Abstract
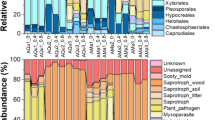
The mountain pine beetle (MPB) is a native bark beetle of western North America that attacks pine tree species, particularly lodgepole pine. It is closely associated with the ophiostomatoid ascomycetes Grosmannia clavigera, Leptographium longiclavatum, Ophiostoma montium, and Ceratocystiopsis sp.1, with which it is symbiotically associated. To develop a better understanding of interactions between beetles, fungi, and host trees, we used target-specific DNA primers with qPCR to assess the changes in fungal associate abundance over the stages of the MPB life cycle that occur in galleries under the bark of pine trees. Multivariate analysis of covariance identified statistically significant changes in the relative abundance of the fungi over the life cycle of the MPB. Univariate analysis of covariance identified a statistically significant increase in the abundance of Ceratocystiopsis sp.1 through the beetle life cycle, and pair-wise analysis showed that this increase occurs after the larval stage. In contrast, the abundance of O. montium and Leptographium species (G. clavigera, L. longiclavatum) did not change significantly through the MPB life cycle. From these results, the only fungus showing a significant increase in relative abundance has not been formally described and has been largely ignored by other MPB studies. Although our results were from only one site, in previous studies we have shown that the fungi described were all present in at least ten sites in British Columbia. We suggest that the role of Ceratocystiopsis sp.1 in the MPB system should be explored, particularly its potential as a source of nutrients for teneral adults.



Similar content being viewed by others
References
Bright D, Stock M (1982) Taxonomy and geographic variation. In: Mitton JB, Sturgeon KB (eds) Bark beetles in North American conifers: a system for the study of evolutionary biology. University of Texas Press, Austin
Six DL, Wingfield MJ (2011) The role of phytopathogenicity in bark beetle–fungus symbioses: a challenge to the classic paradigm. Annu Rev Entomol 56:255–272
Harrington TC (2005) Ecology and evolution of mycophagous bark beetles and their fungal partners. In: Vega FE, Blackwell M (eds) Insect–fungal associations: ecology and evolution. Oxford University Press, New York, pp 257–291
BC MOF (2011) Mountain pine beetle. http://www.for.gov.bc.ca/hfp/mountain_pine_beetle/. Accessed 29 Oct 2011
Safranyik L, Carroll A (2007) The biology and epidemiology of the mountain pine beetle in lodgepole pine forests. In: The mountain pine beetle: a synthesis of biology, management and impacts on lodgepole pine. Natural Resources Canada, Pacific Forest Service, Victoria
Whitney HS (1971) Association of Dendroctonus ponderosae (Coleoptera: Scolytidae) with blue stain fungi and yeasts during brood development in lodgepole pine. Can Entomol 103:1495–1503
Bentz B, Logan J, Amman G (1991) Temperature-dependent development of the mountain pine-beetle (Coleoptera, Scolytidae) and simulation of its phenology. Can Entomol 123:1083–1094
Paine TD, Raffa KF, Harrington T (1997) Interactions among scolytid bark beetles, their associated fungi, and live host conifers. Annu Rev Entomol 42:179–206
Lee S, Kim J-J, Breuil C (2006) Diversity of fungi associated with the mountain pine beetle, Dendroctonus ponderosae and infested lodgepole pines in British Columbia. Fungal Divers 22:91–105
Yamaoka Y, Hiratsuka Y, Maruyama P (1995) The ability of Ophiostoma clavigerum to kill mature lodgepolepine trees. Eur J Forest Pathol 25:401–404
Lee S, Kim J, Breuil C (2006) Pathogenicity of Leptographium longiclavatum associated with Dendroctonus ponderosae to Pinus contorta. Can J Forest Res 36:2864–2872
Rumbold C (1941) A blue stain fungus, Ceratostomella montium n. sp., and some yeasts associated with two species of Dendroctonus. J Agric Res 62:589–601
Robinson R (1962) Blue stain fungi in lodgepole pine (Pinus contorta Dougl. var. latifolia Engelm.) infested by the mountain pine beetle (Dendroctonus monticolae Hopk.). Can J Bot 40:609–614
Whitney HS, Farris SH (1970) Maxillary mycangium in mountain pine beetle. Science 167:54–55
Six DL (2003) Bark beetle–fungus symbioses. In: Bourtzis K, Miller TA (eds) Insect symbiosis. CRC, Boca Raton
Six DL (2003) A comparison of mycangial and phoretic fungi of individual mountain pine beetles. Can J Forest Res 33:1331–1334
