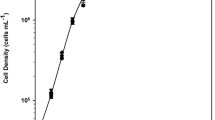
Abstract
Ecophysiological investigations on the salinity acclimation of the cyanobacterium Prochlorothrix hollandica SAG 10.89 led to significantly revised salinity tolerance limits. Besides potential effects of cultivation techniques, clear ion composition effects mainly explain formerly described hypersensitivity to NaCl-mediated salinity and lack of osmolyte detection. An extraordinarily broad plasticity of cellular chlorophyll a/b ratios occurred with variations of NaCl-induced salinity. Photosynthesis characteristics, pigment regulation, respiration, and biomass yield in growth medium with field-like ion composition indicated generally reduced acclimation pressure. A simultaneously significant increase in osmolyte (sucrose) accumulation indicated more efficient osmotic acclimation. Minor growth inhibition up to salinities of 10 practical salinity units enlarged the potential habitat of P. hollandica but at the most to about 300,000 km2 in the Baltic Sea. This supports probable observations of Prochlorothrix sp. in phytoplankton assemblages of open waters in Baltic Sea-monitoring studies. Brackish habitats differ from so far known habitats of Prochlorothrix spp. in turbidity, productivity, and plankton composition. Adjusted physiological features dispel fundamental doubts on the establishment of filamentous prochlorophytes in brackish waters.






Similar content being viewed by others
References
Allakhverdiev SI, Nishiyama Y, Suzuki, I, Tasaka Y, Murata N (1999) Genetic engineering of the unsaturation of fatty acids in membrane lipids alters the tolerance of Synechocyctis to salt stress. Proc Natl Acad Sci USA 96:5862–5867
Allakhverdiev SI, Sakamoto A, Nishiyama Y, Inaba M, Murata N (2000) Ionic and osmotic effects of NaCl-induced inactivation of photosystems I and II in Synechococcus sp. Plant Physiol 123:1047–1056
Andrizhiyevskaya EG, Schwabe TME, Germano M, D’Haene S, Kruip J, van Grondelle R, Dekker JP (2002) Spectroscopic properties of PSI-IsiA supercomplexes from the cyanobacterium Synechococcus PCC 7942. Biochim Biophys Acta 1556:265–272
BACC (2006) Assessment of climate change for the Baltic Sea basin—the BACC project. Int Conf, 22–23 May 2006, Göteborg, Sweden. Chapter summaries, available at: http://dvsun3.gkss.de/ BACC/
Bumba L, Prasil O, Vacha F (2005) Antenna ring around trimeric Photosystem I in chlorophyll b containing cyanobacterium Prochlorothrix hollandica. Biochim Biophys Acta 1708:1–5
Burger-Wiersma T, Veenhuis M, Korthals HJ, van der Wiel CCM, Mur LR (1986) A new prokaryote containing chlorophylls a and b. Nature 320:262–264
Burger-Wiersma T, Stal LJ, Mur LR (1989) Prochlorothrix hollandica. gen. nov., sp. nov., a filamentous oxygenic photoautotrophic prokaryote containing chlorophylls a and b: assignment to Prochlorotrichaceae fam. nov. and order Prochlorales Florenzano, Balloni, and Materassi 1986, with emendation of the ordinal description. Int J Syst Bacteriol 39:250–257
Burger-Wiersma T, Post AF (1989) Functional analysis of the photosynthetic apparatus of Prochlorothrix hollandica (Prochlorales), a chlorophyll b containing prokaryote. Plant Physiol 91:770–774
Burger-Wiersma T, Matthijs HCP (1990) The biology of the prochlorales. In: Codd GA (ed) Autotrophic microbiology and one-carbon metabolism. Kluwer, Dordrecht, pp 1–24
Chisholm SW, Olsen RJ, Zettler ER, Waterbury JB, Goericke, R, Welschmeyer N (1988) A novel free-living prochlorophyte abundant in the oceanic euphotic zone. Nature 334:340–343
