys-rich antimicrobial peptide: activity and mechanism. Biochim Biophys Acta 1645:172–182
Park Y, Park SN, Park S-C, Shin SO, Kim J-Y, Kang S-J, Kim M-H, Jeong C-Y, Hahm K-S (2006) Synergism of Leu–Lys rich antimicrobial peptides and chloramphenicol against bacterial cells. Biochim Biophys Acta 1764:24–32
Sani MA, Loudet C, Grobner G, Dufourc EJ (2007) Pro-apoptotic bax-α1 synthesis and evidence for β-sheet to α-helix conformational change as triggered by negatively charged lipid membranes. J Pept Sci 13:100–106
Seelig J (1978) 31P Nuclear magnetic resonance and the headgroup structure of phospholipids in membranes. Biochim Biophys Acta 515:105–140
Separovic F, Cornell B, Pace R (2000) Orientation dependence of NMR relaxation time, T1ρ, in lipid bilayers. Chem Phys Lipids 107:159–167
Shai Y (1999) Mechanism of the binding insertion and destabilisation of phospholipid bilayer membranes by α-helical antimicrobial and cell non-selective membrane-lytic peptides. Biochim Biophys Acta 1462:55–70
Shai Y, Oren Z (2001) From “carpet” mechanism to de novo designed diastereomeric cell-selective antimicrobial peptides. Peptides 22:1629–1641
Shakil S, Khan R, Zarrilli R, Khan AU (2008) Aminoglycosides versus bacteria: a description of the action resistance mechanism, and nosocomial battleground. J Biomed Sci 15:5–14
Silvius JR (1982) Thermotropic phase transitions of pure lipids in model membranes and their modifications by membrane proteins lipid-protein interactions. Wiley, New York
Sreerama N, Woody RW (1993) A self-consistent method for the analysis of protein secondary structure from circular dichroism. Anal Biochem 209:32–44
Sreerama N, Woody RW (2000) Estimation of protein secondary structure from circular dichroism spectra: comparison of CONTIN, SELCON, and CDSSTR methods with an expanded reference set. Anal Biochem 287:252–260
Sreerama N, Woody RW (2004) On the analysis of membrane protein circular dichroism spectra. Protein Sci 13:100–112
Whittall KP, Sternin E, Bloom M, MacKay AL (1989) J Magn Reson 84:64–71
Yang L, Harroun TA, Weiss TM, Ding L, Huang HW (2001) Barrel-stave model or toroidal model? A case study on melittin pores. Biophys J 81:1475–1785
Acknowledgments
DIF is the recipient of an Australian Postgraduate Award. JDG is the recipient of an Australian Research Council Future Fellowship (FT0991558).
Author information
Authors and Affiliations
Corresponding author
Additional information
D. I. Fernandez and M.-A. Sani contributed equally to the publication.
Membrane-active peptides: 455th WE-Heraeus-Seminar and AMP 2010 Workshop.
Rights and permissions
About this article
Cite this article
Fernandez, D.I., Sani, MA., Gehman, J.D. et al. Interactions of a synthetic Leu–Lys-rich antimicrobial peptide with phospholipid bilayers. Eur Biophys J 40, 471–480 (2011). https://doi.org/10.1007/s00249-010-0660-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-010-0660-5