Abstract
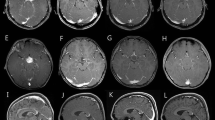
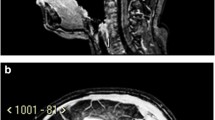
Background: Cerebellar low-grade astrocytomas (CLGAs) of childhood are benign tumours and are usually curable by surgical resection alone or combined with adjuvant radiotherapy. Objective: To undertake a retrospective study of our children with CLGA to determine the optimum schedule for surveillance imaging following initial surgery. In this report we describe the phenomenon of spontaneous regression of residual tumour and discuss its prognostic significance regarding future imaging. Materials and methods: A retrospective review was conducted of children treated for histologically proven CLGA at Great Ormond Street Hospital from 1988 to 1998. Results: Of 83 children with CLGA identified, 13 (15.7%) had incomplete resections. Two children with large residual tumours associated with persistent symptoms underwent additional treatment. Eleven children were followed by surveillance imaging alone for a mean of 6.83 years (range 2–13.25 years). Spontaneous tumour regression was seen in 5 (45.5%) of the 11 children. There were no differences in age, gender, symptomatology, histological grade or Ki-67 fractions between those with spontaneous tumour regression and those with progression. There was a non-significant trend that larger volume residual tumours progressed. Conclusions: Residual tumour followed by surveillance imaging may either regress or progress. For children with residual disease we recommend surveillance imaging every 6 months for the first 2 years, every year for years 3, 4 and 5, then every second year if residual tumour is still present 5 years after initial surgery. This would detect not only progressive or recurrent disease, but also spontaneous regression which can occur later than disease progression.

Similar content being viewed by others
References
Cushing H (1931) Experiences with the cerebellar astrocytoma. A critical review of seventy-six cases. Surg Gynecol Obstet 52:129–204
Due-Tonnessen BJ, Helseth E, Scheie D, et al (2002) Long term outcome after resection of benign cerebellar astrocytomas in children and young adults (0–19 years): report of 110 consecutive cases. Pediatr Neurosurg 37:71–80
Pencalet P, Maixner W, Sainte-Rose C, et al (1999) Benign cerebellar astrocytomas in children. J Neurosurg 90:265–273
Palma L, Russo A, Celli P (1984) Prognosis of the so-called “diffuse” cerebellar astrocytomas. Neurosurgery 15:315–317
Desai KI, Nadkami TD, Muzumdar DP, et al (2001) Prognostic factors for cerebellar astrocytomas in children: a study of 102 cases. Pediatr Neurosurg 35:311–317
Smoots D, Russell Geyer J, Lieberman DM, et al (1998) Predicting disease progression in childhood cerebellar astrocytoma. Childs Nerv Syst 14:636–648
Sgouros S, Fineron PW, Hockley AD (1995) Cerebellar astrocytoma of childhood; long term follow up. Childs Nerv Syst 11:89–96
Saunders DE, Hayward RD, Phipps KP, et al (2003) Surveillance neuroimaging of intracranial medulloblastoma in children: how effective, how often, and for how long? J Neurosurg 99:280–286
Good CD, Wade AM, Hayward RD, et al (2001) Surveillance neuroimaging of childhood intracranial ependymoma: how effective, how often, and for how long? J Neurosurg 94:27–32
Dirven CM, Mooj JJ, Molenaar WM (1997) Cerebellar pilocytic astrocytomas: a treatment protocol based upon analysis of 73 cases and a review of the literature. Childs Nerv Syst 13:17–23
Fernandez C, Figarwalla-Branger D, Girard N, et al (2003) Pilocytic astrocytomas in children: prognostic factors—a retrospective study of 80 cases. Neurosurgery 53:544–553
Saunders DE, Phipps KP, Wade AM, et al (2005) Surveillance imaging strategies following surgery and/or radiotherapy for childhood cerebellar low-grade astrocytomas. J Neurosurg 102:172–178
Schneider JH, Raffel C, McComb JG (1992) Benign cerebellar astrocytomas of childhood. Neurosurgery 30:58–62
Davis CH, Jogelkar VM (1981) Cerebellar astrocytomas in children and young adults. J Neurol Neurosurg Psychiatry 44:820–828