Cunningham FX, Gantt E (1998) Genes and enzymes of carotenoid biosynthesis in plants. Annu Rev Plant Physiol Plant Mol Biol 49:557–583
Demmig-Adams B (1990) Carotenoids and photoprotection in plants: a role for the xanthophyll zeaxanthin. Biochim Biophys Acta 1020:1–24
Erdmann N (1983) Organic osmoregulatory solutes in blue-green algae. Z Pflanzenphysiol 110:147–155
Erdmann N, Hagemann M, Berg C, Fulda S (1990) Basis of salt adaptation in cyanobacteria. Limnologica (Berlin) 20:57–60
Ferjani A, Mustardy L, Sulpice R, Marin K, Isuzuki I, Hagemann M, Murata N (2003) Glucosylglycerol, a compatible solute, sustains cell division under salt stress. Plant Physiol 131(4):1628–1637
Geiß U, Bergmann I, Blank M, Schumann R, Hagemann M, Schoor A (2003) Detection of Prochlorothrix in brackish waters by specific amplification of pcb genes. Appl Environ Microbiol 69:6243–6249
Golden SS, Morden CW, Greer KL (1993) Comparison of sequences and organization of photosynthesis genes among the prochlorophyte Prochlorothrix hollandica, cyanobacteria, and chloroplasts. In: Lewin RA (ed) Origins of plastids: symbiogenesis, prochlorophytes and the origins of chloroplasts. Chapman & Hall, New York, pp 141–158
Golecki JR, Jürgens UJ (1989) Ultrastructural studies on the membrane systems and cell inclusions of the filamentous prochlorophyte Prochlorothrix hollandica. Arch Microbiol 152:77–82
Hagemann M, Fulda S, Schubert H (1994) DNA, RNA and protein synthesis in the cyanobacterium Synechocystis sp. PCC 6803 adapted to different salt concentrations. Cur Microbiol 28:201–207
Hincha DK, Hagemann M (2004) Stabilization of model membranes during drying by compatible solutes involved in the stress tolerance of plants and microorganisms. Biochem J 383:277–283
Ihalainen JA, D’Haene S, Yeremenko N, van Roon H, Arteni AA, Boekema EJ, van Grondelle R, Matthijs HC, Dekker JP (2005) Aggregates of the chlorophyll-binding protein IsiA (CP43')dissipate energy in cyanobacteria. Biochemistry 44:10846–10853
Jakobsen TS, Hansen PB, Jeppesen E, Søndergaard M (2004) Cascading effect of three-spined stickleback Gasterosteus aculeatus on community composition, size, biomass and diversity of phytoplankton in shallow, eutrophic brackish lagoons. Mar Ecol Prog Ser 279:305–309
Joset F, Jeanjean R, Hagemann M (1996) Dynamics of the response of cyanobacteria to salt stress: deciphering the molecular events. Physiol Plant 96:738–744
Kirst GO (1979) Osmotische Adaptation der marinen Planktonalge Platymonas subcordiformis. Ber Deutsch Bot Ges 92:31–42
Lefort-Tran M, Pouphile M, Spath S, Packer L (1988) Cytoplasmic membrane changes during adaptation of the fresh water cyanobacterium Synechococcus 6311 to salinity. Plant Physiol 87:767–775
Lewin RA (1975) A marine Synechocystis (Cyanophyta, Chlorococcales) epizoic on ascidians. Phycol 14:153–169
Lewin RA (1976) Prochlorophyta as a proposed new division of algae. Nature 261:697–698
Lewin RA (1977) Prochloron, type genus of the Prochlorophyta. Phycologia 16:217
Mathews-Roth MM (1987) Photoprotection by carotenoids. Fed Proc 46:1890–1893
Nishiyama Y, Los DA, Murata N (1999) PsbU, a protein associated with photosystem II, is required for the acquisition of cellular thermotolerance in Synechococcus sp. PCC 7002. Plant Physiol 120:176–182
Oren A (1999) Bioenergetic aspects of halophilism. Microbiol Mol Biol Rev 63:334–348
Partensky F, Hess WR, Vaulot D (1999) Prochlorococcus, a marine photosynthetic prokaryote of global significance. Microbiol Mol Biol Rev 63:106–127
Pinevich AV, Skulberg OM, Matthijs HCP, Schubert H, Willen E, Gavrilova OV, Velichko, NV (1999) Characterisation of a novel Chlorophyll b-containing Prochlorothrix species (Prochlorophyta) and its photosynthetic apparatus. Microbios 100:159–174
