Abstract
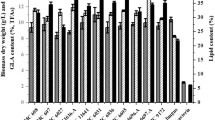
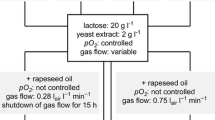
Microbial co-culture simulates the natural ecosystem through the combination of artificial microbes. This approach has been widely applied in the study of activating silent genes to reveal novel secondary metabolites. However, there are still challenges in determining the biosynthetic pathways. In this study, the effects of microbial co-culture on the morphology of the microbes were verified by the morphological observation. Subsequently, through the strategy combining substrate feeding, stable isotope labeling, and gene expression analysis, the biosynthetic pathways of five benzoic acid derivatives N1-N4 and N7 were demonstrated: the secondary metabolite 10-deoxygerfelin of A. sydowii acted as an inducer to induce B. subtilis to produce benzoic acid, which was further converted into 3-OH-benzoic acid by A. sydowii. Subsequently, A. sydowii used 3-OH-benzoic acid as the substrate to synthesize the new compound N2, and then N1, N3, N4, and N7 were biosynthesized upon the upregulation of hydrolase, hydroxylase, and acyltransferase during co-culture. The plate zone analysis suggested that the biosynthesis of the newly induced compounds N1-N4 was mainly attributed to A. sydowii, and both A. sydowii and B. subtilis were indispensable for the biosynthesis of N7. This study provides an important basis for a better understanding of the interactions among microorganisms, providing new ideas for studying the biosynthetic pathways of the newly induced secondary metabolites in co-culture.







Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Blunt JW, Copp BR, Hu WP et al (2019) Marine natural products. Nat Prod Rep 36:122–173
Raad II, Zakhem EA, Helou GE et al (2015) Clinical experience of the use of voriconazole, caspofungin or the combination in primary and salvage therapy of invasive aspergillosis in haematological malignancies. Int J Antimicrob Agents 45:283–288
Wang B, Waters AL, Sims JW et al (2013) Complex marine natural products as potential epigenetic and production regulators of antibiotics from a marine Pseudomonas aeruginosa. Microb Ecol 65:1068–1075
Zhang Q, Lt Z, Li YR et al (2020) Comparative transcriptomics and transcriptional regulation analysis of enhanced laccase production induced by co-culture of Pleurotus eryngii var. ferulae with Rhodotorula mucilaginosa. Appl Microbiol Biotechnol 104:241–255
Baschien C, Rode G, Böckelmann U et al (2009) Interactions between hyphosphere-associated bacteria and the fungus Cladosporium herbarum on aquatic leaf litter. Microb Ecol 58:642–650
Meng LH, Li XM, Li HL et al (2020) Chermebilaenes A and B, new bioactive meroterpenoids from co-cultures of marine-derived isolates of Penicillium bilaiae MA-267 and Penicillium chermesinum EN-480. Mar Drugs 18:339
Liu N, Peng S, Yang J et al (2019) Structurally diverse sesquiterpenoids and polyketides from a sponge-associated fungus Aspergillus sydowii SCSIO41301. Fitoterapia 135:27–32
Yang SQ, Li XM, Li X et al (2018) New citrinin analogues produced by coculture of the marine algal-derived endophytic fungal strains Aspergillus sydowii EN-534 and Penicillium citrinum EN-535 - ScienceDirect. Phytochem Lett 25:191–195
Sharma A, Diwevidi VD, Singh S et al (2013) Biological control and its important in agriculture. Int J Biotechnol Bioeng Res 4:175–180
Sun Y, Liu WC, Shi X et al (2021) Inducing secondary metabolite production of Aspergillus sydowii through microbial co-culture with Bacillus subtilis. Microb Cell Fact 20:42
Xu XY, Shen XT, Yuan XJ et al (2018) Metabolomics investigation of an association of induced features and corresponding fungus during the co-culture of Trametes versicolor and Ganoderma applanatum. Front Microbiol 8:2647
Shinji K, Michio S, Yuta T et al (2016) Evaluation of biosynthetic pathway and engineered biosynthesis of alkaloids. Molecules 21:1078
Yun JW, Dong HK, Song SK (1997) Enhanced production of fructosyltransferase and glucosyltransferase by substrate-feeding cultures of Aureobasidium pullulans. J Ferment Bioeng 84:261–263
