Abstract
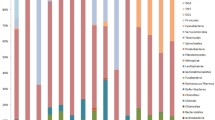
In search for microbiological indicators of coral health and coral diseases, community profiles of coral-associated epizoic prokaryotes were investigated because of their dual potential as a source of coral pathogens and their antagonists. In pairwise samples of visually healthy and diseased coral specimens from Bolinao Bay (Pangasinan, Philippines), mixed biofilm communities of ectoderm- and mucus-colonizing epizoic prokaryotes were compared using fluorescent in situ hybridization (FISH). Oligonucleotide probes targeted 13 phylotypes representing the main taxonomic groups of marine prokaryotes. Coral taxa tended to show specific community profiles. An attempt to separate the profiles of healthy and diseased specimens by applying principal component analysis (PCA) to a (nonselective) collection of corals (affected by various diseases) proved unsuccessful. On the other hand, separate PCA clusters were obtained from healthy and diseased corals belonging to a single species (Pocillopora damicornis) only. This cluster formation was dominated by principal component 1 with the genus Vibrio accounting for 18%. At the same time, reef-site-specific clusters were formed as well. At a reef site exposed to pollution from intensive fish cage (Chanos chanos) farming, healthy P. damicornis were mainly (93%) colonized by unicellular cyanobacteria. The formal calculation of diversity parameters suggested that evenness in particular was driven by both health status and reef site location. Despite the low resolution of taxonomic levels achieved with FISH probes targeting only large phylotype groups, significant differences between healthy and diseased corals and also between polluted and nonpolluted reef sites were observed.







Similar content being viewed by others
References
Ainsworth TD, Fine M, Blackall LL, Hoegh-Guldberg O (2006) Fluorescence in situ hybridization and spectral imaging of coral-associated bacterial communities. Appl Environ Microbiol 72:3016–3020
Amann R, Ludwig W, Schleifer K (1995) Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microb Rev 59:143–169
Ben-Haim Y, Thompson FL, Thompson CC, Cnockaert MC, Hoste B, Swings J, Rosenberg E (2003a) Vibrio coralliilyticus sp nov., a temperature dependent pathogen of the coral Pocillopora damicornis. Int J Syst Evol Microbiol 53:309–315
Ben-Haim Y, Zicherman-Keren M, Rosenberg E (2003b) Temperature related bleaching and lysis of the coral Pocillopora damicornis by the novel pathogen Vibrio coralliilyticus. Appl Environ Microbiol 69:4236–4242
Bourne D, Munn C (2005) Diversity of bacteria associated with the coral Pocillopora damicornis from the Great Barrier Reef. Environ Microbiol 7:1162–1174
Bruno JF, Selig ER (2007) Regional decline of coral cover in the Indo-Pacific: timing, extent, and subregional comparisons. PLoS Biology 2:e711 DOI 10.1371/journal.pone.0000711
Cervino JM, Hayes RL, Polson PW, Polson SC, Goreau TJ, Martinez RJ, Smith GW (2004) Relationship of Vibrio species infection and elevated temperature to yellow blotch/band disease in Caribbean corals. Appl Environ Microbiol 70:6855–6864
Cooney R, Pantos O, Le Tassier MDA, Barer MR, O’Donnell AG, Bythell JC (2002) Characterization of the bacterial consortium associated with black band disease in coral using molecular microbiological techniques. Environ Microbiol 4:401–413
Das S, Lyla PS, Khan SA (2006) Marine microbial diversity and ecology: importance and future perspectives. Current Science 90:1325–1335
Denner E, Smith G, Busse H, Schumann P, Narzt T, Polson SW, Lubitz W, Richardson L (2003) Aurantimonas coralicida gen nov sp nov, the causative agent of white plague type II in Caribbean scleractinian corals. Int J Sys Evol Microbiol 53:1115–1122
Dove SG, Hoegh-Guldberg O, Rangananthan S (2001) Major colour patterns of reef-building corals are due to a family of GFP-like proteins. Coral Reefs 19:197–204