Platt T, Gallegos CL, Harrison WG (1980) Photoinhibition of photosynthesis in natural assemblages of marine phytoplankton. J Mar Res 38:687–701
Post AF, Arieli B (1997) Photosynthesis of Prochlorothrix hollandica under sulfide-rich anoxic conditions. Appl Environ Microbiol 63:3507–3511
Provasoli L (1968) Media and prospect for the cultivation of marine algae. In: Watanabe A, Hattori A (eds) Cultures and collections of algae. Proceedings of US–Japan Conference, Hakone, Sept. 1966. The Japanese Society of Plant Physiologists, Hakone, Japan, pp 63–75
Reed RH, Stewart WDP (1985) Osmotic adjustment and organic solute accumulation in unicellular cyanobacteria from freshwater and marine habitats. Mar Biol 88:1–9
Reed RH, Stewart WDP (1988) The responses of cyanobacteria to salt stress. In: Rogers LJ, Gallon JR (eds) Biochemistry of the algae and cyanobacteria, vol. 12. Clarendon, Oxford, pp 217–231
Rippka R, Deruelles J, Waterbury JB, Herdmann M, Stainer RY (1979) Generic asignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
Scanlan DJ, West NJ (2002) Molecular ecology of the marine cyanobacterial genera Prochlorococcus and Synechococcus. FEMS Microbiol Ecol 40(1):1–12
Schoor A, Erdmann N, Effmert U, Mikkat S (1995) Determination of the cyanobacterial osmolyte glycosylglycerol by high-performance liquid chromatography. J Chromatogr A 704:89–97
Schoor A, Hagemann M, Erdmann N (1999) Glucosylglycerol-phosphate synthase: target for ion-mediated regulation of osmolyte synthesis in the cyanobacterium Synechocystis sp. strain PCC 6803. Arch Microbiol 171:101–106
Schubert H, Fulda S, Hagemann M (1993) Effects of adaptation to different salt concentrations on photosynthesis and pigmentation of the cyanobacterium Synechocystis sp. PCC 6803. Plant Physiol 142:291–295
van den Hoek C, Jahns HM, Mann DG (eds) (1993) Algen. Thieme, Stuttgart, pp 15–34
van der Staay GWM, Brouwer A, Baard RL, van Mourik F, Matthijs HCP (1992) Separation of photosystems I and II from the oxychlorobacterium (prochlorophyte) Prochlorothrix hollandica and association of chlorophyll b binding antennae with photosystem II. Biochim Biophys Acta 1102:220–228
Walsby AE (1997) Numerical integration of phytoplankton photosynthesis through time and depth in a water column. New Phytol 136:189–209
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. Plant Physiol 144:307–313
Wolfstein K, Hartig P (1998) The photosynthetic light dispensation system: application to microphytobenthic primary production measurements. Mar Ecol Prog Ser 166:63–71
Yeremenko N, Kouril R, Ihalainen JA, D’Haene S, van Oosterwijk N, Andrizhiyevskaya EG, Keegstra W, Dekker HL, Hagemann M, Boekema EJ, Matthijs HC, Dekker JP (2004) Supramolecular organization and dual function of the IsiA chlorophyll-binding protein in cyanobacteria. Biochemistry 43:10308–10313
Acknowledgments
We thank S. Görs for a chromatographic verification of sucrose accumulation. The detailed and helpful suggestions of an anonymous reviewer are kindly acknowledged. This work was partially supported by a grant of the Deutsche Forschungsgemeinschaft (DFG).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bergmann, I., Geiß-Brunschweiger, U., Hagemann, M. et al. Salinity Tolerance of the Chlorophyll b-synthesizing Cyanobacterium Prochlorothrix hollandica Strain SAG 10.89. Microb Ecol 55, 685–696 (2008). https://doi.org/10.1007/s00248-007-9311-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-007-9311-5