Abstract
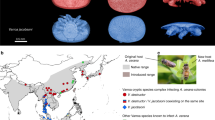
Host-parasite co-evolution is a process of reciprocal, adaptive genetic change. In natural conditions, parasites can shift to other host species, given both host and parasite genotypes allow this. Even though host-parasite co-evolution has been extensively studied both theoretically and empirically, few studies have focused on parasite gene flow between native and novel hosts. Nosema ceranae is a native parasite of the Asian honey bee Apis cerana, which infects epithelial cells of mid-guts. This parasite successfully switched to the European honey bee Apis mellifera, where high virulence has been reported. In this study, we used the parasite N. ceranae and both honey bee species as model organisms to study the impacts of two-host habitat sharing on parasite diversity and virulence. SNVs (Single Nucleotide Variants) were identified from parasites isolated from native and novel hosts from sympatric populations, as well as novel hosts from a parapatric population. Parasites isolated from native hosts showed the highest levels of polymorphism. By comparing the parasites isolated from novel hosts between sympatric and parapatric populations, habitat sharing with the native host significantly enhanced parasite diversity, suggesting there is continuing gene flow of parasites between the two host species in sympatric populations.


Similar content being viewed by others
Data availability
Data generated from this study have been deposited in NCBI BioProject PRJNA602377 with accession # SAMN13893430 and SAMN19893431. Additional comparative data were downloaded from NCBI BioProject PRJNA209464 with accession # SAMN02213592.
References
Acevedo MA, Dillemuth FP, Flick AJ, Faldyn MJ, Elderd BD (2019) Virulence-driven trade-offs in disease transmission: a meta-analysis. Evol Int J Org Evol 73(4):636–647
Akey JM, Zhang G, Zhang K, Jin L, Shriver MD (2002) Interrogating a high-density SNP map for signatures of natural selection. Genome Res 12(12):1805–1814
Anderson D, East IJ (2008) The latest buzz about colony collapse disorder. Science 319:724–725
Antunez K, Martin-Hernandez R, Prieto L, Meana A, Zunino P, Higes M (2009) Immune suppression in the honey bee (Apis mellifera) following infection by Nosema ceranae (Microsporidia). Environ Microbiol 11(9):2284–2290
Bataillon T, Duan J, Hvilsom C, Jin X, Li Y, Skov L et al (2015) Inference of purifying and positive selection in three subspecies of chimpanzees (Pan troglodytes) from exome sequencing. Genom Biol Evol 7(4):1122–1132
Castillo JA, Agathos SN (2019) A genome-wide scan for genes under balancing selection in the plant pathogen Ralstonia solanacearum. BMC Evol Biol 19(1):123
Chaimanee V, Pettis JS, Chen Y, Evans JD, Khongphinitbunjong K, Chantawannakul P (2013) Susceptibility of four different honey bee species to Nosema ceranae. Vet Parasitol 193(1–3):260–265
Chen YP, Pettis JS, Zhao Y, Liu X, Tallon LJ, Sadzewicz LD et al (2013) Genome sequencing and comparative genomics of honey bee microsporidia, Nosema apis reveal novel insights into host-parasite interactions. BMC Genom 14(1):451
Cox-Foster DL, Conlan S, Holmes E, Palacios G, Evans JD, Moran NA et al (2007) A metagenomic survey of microbes in honey bee colony collapse disorder. Science 318(5848):283–287
de Jong MA, Wahlberg N, van Eijk M, Brakefield PM, Zwaan BJ (2011) Mitochondrial DNA signature for range-wide populations of Bicyclus anynana suggests a rapid expansion from recent refugia. PLoS ONE 6(6):e21385
Engelstädter J, Fortuna NZ (2019) The dynamics of preferential host switching: host phylogeny as a key predictor of parasite distribution. Evolution 73(7):1330–1340
Evans JD (2006) Beepath: an ordered quantitative-PCR array for exploring honey bee immunity and disease. J Invertebr Pathol 93(2):135–139
Evans JD, Aronstein K, Chen YP, Hetru C, Imler J-L, Jiang H et al (2006) Immune pathways and defence mechanisms in honey bees Apis mellifera. Insect Mol Biol 15(5):645–656
Frank SA (2012) Natural selection. III. Selection versus transmission and the levels of selection. J Evol Biol 25(2):227–243
Fries I, Feng F, da Silva A, Slemenda SB, Pieniazek NJ (1996) Nosema ceranae n. sp. (Microspora, Nosematidae), morphological and molecular characterization of a microsporidian parasite of the Asian honey bee Apis cerana (Hymenoptera, Apidae). Eur J Protistol 32(3):356–365
Fries I, Chauzat M-P, Chen Y-P, Doublet V, Genersch E, Gisder S et al (2013) Standard methods for Nosema research. J Apicult Res 52(1):1–28
Fu YX, Li WH (1993) Statistical tests of neutrality of mutations. Genetics 133(3):693–709
Fürst MA, McMahon DP, Osborne JL, Paxton RJ, Brown MJF (2014) Disease associations between honeybees and bumblebees as a threat to wild pollinators. Nature 506(7488):364–366
Gómez-Moracho T, Maside X, Martin-Hernandez R, Higes M, Bartolome C (2014) High levels of genetic diversity in Nosema ceranae within Apis mellifera colonies. Parasitology 141(4):475–481.
