Abstract
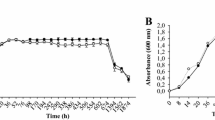
The biological control bacterium Pseudomonas chlororaphis (aureofaciens) strain 30-84 employs two quorum sensing (QS) systems: PhzR/PhzI regulates the production of the antibiotics phenazine-1-carboxylic acid, 2-hydroxy-phenazine-1-carboxylic acid, and 2-hydroxy-phenazine, whereas CsaR/CsaI regulates currently unknown aspects of the cell surface. Previously characterized derivatives of strain 30-84 with mutations in each QS system and in the phenazine biosynthetic genes were screened for their ability to form surface-attached biofilm populations in vitro, using microtiter plate and flow cell biofilm assays, and on seeds and roots. Results from in vitro, seed, and root studies demonstrated that the PhzR/PhzI and the CsaR/CsaI QS regulatory systems contribute to the establishment of biofilms, with mutations in PhzR/PhzI having a significantly greater effect than mutations in CsaR/CsaI. Interestingly, phenazine antibiotic production was necessary for biofilm formation to the same extent as the PhzR/PhzI QS system, suggesting the loss of phenazines was responsible for the majority of the biofilm defect in these mutants. In vitro analysis indicated that genetic complementation or AHL addition to the growth medium restored the ability of the AHL synthase phzI mutant to form biofilms. However, only phenazine addition or genetic complementation of the phenazine biosynthetic mutation in trans restored biofilm formation by mutants defective in the transcriptional activator phzR or the phzB structural mutant. QS and phenazine production were also involved in the establishment of surface-attached populations on wheat seeds and plant roots, and, as observed in vitro, the addition of AHL extracts restored the ability of phzI mutants, but not phzR mutants, to form surface attached populations on seeds. Similarly, the presence of the wild type in mixtures with the mutants restored the ability of the mutants to colonize wheat roots, demonstrating that AHL and/or phenazine production by the wild-type population could complement the AHL- and phenazine-deficient mutants in situ. Together, these data demonstrate that both QS systems are involved in the formation of surface-attached populations required for biofilm formation by P. chlororaphis strain 30-84, and indicate a new role for phenazine antibiotics in rhizosphere community development beyond inhibition of other plant-associated microorganisms.





Similar content being viewed by others
References
Abken, HJ, Tietze, M, Brodersen, J, Baumer, S, Beifuss, U, Deppenmeier, U (1998) Isolation and characterization of methanophenazine and function of phenazines in membrane-bound electron transport of Methanosarcina mazei Go1. J Bacteriol 180: 2027–2032
Arevalo-Ferro, C, Reil, G, Gorg, A, Eberl, L, Riedel, K (2005) Biofilm formation of Pseudomonas putida IsoF: the role of quorum sensing as assessed by proteomics. Syst Appl Microbiol 28: 87–114
Bauer, WD, Mathesius, U (2004) Plant responses to bacterial quorum sensing signals. Curr Opin Plant Biol 7: 429–433
Beloin, C, Ghigo, JM (2005) Finding gene-expression patterns in bacterial biofilms. Trends Microbiol 13: 16–19
Cao, H, Baldini, RL, Rahme, LG (2001) Common mechanisms for pathogens of plants and animals. Annu Rev Phytopathol 39: 259–284
Cha, C, Gao, P, Chen, YC, Shaw, PD, Farrand, SK (1998) Production of acyl-homoserine lactone quorum-sensing signals by gram-negative plant-associated bacteria. Mol Plant Microbe Interact 11: 1119–1129
Christensen, BB, Sternberg, C, Andersen, JB, Palmer Jr, RJ, Nielsen, AT, Givskov, M, Molin, S (1999) Molecular tools for study of biofilm physiology. Methods Enzymol 310: 20–42
Cucarella, C, Solano, C, Valle, J, Amorena, B, Lasa, I, Penades, JR (2001) Bap, a Staphylococcus aureus surface protein involved in biofilm formation. J Bacteriol 183: 2888–2896