Ducklow HW, Mitchell M (1979) Composition of mucus released by coral reef coelenterates. Limnol Oceanogr 24:706–714
Fine M, Loya Y (2004) Coral bleaching in a temperate sea: from colony physiology to population ecology. In: Rosenberg E, Loya Y (eds) Coral health and disease. Springer, Berlin, pp 143–156
Frias-Lopez J, Zerkle A, Bonheyo G, Fouke W (2002) Partitioning of bacterial communities between seawater and healthy, black band diseased, and dead coral surfaces. Appl Environ Microbiol 68:2214–2228
Gil-Agudelo DL, Myers C, Smith GW, Kim K (2006) Changes in the microbial communities associated with Gorgonia ventalina during aspergillosis infection. Dis Aquat Org 69:89–94
Giovannoni S, Rappe M (2000) Evolution, diversity, and molecular ecology of marine prokaryotes. In: Kirchman DL (ed) Microbial ecology of the oceans. Wiley-Liss, New York, pp 47–84
Harvell D, Aronson R, Baron N, Connell J, Dobson A, Ellner S, Gerber L, Kim K, Kuris A, McCallum H, Lafferty K, McKay B, Porter J, Pascual M, Smith G, Sutherland K, Ward J (2004) The rising tide of ocean diseases: unsolved problems and research priorities. Front Ecol Environ 2:375–382
Hoegh-Guldberg O (2004) Coral reefs and projections of future change. In: Rosenberg E, Loya Y (eds) Coral health and disease. Springer, Berlin, pp 463–484
Jokiel PL (2004) Temperature stress and coral bleaching. In: Rosenberg E, Loya Y (eds) Coral health and disease. Springer, Berlin, pp 401–426
Kellogg C (2004) Tropical Archaea: diversity associated with the surface microlayer of corals. Mar Ecol Prog Ser 273:81–88
Klaus JS, Janse I, Heikoop JM, Sandford RA, Fouke BW (2007) Coral microbial communities, zooxanthellae and mucus along gradients of seawater depth and coastal pollution. Environ Microbiol 9:1291–1305
Kobayashi M, Ishizuka T, Katayama M, Kanehisa M, Bhattacharyya-Pakrasi M, Pakrasi HB, Ikeuchi M (2004) Response to oxidative stress involves a novel peroxiredoxin gene in the unicellular cyanobacterium Synechocystis sp. PCC 6803. Plant Cell Physiol 45:290–299
Kooperman N, Ben-Dov E, Kramarsky-Winter E, Barak Z, Kushmaro A (2007) Coral mucus-associated bacterial communities from natural and aquarium environments. FEMS Microbiol Lett 276:106–113
Koren O, Rosenberg E (2006) Bacteria associated with mucus and tissues of the coral Oculina patagonica in summer and winter. Appl Environ Microbiol 72:5254–5259
Kushmaro A, Banin E, Loya Y, Stackebrandt E, Rosenberg E (2001) Vibrio shiloi sp nov, the causative agent of bleaching of the coral Oculina patagonica. Int J Syst Evol Microbiol 51:1383–1388
Lafti A, Jeanjean R, Lemeille S, Havaux M, Zhang C-C (2005) Iron starvation leads to oxidative stress in Anabaena sp. strain PCC 7120. J Bacteriol 187:6596–6598
Lesser M, Mazel C, Gurbunov M, Falkowski PG (2004) Discovery of symbiotic nitrogen-fixing cyanobacteria in corals. Science 305:997–1000
Loya Y (2004) The coral reefs of Eilat—past, present, future: three decades of coral community structure studies. In: Rosenberg E, Loya Y (eds) Coral health and disease. Springer, Berlin, pp 1–34
Pantos O, Bythell JC (2006) Bacterial community structure associated with white band disease in the elkhorn coral Acropora palmata determined using culture-independent 16S rRNA techniques. Dis Aquat Org 69:79–88
Pantos O, Cooney R, Le Tissler MDA, Barer MR, O’Donnell AG, Bythell JC (2003) The bacterial ecology of a plague-like disease affecting the Caribbean coral Montastrea annularis. Environ Microbiol 5:370–382
Patterson KL, Porter JW, Ritchie KB, Polson SW, Mueller E, Peters EC, Santavy DL, Smith GW (2002) The etiology of white pox, a lethal disease of the Caribbean elkhorn coral, Acropora palmata. Proc Natl Acad Sci U S A 99:8725–8730
Pernthaler J, Glockner FO, Schoenhuber W, Amann R (2001) Fluorescence in situ hybridization (FISH) with rRNA-targeted oligonucleotide probes. Methods Microbiol 30:207–226