Gómez-Moracho T, Bartolomé C, Bello X, Martín-Hernández R, Higes M, Maside X (2015) Recent worldwide expansion of Nosema ceranae (Microsporidia) in Apis mellifera populations inferred from multilocus patterns of genetic variation. Infect Genet Evol 31:87–94
Gómez-Moracho T, Bartolomé C, Martín-Hernández R, Higes M, Maside X (2015) Evidence for weak genetic recombination at the PTP2 locus of Nosema ceranae. Environ Microbiol 17(4):1300–1309
Graystock P, Goulson D, Hughes WOH (2015) Parasites in bloom: flowers aid dispersal and transmission of pollinator parasites within and between bee species. Proc Biol Sci 282(1813):20151371
Grosberg RK, Hart MW (2000) Mate selection and the evolution of highly polymorphic self/nonself recognition genes. Science 289(5487):2111–2114
Higes M, Martin R, Meana A (2006) Nosema ceranae, a new microsporidian parasite in honeybees in Europe. J Invertebr Pathol 92(2):93–95
Higes M, Martín-Hernández R, Botías C, Bailón EG, González-Porto AV, Barrios L et al (2008) How natural infection by Nosema ceranae causes honeybee colony collapse. Environ Microbiol 10(10):2659–2669
Higes M, Martín-Hernandez R, Garrido-Bailón E, Gonzales-Porto A, García-Palencia P, Meana A et al (2009) Honeybee colony collapse due to Nosema ceranae in professional apiaries. Environ Microbiol Rep 1
Hovestadt T, Thomas JA, Mitesser O, Schönrogge K (2019) Multiple host use and the dynamics of host switching in host–parasite systems. Insect Conserv Divers 12(6):511–522
Huang Q, Kryger P, Le Conte Y, Moritz RFA (2012) Survival and immune response of drones of a Nosemosis tolerant honey bee strain towards N. ceranae infections. J Invertebr Pathol 109(3):297–302
Huang Q, Wu ZH, Li WF, Guo R, Xu JS, Dang XQ et al (2021) Genome and evolutionary analysis of Nosema ceranae: a microsporidian parasite of honey bees. Front Microbiol 12:645353
Johnson PTJ, Preston DL, Hoverman JT, LaFonte BE (2013) Host and parasite diversity jointly control disease risk in complex communities. Proc Natl Acad Sci 110(42):16916–16921
Kofler R, Pandey RV, Schlötterer C (2011) poPoolation2: identifying differentiation between populations using sequencing of pooled DNA samples (Pool-Seq). Bioinformatics 27(24):3435–3436
Kofler R, Orozco-terWengel P, De Maio N, Pandey RV, Nolte V, Futschik A et al (2011) PoPoolation: a toolbox for population genetic analysis of next generation sequencing data from pooled individuals. PLoS ONE 6(1):e15925
Korneliussen TS, Moltke I, Albrechtsen A, Nielsen R (2013) Calculation of Tajima’s D and other neutrality test statistics from low depth next-generation sequencing data. BMC Bioinform 14(1):289
Levin BR, Bull JJ (1994) Short-sighted evolution and the virulence of pathogenic microorganisms. Trends Microbiol 2(3):76–81
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25(14):1754–1760
Li W, Evans J, Li J, Su S, Hamilton M, Chen Y (2017) Spore load and immune response of honey bees naturally infected by Nosema ceranae. Parasitol Res 116:1–10
Lievens EJP, Perreau J, Agnew P, Michalakis Y, Lenormand T (2018) Decomposing parasite fitness reveals the basis of specialization in a two-host, two-parasite system. Evol Lett 2(4):390–405
Mayack C, Naug D (2009) Energetic stress in the honeybee Apis mellifera from Nosema ceranae infection. J Invertebr Pathol 100(3):185–188
Morrison LJ, McLellan S, Sweeney L, Chan CN, MacLeod A, Tait A, Turner CMR (2010) Role for parasite genetic diversity in differential host responses to Trypanosoma brucei Infection. Infect Immun 78(3):1096–1108