Davey, ME, Caiazza, NC, O'Toole, GA (2003) Rhamnolipid surfactant production affects biofilm architecture in Pseudomonas aeruginosa PAO1. J Bacteriol 185: 1027–1036
Davies, DG, Parsek, MR, Pearson, JP, Iglewski, BH, Costerton, JW, Greenberg, EP (1998) The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science 280: 295–298
Deziel, E, Comeau, Y, Villemur, R (2001) Initiation of biofilm formation by Pseudomonas aeruginosa 57RP correlates with emergence of hyperpiliated and highly adherent phenotypic variants deficient in swimming, swarming, and twitching motilities. J Bacteriol 183: 1195–1204
Fuqua, C, Parsek, MR, Greenberg, EP (2001) Regulation of gene expression by cell-to-cell communication: acyl-homoserine lactone quorum sensing. Annu Rev Genet 35: 439–468
Genavaux, P, Muller, S, Bauda, P (1996) A rapid screening procedure to identify mini-Tn10 insertion mutants of Escherichia coli K-12 with altered adhesion properties. FEMS Microbiol Lett 142: 27–30
Heilmann, C, Gerke, C, Perdreau-Remington, F, Goetz, F (1996) Characterization of Tn917 insertion mutants of Staphylococcus epidermitis affected in biofilm formation. Infect Immun 64: 277–282
Hernandez, ME, Newman, DK (2001) Extracellular electron transfer. Cell Mol Life Sci 58: 1562–1571
Heydorn, A, Ersboll, BK, Hentzer, M, Parsek, MR, Givskov, M, Molin, S (2000) Experimental reproducibility in flow-chamber biofilms. Microbiology 146: 2409–2415
Klausen, M, Heydorn, A, Ragas, P, Lambertsen, L, Aaes-Jorgensen, A, Molin, S, Tolker-Neilsen, T (2003) Biofilm formation by Pseudomonas aeruginosa wild type, flagella and type IV pili mutants. Mol Microbiol 48: 1511–1524
Kierek-Pearson, K, Karatan, E (2005) Biofilm development in bacteria. Adv Appl Microbiol 57: 79–111
Laursen, JB, Nielsen, J (2004) Phenazine natural products: biosynthesis, synthetic analogues, and biological activity. Chem Rev 104: 1663–1686
Lazazzera, BA (2005) Lessons from DNA microarray analysis: the gene expression profile of biofilms. Curr Opin Microbiol 2: 222–227
Loo, CY, Corliss, DA, Ganeshkumar, N (2000) Streptococcus gordonii biofilm formation: identification of genes that code for biofilm phenotypes. J Bacteriol 182: 1374–1382
Mazzola, M, Cook, RJ, Thomashow, LS, Weller, DM, Pierson, LS (1992) Contribution of phenazine antibiotic biosynthesis to the ecological competence of fluorescent pseudomonads in soil habitats. Appl Environ Microbiol 58: 2616–2624
McLean, RJ, Whiteley, M, Stickler, DJ, Fuqua, WC (1997) Evidence of autoinducer activity in naturally occurring biofilms. FEMS Microbiol Lett 154: 259–263
Miller, MB, Bassler, BL (2001) Quorum sensing in bacteria. Annu Rev Microbiol 55: 165–199
Morris, CA, Monier, JM (2003) The ecological significance of biofilm formation by plant-associated bacteria. Annu Rev Phytopathol 41: 429–453
O'Toole, G, Kaplan, HB, Kolter, R (2000) Biofilm formation as microbial development. Annu Rev Microbiol 54: 49–79
O'Toole, GA, Kolter, R (1998) Flagellar and twitching motility are necessary for Pseudomonas aeruginosa biofilm development. Mol Microbiol 30: 295–304
O'Toole, GA, Kolter, R (1998) Initiation of biofilm formation in Pseudomonas fluorescens WC365 proceeds via multiple, convergent signaling pathways: a genetic analysis. Mol Microbiol 28: 449–461
Parsek, MR, Greenberg, EP (2005) Sociomicrobiology: the connections between quorum sensing and biofilms. Trends Microbiol 13: 27–33
Pierson III, LS, Keppenne, VD, Wood, DW (1994) Phenazine antibiotic biosynthesis in Pseudomonas aureofaciens 30-84 is regulated by PhzR in response to cell density. J Bacteriol 176: 3966–3974
Pierson, LS III, Thomashow, LS (1992) Cloning and heterologous expression of the phenazine biosynthetic locus from Pseudomonas aureofaciens 30-84. Mol Plant Microbe Interact 5: 330–339