Kleihues P, Cavenee WK (2000) Astrocytic tumours. World Health Organization classification of tumours. Pathology and genetics of tumours of the nervous system. IARC Press, Lyon
Cairncross JG, Pexman W, Rathbone MP (1985) Post surgical contrast enhancement mimicking residual brain tumour. Can J Neurol Sci 12:75
Nicoletti GF, Barone F, Passanassi M, et al (1994) Linear contrast enhancement at the operative site on early post operative CT after removal of brain tumors. J Neurosurg Sci 38:131–135
Knauth M, Aras N, Wirtz CR, et al (1999) Surgically induced intracranial contrast enhancement: potential source of diagnostic error in intra-operative MR imaging. AJNR 20:1547–1553
Rollins NK, Nisen P, Shapiro KN (1998) The use of early post operative MR in detecting residual juvenile cerebellar pilocytic astrocytoma. AJNR 19:151–156
Griffiths PD (1999) A protocol for imaging paediatric brain tumours. United Kingdom Children’s Cancer Study Group (UKCCSG) and Societe Francaise D’Oncologie Pediatrique (SFOP) panelists. Clin Oncol 11:290–294
Forsting M, Albert FK, Kunze S, et al (1993) Extirpation of glioblastomas: MR and CT follow-up of residual tumor and regrowth patterns. AJNR 14:77–87
McKeever PE, Strawderman MS, Yamini B, et al (1998) MIB-1 proliferation index predicts survival among patients with grade II astrocytoma. J Neuropathol Exp Neurol 57:931–936
Shinoda J, Sakai N, Nakatani K, et al (1998) Prognostic factors in supratentorial WHO grade II astrocytomas in adults. Br J Neurosurg 12:318–324
Wakimoto H, Aoyagi M, Nakayama T, et al (1996) Prognostic significance of Ki-67 labelling indices obtained using MIB-1 monoclonal antibody in patients with supratentorial astrocytomas. Cancer 77:373–380
Wessels PH, Hopman AH, Kubat B, et al (2003) Proliferation and aneusomy predict survival of young patients with astrocytoma grade II. Br J Cancer 89:128–134
Hilton DA, Love S, Barber R, et al (1998) Accumulation of p53 and Ki-67 expression do not predict survival in patients with fibrillary astrocytomas or the response of these tumors to radiotherapy. Neurosurgery 42:724–729
Czech T, Slavc I, Aicholzer M, et al (1999) Proliferative activity as measured by MIB-1 labeling index and long-term outcome of visual pathway astrocytomas in children. J Neurooncol 42:143–150
Dirven CM, Koudstaal J, Mooj JJ, et al (1998) The proliferative potential of the pilocytic astrocytoma: the relation between MIB-1 labelling and clinical and neuroradiological follow-up. J Neurooncol 37:9–16
Gottschalk S, Tavakolian R, Buske A, et al (1999) Spontaneous remission of chiasmatic/hypothalamic masses in neurofibromatosis type 1: report of two cases. Neuroradiology 41:199–201
Parsa CF, Hoyt CS, Lesser RL, et al (2001) Spontaneous regression of optic gliomas: thirteen cases documented by serial neuroimaging. Arch Ophthalmol 119:516–529
Schmandt SM, Packer RJ, Vezina LG, et al (2000) Spontaneous regression of low-grade astrocytomas in childhood. Pediatr Neurosurg 32:132–136
Venes JL, Latack J, Kandt RS (1984) Postoperative regression of opticochiasmatic astrocytoma: a case for expectant therapy. Neurosurgery 15:421–423
Borit A, Richardson EP Jr (1982) The biological and clinical behaviour of pilocytic astrocytomas of the optic pathways. Brain 105(Pt 1):161–187
Hoffmann HJ (1994) Management and outcome of low-grade astrocytomas of the midline in children: a retrospective review. Neurosurgery 35:343–344
Folkman J (1995) Seminars in medicine of the Beth Israel Hospital, Boston: clinical applications of research on angiogenesis. N Engl J Med 333:1757–1763
Elston DM (2004) Mechanisms of regression. Clin Med Res 2:85–88
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gunny, R.S., Hayward, R.D., Phipps, K.P. et al. Spontaneous regression of residual low-grade cerebellar pilocytic astrocytomas in children. Pediatr Radiol 35, 1086–1091 (2005). https://doi.org/10.1007/s00247-005-1546-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-005-1546-z