Nelson CW, Moncla LH, Hughes AL (2015) SNPGenie: estimating evolutionary parameters to detect natural selection using pooled next-generation sequencing data. Bioinformatics 31(22):3709–3711
Pelin A, Selman M, Aris-Brosou S, Farinelli L, Corradi N (2015) Genome analyses suggest the presence of polyploidy and recent human-driven expansions in eight global populations of the honeybee pathogen Nosema ceranae. Environ Microbiol 17(11):4443–4458
Peters MJ, Suwannapong G, Pelin A, Corradi N (2019) Genetic and genome analyses reveal genetically distinct populations of the bee pathogen Nosema ceranae from Thailand. Microb Ecol 77(4):877–889
Pilosof S, Fortuna MA, Cosson J-F, Galan M, Kittipong C, Ribas A et al (2014) Host–parasite network structure is associated with community-level immunogenetic diversity. Nat Commun 5(1):5172
R Core Team (2013) R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing. http://www.R-project.org/
Rodrigues JA, Acosta-Serrano A, Aebi M, Ferguson MAJ, Routier FH, Schiller I et al (2015) Parasite glycobiology: a bittersweet symphony. PLoS Pathog 11(11):e1005169
Sagastume S, Martín-Hernández R, Higes M, Henriques-Gil N (2016) Genotype diversity in the honey bee parasite Nosema ceranae: multi-strain isolates, cryptic sex or both? BMC Evol Biol 16(1):216.
Sarvari M, Mikani A, Mehrabadi M (2020) The innate immune gene Relish and Caudal jointly contribute to the gut immune homeostasis by regulating antimicrobial peptides in Galleria mellonella. Dev Comp Immunol 110:103732
Smith AL, Hodkinson TR, Villellas J, Catford JA, Csergő AM, Blomberg SP et al (2020) Global gene flow releases invasive plants from environmental constraints on genetic diversity. Proc Natl Acad Sci 117(8):4218–4227
Spiess AN (2018) qpcR: Modelling and analysis of real-time PCR data. R Package, Version 1.4–1
Stajich JE, Hahn MW (2005) Disentangling the effects of demography and selection in human history. Mol Biol Evol 22(1):63–73
Stam R, Silva-Arias GA, Tellier A (2019) Subsets of NLR genes show differential signatures of adaptation during colonization of new habitats. New Phytol 224(1):367–379
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123(3):585–595
Van der Auwera GA, Carneiro MO, Hartl C, Poplin R, Del Angel G, Levy-Moonshine A et al (2013) From FastQ data to high confidence variant calls: the Genome Analysis Toolkit best practices pipeline. Curr Protocols Bioinform 43:1–33
Williams GR, Shutler D, Burgher-MacLellan KL, Rogers REL (2014) Infra-population and -community dynamics of the parasites Nosema apis and Nosema ceranae, and consequences for honey bee (Apis mellifera) Hosts. PLoS ONE 9(7):e99465
Wilmes M, Sahl H-G (2014) Defensin-based anti-infective strategies. Int J Med Microbiol 304(1):93–99
Acknowledgements
The project is supported by initiation package of Jiangxi Agricultural University (050014/923230722), National Natural Science Foundation of China #32060778 and the earmarked fund for Jiangxi Agriculture Research System (JXARS-14).
Author information
Authors and Affiliations
Contributions
QH designed the experiment. QH and LK conducted the experiment. QH, LK, LZZ, WYY, ZJZ and JDE organized the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ke, L., Yan, W.Y., Zhang, L.Z. et al. Honey Bee Habitat Sharing Enhances Gene Flow of the Parasite Nosema ceranae. Microb Ecol 83, 1105–1111 (2022). https://doi.org/10.1007/s00248-021-01827-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-021-01827-3