Kim J, Allen E, Humble L, Breuil C (2005) Ophiostomatoid and basidiomycetous fungi associated with green, red, and grey lodgepole pines after mountain pine beetle (Dendroctonus ponderosae) infestation. Can J Forest Res 35:274–284
Plattner A et al (2009) Resolving taxonomic and phylogenetic incongruence within species Ceratocystiopsis minuta. Mycologia 101:878–887
Plattner A (2008) Pathogenicity and taxonomy of fungi associated with the mountain pine beetle in British Columbia. University of British Columbia, Vancouver
Hsiau PTW, Harrington TC (2003) Phylogenetics and adaptations of basidiomycetous fungi fed upon by bark beetles (Coleoptera: Scolytidae). Symbiosis 34:111–131
Six DL, Paine TD (1998) Effects of mycangial fungi and host tree species on progeny survival and emergence of Dendroctonus ponderosae (Coleoptera: Scolytidae). Environ Entomol 27:1393–1401
Reid RW (1961) Moisture changes in lodgepole pine before and after attack by the mountain pine beetle. Forestry Chronicle 368–375
Clayton RB (1964) The utilization of sterols by insects. J Lipid Res 15:3–19
Norris DM, Baker JM, Chu HM (1969) Symbiotic interrelationships between microbes and ambrosia beetles. III. Ergosterol as the source of sterol to the insect. Ann Entomol Soc Am 62:413–414
Bentz B, Six DL (2006) Ergosterol content of fungi associated with Dendroctonus ponderosae and Dendroctonus rufipennis (Coleoptera: Curculionidae, Scolytinae). Ann Entomol Soc Am 99:189–194
Bleiker KP, Six DL (2007) Dietary benefits of fungal associates to an eruptive herbivore: potential implications of multiple associates on host population dynamics. Environ Entomol 36:1384–1396
Ayres M, Wilkens R, Ruel J, Lombardero M, Vallery E (2000) Nitrogen budgets of phloem-feeding bark beetles with and without symbiotic fungi. Ecology 81:2198–2210
Adams AS, Six DL (2007) Temporal variation in mycophagy and prevalence of fungi associated with developmental stages of Dendroctonus ponderosae (Coleoptera: Curculionidae). Environ Entomol 36:64–72
Luchi N, Capretti P, Pinzani P, Orlando C, Pazzagli M (2005) Real-time PCR detection of Biscogniauxia mediterranea in symptomless oak tissue. Lett Appl Microbiol 41:61–68
Bahnweg G et al (2000) Controlled inoculation of Norway spruce (Picea abies) with Sirococcus conigenus: PCR-based quantification of the pathogen in host tissue and infection-related increase of phenolic metabolites. Trees-Struct Funct 14:435–441
Schena L, Nigro F, Ippolito A, Gallitelli D (2004) Real-time quantitative PCR: a new technology to detect and study phytopathogenic and antagonistic fungi. Eur J Plant Pathol 110:893–908
Karlsson M, Hietala A, Kvaalen H, Solheim H (2007) Quantification of host and pathogen DNA and RNA transcripts in the interaction of Norway spruce with Heterobasidion parviporum. Physiol Mol Plant Pathol 70:99–109
Schweigkofler W et al (2005) Detection and quantification of Leptographium wageneri, the cause of black-stain root disease, from bark beetles (Coleoptera: Scolytidae) in Northern California using regular and real-time PCR. Can J For Res 35:1798–1808
Carroll A, Taylor S, Régnière J, Safranyik L (2003) Effects of climate change on range expansion by the mountain pine beetle in British Columbia. In: Mountain pine beetle symposium: challenges and solutions Oct. 30–31. Kelowna, BC
Khadempour L, Massoumi Alamouti S, Hamelin RC, Bohlmann J, Breuil C (2010) Target-specific PCR primers can detect and differentiate ophiostomatoid fungi from microbial communities associated with the mountain pine beetle Dendroctonus ponderosae. Fungal Biol 114:825–833
Cullings KW (1992) Design and testing of a plant-specific PCR primer for ecological and evolutionary studies. Mol Ecol 1:233–240
West BT, Welch KB, Galecki AT (2006) Two-level models for clustered data: the rat pup example. In: West BT, Welch KB, Galecki AT (eds) Linear mixed models: a practical guide using statistical software. Chapman Hall/CRC, Boca Raton, p 376
Blom G (1958) Statistical estimates and transformed beta-variables. Wiley, New York
Holm S (1979) A simple sequentially rejective multiple test procedure. Scand J Stat 6:65–70
Raffa KF, Berryman A (1983) The role of host plant resistance in the colonization behavior and ecology of bark beetles (Coleoptera: Scolytidae). Ecological Monographs 53:27–49
Bleiker KP, Six DL (2009) Competition and coexistence in a multi-partner mutualism: interactions between two fungal symbionts of the mountain pine beetle in beetle-attacked trees. Microb Ecol 57:191–202 http://www.springerlink.com/index/10.1007/s00248-008-9395-6.