Pierson III, LS, Wood, DW, Pierson, EA, Chancey, ST (1998) Homoserine lactone-mediated gene regulation in plant-associated bacteria. Annu Rev Phytopathol 36: 207–225
Pratt, LA, Kolter, R (1998) Genetic analysis of Escherichia coli biofilm formation: roles of flagella, motility, chemotaxis and type I pili. Mol Microbiol 30: 285–293
Ramey, BE, Koutsoudis, M, von Bodman, SB, Fuqua, C (2004) Biofilm formation in plant–microbe associations. Curr Opin Microbiol 7: 602–609
Rice, SA, Koh, KS, Queck, SY, Labbate, M, Lam, KW, Kjellberg, S (2005) Biofilm formation and sloughing in Serratia marcescens are controlled by quorum sensing and nutrient cues. J Bacteriol 187: 3477–3485
Rupp, ME, Ulphani, JS, Fey, PD, Mack, D (1999) Characterization of Staphylococcus epidermidis polysaccharide intercellular adhesion/hemagglutinin in the pathogenesis of intravascular catheter-associated infection in a rat model. Infect Immuol 67: 2656–2659
Sauer, K, Camper, AK (2001) Characterization of phenotypic changes in Pseudomonas putida in response to surface-associated growth. J Bacteriol 183: 6579–6589
Schuster, M, Lostroh, CP, Ogi, T, Greenberg, EP (2003) Identification, timing, and signal specificity of Pseudomonas aeruginosa quorum-controlled genes: a transcriptome analysis. J Bacteriol 185: 2066–2079
Staskawicz, B, Dahlbreck, D, Keen, N, Napoli, C (1987) Molecular characterization of cloned avirulence genes from race 0 and race 1 of Pseudomonas syringae pv. glycinea. J Bacteriol 169: 5789–5794
Turner, JM, Messenger, AJ (1986) Occurrence, biochemistry and physiology of phenazine pigment production. Adv Microb Physiol 27: 211–275
Ulrich, RL, Hines, HB, Parthasarathy, N, Jeddeloh, JA (2003) Mutational analysis and biochemical characterization of the Burkholderia thailandensis DW503 quorum-sensing network. J Bacteriol 186: 4350–4360
Von Bodman, SB, Bauer, WD, Coplin, DL (2003) Quorum sensing in plant-pathogenic bacteria. Annu Rev Phytopathol 41: 455–482
Wagner, VE, Bushnell, D, Passador, L, Brooks, AI, Iglewski, BH (2003) Microarray analysis of Pseudomonas aeruginosa quorum-sensing regulons: effects of growth phase and environment. J Bacteriol 185: 2080–2095
Watnick, PI, Kolter, R (1999) Steps in the development of a Vibrio cholerae El Tor biofilm. Mol Microbiol 34: 586–595
Whitehead, NA, Barnard, AM, Slater, H, Simpson, NJ, Salmond, GP (2001) Quorum-sensing in Gram-negative bacteria. FEMS Microbiol Rev 25: 365–404
Wood, DW, Pierson III, LS (1996) The phzI gene of Pseudomonas aureofaciens 30-84 is responsible for the production of a diffusible signal required for phenazine antibiotic production. Gene 168: 49–53
Wood, DW, Gong, F, Aykin, MM, Williams, P, Pierson III, LS (1997) N-Acyl-homoserine lactone-mediated regulation of phenazine gene expression by Pseudomonas aureofaciens 30-84 in the wheat rhizosphere. J Bacteriol 179: 7663–7670
Yoshida, A, Ansai, T, Takehara, T, Kuramitsu, HK (2005) LuxS-based signaling affects Streptococcus mutans biofilm formation. Appl Environ Microbiol 71: 2372–2380
Zhang, Z, Pierson III, LS (2001) A second quorum-sensing system regulates cell surface properties but not phenazine antibiotic production in Pseudomonas aureofaciens. Appl Environ Microbiol 67: 4305–4315
Acknowledgments
We thank Patricia Figuli, Shaina Schwartz, and Dr. Cheryl Whistler for technical assistance and Christopher Rensing for critical discussions. This work was supported by USDA NRICGP grant No. 2001-02684.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maddula, V.S.R.K., Zhang, Z., Pierson, E.A. et al. Quorum Sensing and Phenazines are Involved in Biofilm Formation by Pseudomonas chlororaphis (aureofaciens) Strain 30-84. Microb Ecol 52, 289–301 (2006). https://doi.org/10.1007/s00248-006-9064-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-006-9064-6