Arai Y, Takahashi, et al (1985) Directed biosynthesis of new saframycin derivatives with resting cells of Streptomyces lavendulae. Antimicrob Agents Ch 28:5–11
Shlomi T, Fan J, Tang B et al (2014) Quantitation of Cellular Metabolic Fluxes of Methionine. Anal Chem 86:1583–1591
Jang C, Chen L, Rabinowitz JD (2018) Metabolomics and isotope tracing. Cell 173:822–837
Hammerl R, Frank O, Hofmann T (2017) Differential off-line lC-NMR (DOLC-NMR) metabolomics to monitor tyrosine-induced metabolome alterations in Saccharomyces cerevisiae. J Agr Food Chem 65:3230–3241
Fu Q, Dudley S, Sun C et al (2018) Stable isotope labeling-assisted metabolite probing for emerging contaminants in plants. Anal Chem 90:11040–11047
Wang H, Peng L, Ding Z et al (2015) Stimulated laccase production of Pleurotus ferulae JM301 fungus by Rhodotorula mucilaginosa yeast in co-culture. Process Biochem 50:901–905
Zhong Z, Li N, Liu L et al (2018) Label-free differentially proteomic analysis of interspecific interaction between white-rot fungi highlights oxidative stress response and high metabolic activity. Fungal Biol-UK 122:774–784
Sun Y, Shi X, Xing Y et al (2022) Co-culture of Aspergillus sydowii and Bacillus subtilis induces the production of antibacterial metabolites. Fungal Biol-UK 126:320–332
Li YC, Tao WY, Cheng L (2009) Paclitaxel production using co-culture of Taxus suspension cells and paclitaxel-producing endophytic fungi in a co-bioreactor. Appl Microbiol Biotechnol 83:233–239
Chiang YM, Chang SL, Oakley BR et al (2011) Recent advances in awakening silent biosynthetic gene clusters and linking orphan clusters to natural products in microorganisms. Curr Opin Chem Biol 15:137–143
Birkó Z, Bialek S, Buzás K et al (2007) The secreted signaling protein factor C triggers the A-factor response regulon in Streptomyces griseus: overlapping signaling routes*. Mol Cell Proteomics 6:1248–1256
Liang JG, Chu XH, Xiong ZQ et al (2011) Oxygen uptake rate regulation during cell growth phase for improving avermectin B1a batch fermentation on a pilot scale (2 m3). World J Microbiol Biotechnol 27:2639–2644
Shang Z, Salim AA, Capon RJ (2017) Chaunopyran A: Co-cultivation of marine mollusk-derived fungi activates a rare class of 2-Alkenyl-Tetrahydropyran. J Nat Prod 80:1167–1172
Knowles S, Raja H, Wright A et al (2019) Mapping the fungal battlefield: using in situ chemistry and deletion mutants to monitor interspecific chemical interactions between fungi. Front Microbiol 10:285
Olmo AD, Calzada J, Nuez M (2017) Benzoic acid and its derivatives as naturally occurring compounds in foods and as additives: uses, exposure and controversy. Crit Rev Food Sci Nutr 57:3084–3103
Xiang L, Moore BS (2003) Characterization of benzoyl coenzyme a biosynthesis genes in the enterocin-producing bacterium “streptomyces maritimus.” J Bacteriol 185:399–404
Acknowledgements
This work was supported by the grants from the Natural Science Foundation of Liaoning Province of China (2019-ZD-0143) and Science and Technology Development Program of Jilin Province of China (No. 202110401131YY).
Funding
This work was supported by the grants from the Natural Science Foundation of Liaoning Province of China (2019-ZD-0143) and Science and Technology Development Program of Jilin Province of China (No. 202110401131YY).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis, the first draft of the manuscript was written by Yu Sun, Yu Sun and Yue-Sheng Dong conceived and designed the work, Xuan Shi, Liang-Yu He, Quo-Feng Guo and Zhi-Long Xiu participated in the study, Yan Xing polished the language of the paper, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
This is an observational study. The XYZ Research Ethics Committee has confirmed that no ethical approval is required.
Competing Interests
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, Y., Shi, X., He, LY. et al. Biosynthetic Profile in the Co-culture of Aspergillus sydowii and Bacillus subtilis to Produce Novel Benzoic Derivatives. Microb Ecol 85, 1288–1299 (2023). https://doi.org/10.1007/s00248-022-02029-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-022-02029-1