Raymundo L, Rosell K, Reboton C, Kaczmarsky L (2005) Coral diseases on Philippine reefs: genus Porites is a dominant host. Dis Aquat Org 64:181–191
Reshef L, Koren O, Loya Y, Zilber-Rosenberg I, Rosenberg E (2006) The coral probiotic hypothesis. Environ Microbiol 8:2068–2073
Ritchie KB (2006) Regulation of microbial populations by coral surface mucus and mucus associated bacteria. Mar Ecol Prog Ser 322:1–14
Ritchie KB, Smith GW (1995) Preferential carbon utilization by surface bacterial communities from water mass, normal, and white-band diseased Acropora cervicornis. Mol Mar Biol 4:345–352
Rohwer F, Breitbart M, Jara J, Azam F, Knowlton N (2001) Diversity of bacteria associated with the Caribbean coral Montastrea franksi. Coral Reefs 20:85–91
Rohwer F, Seguritan V, Azam F, Knowlton N (2002) Diversity and distribution of coral associated bacteria. Mar Ecol Prog Ser 243:1–10
Rosenberg E, Ben-Haim Y (2002) Microbial diseases of corals and global warming. Environ Microbiol 4:318–328
Rosenberg E, Koren O, Reshef L, Efrony R, Zilber-Rosenberg I (2007) The role of microorganisms in coral health, disease and evolution. Nature Rev Microbiol 5:355–361
Santiago-Vasquez LZ, Bruck TB, Bruck WM, Duque-Alarcon AP, McCarthy PJ, Kerr RG (2007) The diversity of the bacterial communities associated with the azooxanthellate hexacoral Cirrhipathes lutkeni. ISME J 1:654–659
Sekar R, Mills DK, Remily ER, Voss JD, Richardson LL (2006) Microbial communities in the surface mucopolysaccharide layer and the black band microbial mat of black band-diseased Siderastrea siderea. Appl Environ Microbiol 72:5963–5973
Snaidr J, Amann R, Huber I, Ludwig W, Schleifer K-H (1997) Phylogenetic analysis and in situ identification of bacteria in activated sludge. Appl Environ Microbiol 63:2884–2896
Sutherland K, Porter J, Torres C (2004) Disease and immunity in Caribbean and Indo-Pacific zooxanthellae corals. Mar Ecol Prog Ser 266:273–302
Suzuki M, Sherr E, Sherr B (1993) DAPI direct counting underestimates bacterial abundances and average cell size compared to AO direct counting. Limnol Oceanogr 38:1566–1570
Trousselier M, Legendre P (1981) A functional evenness index for microbial ecology. Microb Ecol 7:283–296
Wagner N, Horny M, Daims H (2003) Fluorescence in situ hybridisation for the identification and characterisation of prokaryotes. Curr Opin Microbiol 6:302–309
Webster N, Bourne D (2007) Bacterial community structure associated with the Antarctic soft coral, Alcyonium antarcticum. FEMS Microbiol Ecol 59:81–94
Wegley L, Yu Y, Breitbart M, Casas V, Kline DI, Rohwer F (2004) Coral associated Archaea. Mar Ecol Prog Ser 273:89–96
Yang X, Shimizu Y, Steiner JR, Clardy J (1993) Nostoclides I and II, extracellular metabolism from a symbiotic cyanobacterium, Nostoc sp. Tetrahedron Lett 34:761–764
Zak JC, Willig MR, Moorhead DL, Wildman HG (1994) Functional diversity of microbial communities: a quantitative approach. Soil Biol Biochem 26:1101–1108
Zhu J, Miller MB, Vance RE, Dziejman M, Bassler BL, Mekalanos JJ (2002) Quorum-sensing regulators control virulence gene expression in Vibrio cholerae. Proc Natl Acad Sci U S A 99:3129–3134
Acknowledgements
Authors thank H.P. Grossart for an introduction to the FISH technique and H Dries for her help in collecting samples from corals in situ. This investigation was supported by a Center of Excellence grant to W.R. in the framework of an international GEF/World Bank funded Coral Reef Project, as well as support from DAAD (German Academic Exchange Service) to W.R. and H.P.G.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arboleda, M., Reichardt, W. Epizoic Communities of Prokaryotes on Healthy and Diseased Scleractinian Corals in Lingayen Gulf, Philippines. Microb Ecol 57, 117–128 (2009). https://doi.org/10.1007/s00248-008-9400-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-008-9400-0