Abstract
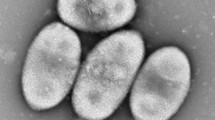
Endosymbiotic bacteria were observed to inhabit the cytoplasm of the freshwater ciliateParamecium novaurelia. Transmission electron microscopy and toxicity tests with sensitive paramecia showed that the endosymbionts belong to the genusCaedibacter. The bacteria conferred a killer trait to their host paramecia. The production of a proteinaceous inclusion body (“R-body”) in the bacterial cell makes them toxic to other paramecia after they become enclosed in food vacuoles. R-bodies ofCaedibacter sp were associated with phages, which are known in most otherCaedibacter species to code for the R-body proteins. The killer-effect ofP. novaurelia on sensitiveP. caudatum strains was of the “paralysis” type, which is a characteristic of the symbiont speciesCaedibacter caryophila. Until nowC. caryophila was known to inhabit the macronucleus ofParamecium caudatum only. Sequencing of the 16S rRNA-gene proved thatCaedibacter sp from the cytoplasm ofP. novaurelia belongs to the speciesC. caryophila as well. The rDNA-sequence of 1695 bp length differed in a total of only 1 bp from the corresponding gene inC. caryophila from the macronucleus ofP. caudatum. The results indicate that the infection of specific host cell compartments may depend on host genes, but not on different traits of the infecting symbiont species. The occurrence of killer and sensitive paramecia strains together in one pond is discussed with respect to the competitive advantage of the killer trait.
Similar content being viewed by others
Referrnces
Austin ML (1948) The killing substance, paramecin: activity of single particles. Am Nat 82:51–59
Austin ML (1948) The killing action and rate of production of single particles of paramecin 51. Physiol Zool 21:69–86
Görtz, H-D (1996) Symbiosis in ciliates. In: Hausmann, K, Bradbury PC (eds) Ciliates: Cells as organisms. Gustav Fischer Verlag, Stuttgart, pp. 441–462
Görtz H-D, Wiemann M (1989) Route of infection of the bacteriaHolospora elegans andHolospora obtusa into the nuclei ofParamecium caudatum. Europ J Protistol 24:101–109
Heruth DP, Pond FR, Dilts JA, Quackenbush RL (1994) Characterization of genetic determinants for R body synthesis and assembly inCaedibacter taeniospiralis 47 and 116. J Bacteriol 176:3559–3567
Kusch J (1998) Local and temporal distribution of different genotypes of pond-dwellingStentor coeruleus. Protist 149:147–154
Kusch J, Heckmann K (1996) Population-structure ofEuplotes cilites revealed by RAPD-fingerprinting. Écoscience 3:378–384
Landis WG (1981) The ecology, role of the killer trait, and interactions of five species of theParamecium aurelia complex inhabiting the littoral zone. Can J Zool 59:1734–1743
Landis WG (1986) The interplay among ecology, breeding systems, and genetics in theParamecium aurelia andParamecium bursaria complexes. In: Corliss JO, Patterson DJ (eds) Progress in Protistology, vol. 1. Biopress Ltd, Bristol, pp 287–307
Landis WG (1987) Factors determining the frequency of the killer trait within populations of theParamecium aurelia complex. Genetics 115:197–205
Landis WG (1988) Ecology. In: Görtz H-D (ed)Paramecium. Springer Verlag, Berlin, pp 419–436
Mueller JA (1963) Separation of kappa particles with infective activity from those with killing activity and identification of the infective particles inParamecium aurelia. Exp Cell Res 30:492–508
Pond FR, Gibson I, Lalucat, J, Quackenbush RL (1989) Rbody-producing bacteria. Microbiol Rev 53:25–67
Preer Jr JR, Preer LB, Jurand A (1974) Kappa and other endosymbionts inParamecium aurelia. Bacteriol Rev 38:113–163
Quackenbush RL (1988) Endosymbionts of killerParamecia. In: Görtz H-D (ed)Paramecium. Springer Verlag, Berlin, pp 406–418
Quackenbush RL, Burbach JA (1983) Cloning and expression of DNA sequences associated with the killer trait ofParamecium tetraurelia stock 47. Proc Natl Acad Sci USA 80:250–254
Schmidt HJ, Görtz H-D, Quackenbush RL (1987)Caedibacter caryophila sp nov, a killer symbiont inhabiting the macronucleus ofParamecium caudatum. Int J Syst Bacteriol 37:459–462
Schmidt HJ, Pond FR, Görtz H-D (1987) Refractile bodies (R bodies) from the macronuclear killer particleCaedibacter caryophila. J Cell Sci 88:177–184
Schmidt HJ, Görtz H-D, Pond FR, Quackenbush RL (1988) Characterization ofCaedibacter endonucleobionts from the macronucleus ofParamecium caudatum and the identification of a mutant with blocked R-body synthesis. Exp Cell Res 174:49–57
Sonneborn TM (1943) Gene and cytoplasm. I. The determination and inheritance of the killer character in variety 4 ofParamecium aurelia. Proc Natl Acad Sci USA 29:329–338
Sonneborn TM (1959)Kappa and related particles inParamecium. Adv Virus Res 6:229–359
Springer N, Ludwig W, Amann R, Schmidt HJ, Görtz H-D, Schleifer K-H (1993) Occurrence of fragmented 16S rRNA in an obligate bacterial endosymbiont ofParamecium caudatum. Proc Natl Acad Sci USA 90:9892–9895
Stoeck T, Schmidt HJ (1997) Fast and accurate identification of European species of theParamecium aurelia complex by RAPD-fingerprints. Microb Ecol 35:311–317
Tallan I (1959) Factors involved in infection by the kappa particles inParamecium aurelia, syngen 4. Physiol Zool 32:78–89
Tallan I (1961) A cofactor required by kappa in the infection ofParamecium aurelia and its possible action. Physiol Zool 34:1–13
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kusch, J., Stremmel, M., Breiner, H.W. et al. The toxic symbiontCaedibacter caryophila in the cytoplasm ofParamecium novaurelia . Microb Ecol 40, 330–335 (2000). https://doi.org/10.1007/s002480000034
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002480000034