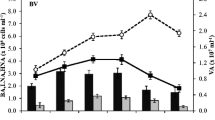
Abstract
To test if the quality and concentration of dissolved nitrogen (N) species could be a selective force in shaping bacterioplankton community structure, competition for various N compounds among five heterotrophic marine bacteria (Pseudomonas strains B, B25, and AX; Bacillus strain A6; Erythrobacter strain F19) was examined. Two of the five strains (AX and B25) were capable of utilizing urea for growth. The five strains were inoculated into dilute (1/1,000 strength) ZoBell medium enriched with various N sources (free amino acids, casein, ammonium, nitrate, or urea). Regardless of the added N source, the communities were either dominated by strain B (at 50 μM N) or strain AX (at 250 μM N). Without any addition of N, strain F19 dominated. If F19 was not included in the community, strain B25 dominated. Despite these differences in community structure, consumption of the added N compounds was surprisingly similar and no advantages of urea for the urea-utilizing bacterium B25 were obvious. To examine if urea could be of selective advantage to the urea-degrading strains B25 and AX, communities with and without B25 were amended with urea N. As expected, strain B25 became dominant when present, but without this strain the non-urea-utilizing strain B outcompeted the urea-utilizing strain AX. Possibly, strain B benefited from N released during catabolism of urea by strain AX. Changes in community composition did not result in major changes in the nitrogen dynamics. The results indicate that dissolved N species can be a selective force in shaping microbial communities. Relative to nutrient generalists, nutrient specialists may either have competitive advantages or stimulate growth of other species by synergetic interactions. Results from the model communities suggest that there may be a large degree of unpredictability in the making of microbial communities, whereas major ecosystem functions such as N cycling appear relatively stable.





Similar content being viewed by others
References
Azam F, Fenchel T, Field JG, Gray JS, Meyer-Reil L-A, Thingstad TF (1983) The ecological role of water-column microbes in the sea. Mar Ecol Prog Ser 10:257–263
Baas-Becking LGM (1934) Geobiologie of Inleiding Tot de Milieukunde. Van Stockkum & Zoon, The Hague
Bell T, Newman JA, Silverman BW, Turner SL, Lilley AK (2005) The contribution of species richness and composition to bacterial services. Nature 436:1157–1160
Cho BC, Park MG, Shim JH, Azam F (1996) Significance of bacteria in urea dynamics in coastal surface waters. Mar Ecol Prog Ser 142:19–26
Cole JR, Chai B, Farris RJ, Wang Q, Kulam SA, McGarrell DM, Garrity GM, Tiedje JM (2005) The Ribosomal Database Project (RDP-II): sequences and tools for high-throughput rRNA analysis. Nucleic Acids Res 33:D294–D296
Cottrell MT, Kirchman DL (2000) Natural assemblages of marine proteobacteria and members of the Cytophaga–Flavobacter cluster consuming low- and high-molecular-weight dissolved organic matter. Appl Environ Microbiol 66:1692–1697
del Giorgio PA, Gasol JM, Vaque D, Mura P, Agusti S, Duarte CM (1996) Bacterioplankton community structure: protists control net production and the proportion of active bacteria in a coastal marine community. Limnol Oceanogr 41:1169–1179
Denner EBM, Vybiral D, Koblizek M, Kampfer P, Busse HJ, Velimirov B (2002) Erythrobacter citreus sp. nov., a yellow-pigmented bacterium that lacks bacteriochlorophyll a, isolated from the western Mediterranean Sea. Int J Syst Evol Microbiol 52:1655–1661