Lee S (2006) Fungi associated with the mountain pine beetle, Dendroctonous ponderosae. University of British Columbia, Vancouver
Upadhyay H (1981) A monograph of Ceratocystis and Ceratocystiopsis. University of Georgia Press, Athens
Gil-Serna J, González-Salgado A (2009) ITS-based detection and quantification of Aspergillus ochraceus and Aspergillus westerdijkiae in grapes and green coffee beans by real-time quantitative PCR. Int J Food Microbiol 131:162–167
Suarez MB et al (2005) Development of real-time PCR (TaqMan) assays for the detection and quantification of Botrytis cinerea in planta. Plant Physiol Biochem 43:890–899
Berbee ML, Taylor JW (1993) Dating the evolutionary radiations of the true fungi. Can J Bot 71:1114–1127
Massoumi Alamouti S et al (2011) Gene genealogies reveal cryptic species and host preferences for the pine fungal pathogen Grosmannia clavigera. Mol Ecol 20:2581–2602
Heid CA, Stevens J, Livak KJ, Williams PM (1996) Real time quantitative PCR. Genome Res 6:986–994
Gamper HA, Young JPW, Jones DL, Hodge A (2008) Real-time PCR and microscopy: are the two methods measuring the same unit of arbuscular mycorrhizal fungal abundance? Fungal Genet Biol 45:581–596
Solheim H (1995) Early stages of blue-stain fungus invasion of lodgepole pine sapwood following mountain pine-beetle attack. Can J Bot 73:70–74
Butler M, Day A (1998) Fungal melanins: a review. Can J Microbiol 44:1115–1136
Six DL, Bentz BJ (2007) Temperature determines symbiont abundance in a multipartite bark beetle–fungus ectosymbiosis. Microb Ecol 54:112–118
Acknowledgments
We thank Dr. J. McLean for insightful discussion on MPB physiology and ecology, and Siew Law, Lynette Lim, Heather Jack, and Renee Morton for technical assistance. This work was supported by grants to CB and JB from the Natural Sciences and Engineering Research Council of Canada (NSERC) and funds to CB and JB for the Tria Project (www.thetriaproject.ca) provided by the Province of British Columbia through Genome Canada, Genome British Columbia and by the Government of Alberta through Genome Alberta.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Cop. sp.1 described in this manuscript is probably the same as the one isolated in BC by Robinson (Bright and Stock 1982), we could not confirm this because, to our knowledge, the Robinson culture has not been preserved in a culture collection. Japanese C. minuta isolates from bark beetle galleries in spruce or pine have been shown to be non-pathogenic and to produce very small or no lesions in hosts (Lee et al. 2006). It is likely that Cop. sp.1 is not pathogenic to pine trees because it shows similar growth characteristics to the Japanese C. minuta, including, for example, slow growth, similar temperature requirements, cycloheximide sensitivity and mainly colonizing bark beetle galleries rather than phloem and sapwood. Compared to the MPB-associated staining fungi, Cop. sp.1 is difficult to detect using fungal isolation, since, on 1% MEA, this species grows at about 1mm/day in contrast to 25 mm/day for G. clavigera. Thus, Cop. sp.1 may have been considered unimportant in the MPB ecosystem, where fungal associates have been studied largely in relation to their effects on host trees (Yamaoka et al. 1995).
Fig. 1
(JPEG 48 kb)
Fig. 2
(JPEG 1522 kb)
Fig. 3
(JPEG 45 kb)
Rights and permissions
About this article
Cite this article
Khadempour, L., LeMay, V., Jack, D. et al. The Relative Abundance of Mountain Pine Beetle Fungal Associates Through the Beetle Life Cycle in Pine Trees. Microb Ecol 64, 909–917 (2012). https://doi.org/10.1007/s00248-012-0077-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-012-0077-z