Abstract
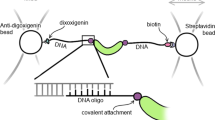
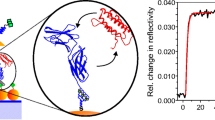
A genetically modified form of the human DNA repair protein O6-alkylguanine-DNA-alkyltransferase (hAGT) was used to immobilize different recombinant hAGT fusion proteins covalently and selectively on gold and glass surfaces. Fusion proteins of hAGT with Glutathione S-Transferase and with tandem repeats of Titin Ig-domains, were produced and anchored via amino-polyethylene glycol benzylguanine. Anchoring was characterized and quantified with surface plasmon resonance, atomic force microscope and fluorescence measurements. Individual fusion proteins were unfolded by single molecule force spectroscopy corroborating the selectivity of the covalent attachment.








Similar content being viewed by others
Abbreviations
- hAGT:
-
O6-alkylguanine-DNA-alkyltransferase
- GST:
-
Glutathione S-Transferase
- PEG:
-
Polyethylene glycol
- BG:
-
Benzylguanine
- SPR:
-
Surface plasmon resonance
- AFM:
-
Atomic force microscope
- EDC:
-
1-ethyl-3-(3-diaminopropyl)carbodiimide hydrochloride
- NHS:
-
N-hydroxysuccinimide
- GST:
-
Glutathione S-transferase
- CMC:
-
Carboxymethylcellulose
References
Butt HJ, Jaschke M (1995) Calculation of thermal noise in atomic force microscopy. Nanotechnology 6:1–7
Damoiseaux R, Keppler A, Johnsson K (2001) Synthesis and applications of chemical probes for human O6-alkylguanine-DNA alkyltransferase. Chembiochem 2:285–287
Daniels DS, Tainer JA (2000) Conserved structural motifs governing the stoichiometric repair of alkylated DNA by O(6)-alkylguanine-DNA alkyltransferase. Mutat Res 460:151–163
Daniels DS, Mol CD, Arvai AS, Kanugula S, Pegg AE, Tainer JA (2000) Active and alkylated human AGT structures: a novel zinc site, inhibitor and extrahelical base binding. Embo J 19:1719–1730
Dietz H, Rief M (2004a) Single molecule force spectroscopy of proteins. In: Proceedings of NATO ASI and SUSSP 59th. Soft condens Matter Phys Mol cell Biol (in press)
Dietz H, Rief M (2004b) Exploring the energy landscape of green fluorescent protein by single molecule mechanical experiments. PNAS 101:16192–16197
Florin E-L, Rief M, Lehmann M, Ludwig M, Dornmair C, Moy T, Gaub HE (1995) Sensing specific molecular interactions with the atomic force microscope. Biosensors Bioelectronics 10:895–901
Gendreizig S, Kindermann M, Johnsson K (2003) Induced protein dimerization in vivo through covalent labeling. J Am Chem Soc 125:14970–14971
Grandbois M, Beyer M, Rief M, Clausen-Schaumann H, Gaub HE (1999) How strong is a covalent bond?. Science 283:1727–1730
Juillerat A, Gronemeyer T, Keppler A, Gendreizig S, Pick H, Vogel H, Johnsson K (2003) Directed evolution of O6-alkylguanine-DNA alkyltransferase for efficient labeling of fusion proteins with small molecules in vivo. Chem Biol 10:313–317
Keppler A, Gendreizig S, Gronemeyer T, Pick H, Vogel H, Johnsson K (2003) A general method for the covalent labeling of fusion proteins with small molecules in vivo. Nat Biotechnol 21:86–89
Kindermann M, George N, Johnsson N, Johnsson K (2003) Covalent and selective immobilization of fusion proteins. J Am Chem Soc 125:7810–7811
Neuert G, Kufer SK, Benoit M, Gaub HE (2004) Modular multichannel surface plasmon spectrometer. Review of Scientific Instruments (in press)
Oesterhelt F, Rief M, Gaub HE (1999) Single molecule force spectroscopy by AFM indicates helical structure of poly(ethylene-glycol) in water. New J Phys, pp 6.1–6.11
Pegg AE, Boosalis M, Samson L, Moschel RC, Byers TL, Swenn K, Dolan ME (1993) Mechanism of inactivation of human O6-alkylguanine-DNA alkyltransferase by O6-benzylguanine. Biochemistry 32:11998–12006
Rief M, Gautel M, Oesterhelt F, Fernandez JM, Gaub HE (1997) Reversible unfolding of individual titin immunoglobulin domains by AFM. Science 276:1109–1112
Acknowledgements
We thank Martin Benoit for advise and help. Special thank also to Jan Barnikow and Kai Johnsson from Covalys Switzerland who provided the BG-anchor and the hAGT vector. This work was supported by the DFG.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kufer, S.K., Dietz, H., Albrecht, C. et al. Covalent immobilization of recombinant fusion proteins with hAGT for single molecule force spectroscopy. Eur Biophys J 35, 72–78 (2005). https://doi.org/10.1007/s00249-005-0010-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-005-0010-1