Docherty KM, Young KC, Maurice PA, Bridgham SD (2006) Dissolved organic matter concentration and quality influences upon structure and function of freshwater microbial communities. Microb Ecol 52:378–388
Eiler A, Langenheder S, Bertilsson S, Tranvik LJ (2003) Heterotrophic bacterial growth efficiency and community structure at different natural organic carbon concentrations. Appl Environ Microbiol 69:3701–3709
Fagerbakke KM, Heldal M, Norland S (1996) Content of carbon, nitrogen, oxygen, sulfur and phosphorus in native aquatic and cultured bacteria. Aquat Microb Ecol 10:15–27
Fernandez A, Huang SY, Seston S, Xing J, Hickey R, Criddle C, Tiedje J (1999) How stable is stable? Function versus community composition. Appl Environ Microbiol 65:3697–3704
Findlay SEG, Sinsabaugh RL, Sobczak WV, Hoostal M (2003) Metabolic and structural response of hyporheic microbial communities to variations in supply of dissolved organic matter. Limnol Oceanogr 48:1608–1617
Frette L, Johnsen K, Jørgensen NOG, Nybroe O, Kroer N (2004) Functional characteristics of culturable bacterioplankton from marine and estuarine environments. Int Microbiol 7:219–227
Gontang EA, Fenical W, Jensen PR (2007) Phylogenetic diversity of gram-positive bacteria cultured from marine sediments. Appl Environ Microbiol 73:3272–3282
Gray ND, Head IM (2001) Linking genetic identity and function in communities of uncultured bacteria. Environ Microbiol 3:481–492
Hareland WA, Crawford RL, Chapman PJ, Dagley S (1975) Metabolic function and properties of 4-hydroxyphenylacetic acid 1-hydroxylase from Pseudomonas acidovorans. J Bact 121:272–285
Johnsen K, Enger O, Jacobsen CS, Thirup L, Torsvik V (1999) Quantitative selective PCR of 16S ribosomal DNA correlates well with selective agar plating in describing population dynamics of indigenous Pseudomonas spp. in soil hot spots. Appl Environ Microbiol 65:1786–1788
Jørgensen NOG (2006) Uptake of urea by estuarine bacteria. Aquat Microb Ecol 42:227–243
Jørgensen NOG, Jacobsen CS (1996) Bacterial uptake and utilization of dissolved DNA. Aquat Microb Ecol 11:263–270
Jørgensen NOG, Jensen RE (1997) Determination of dissolved combined amino acids using microwave-assisted hydrolysis and HPLC precolumn derivatization for labeling of primary and secondary amines. Mar Chem 57:287–297
Jørgensen NOG, Kroer N, Coffin RB, Yang XH, Lee C (1993) Dissolved free amino acids, combined amino acids, and DNA as sources of carbon and nitrogen to marine bacteria. Mar Ecol Prog Ser 98:135–148
Kandel A, Christoffersen K, Nybroe O (1993) Filtration rates of Daphnia cucullata on Alcaligenes eutrophus JMP134 estimated by a fluorescent antibody method. FEMS Microbiol Ecol 12:1–8
Kirchman DL, Keil RG, Wheeler PA (1989) The effect of amino acids on ammonium utilization and regeneration by heterotrophic bacteria in the subarctic Pacific. Deep-Sea Res 36:1763–1776
Kirchman DL, Dittel AI, Findley SEG, Fischer D (2004) Changes in bacterial activity and community structure in response to dissolved organic matter in the Hudson River, New York. Aquat Microb Ecol 35:257
Kirchman DL, Elifantz H, Dittel AI, Malmstrom RR, Cottrell MT (2007) Standing stocks and activity of Archaea and Bacteria in the western Arctic Ocean. Limnol Oceanogr 52:495–507
Koch AL (1996) What size should a bacterium be? A question of scale. Annu Rev Microbiol 50:317–348
Kroer N, Jørgensen NOG, Coffin RB (1994) Utilization of dissolved nitrogen by heterotrophic bacterioplankton: a comparison of three ecosystems. Appl Environ Microbiol 60:4116–4123
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, New York, pp 115–175
Langenheder S, Lindstrom ES, Tranvik LJ (2005) Weak coupling between community composition and functioning of aquatic bacteria. Limnol Oceanogr 50:957–967
Langenheder S, Lindstrom ES, Tranvik LJ (2006) Structure and function of bacterial communities emerging from different sources under identical conditions. Appl Environ Microbiol 72:212–220
Lindroth P, Mopper K (1979) High performance liquid chromatographic determination of subpicomole amounts of amino acids by precolumn fluorescence derivatization with o-phthaldialdehyde. Anal Chem 51:1667–1674
Loreau M, Naeem S, Inchausti P, Bengtsson J, Grime JP, Hector A, Hooper DU, Huston MA, Raffaelli D, Schmid B, Tilman D, Wardle DA (2001) Ecology-biodiversity and ecosystem functioning: current knowledge and future challenges. Science 294:804–808
Madigan MT, Martinko JM (2006) BROCK biology of microorganisms, 11th edn. Prentice Hall, Upper Saddle River
Massana R, Gasol JM, Bjørnsen PK, Blackburn N, Hagström A, Hietanen S, Hygum BH, Kuparinen J, Pedros-Alio C (1997) Measurement of bacterial size via image analysis of epifluorescence preparations: description of an inexpensive system and solutions to some of the most common problems. Scientia Marina 61:397–407
Middelboe M, Borch NH, Kirchman DL (1995) Bacterial utilization of dissolved free amino acids, dissolved combined amino acids and ammonium in the Delaware Bay estuary: effects of carbon and nitrogen limitation. Mar Ecol Prog Ser 128:109–120
Middelboe M, Jørgensen NOG, Kroer N (1996) Effects of viruses on nutrient turnover and growth efficiency of noninfected marine bacterioplankton. Appl Environ Microbiol 62:1991–1997
Pedersen AGU, Thomsen TR, Lomstein BAA, Jørgensen NOG (2001) Bacterial influence on amino acid enantiomerization in a coastal marine sediment. Limnol Oceanogr 46:1358–1369
Price NM, Harrison PJ (1987) Comparison of methods for the analysis of dissolved urea in seawater. Mar Biol 94:307–317
Rubin MA, Leff LG (2007) Nutrients and other abiotic factors affecting bacterial communities in an Ohio River (USA). Microb Ecol 54:374–382
Spiers AJ, Buckling A, Rainey PB (2000) The causes of Pseudomonas diversity. Microbiol 146:2345–2350
Szabó KÉ, Itor POB, Bertilsson S, Tranvik LJ, Eiler A (2007) Importance of rare and abundant populations for the structure and functional potential of freshwater bacterial communities. Aquat Microb Ecol 47:1–10
Vogels GD, van der Drift C (1976) Degradation of purines and pyrimidines by microorganisms. Bacteriol Rev 40:403–468
Worm J, Nybroe O (2001) Input of protein to lake water microcosms affects expression of proteolytic enzymes and the dynamics of Pseudomonas spp. Appl Environ Microbiol 67:4955–4962
Worm J, Jensen LE, Hansen TS, Søndergaard M, Nybroe O (2000) Interactions between proteolytic and non-proteolytic Pseudomonas fluorescens affect protein degradation in a model community. FEMS Microbiol Ecol 32:103–109
ZoBell C (1946) Marine microbiology; a monograph on hydrobacteriology. Chronica Botanica Co., Waltham
Acknowledgements
We thank H. Irming, M.-B. Prahm, R. E. Jensen, and A.-S. B. Hentze for technical assistance, E. Rochelle-Newall for valuable discussions, and four reviewers for constructive criticism. S. Sørensen and J. Worm kindly assisted with the flow cytometry analysis. This work was supported by Grant 9601731 from the Danish Natural Science Research Council.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Frette, L., Jørgensen, N.O.G., Nybroe, O. et al. Effect of Availability of Nitrogen Compounds on Community Structure of Aquatic Bacteria in Model Systems. Microb Ecol 57, 104–116 (2009). https://doi.org/10.1007/s00248-008-9404-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-008-9404-9