Abstract
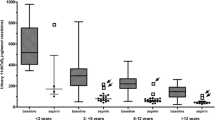
Aspirin (ASA) remains the most common antiplatelet agent used in children. VerifyNow Aspirin Test® (VN) assesses platelet response to ASA, with therapeutic effect defined by the manufacturer as ≤ 549 aspirin reaction units (ARU). Single-center, observational, analysis of 195 children (< 18 years-old) who underwent first VN between 2015 and 2020. Primary outcome was proportion of patients with ASA biochemical resistance (> 549 ARU). Secondary outcomes included incidence of new clinical thrombotic and bleeding events during ≤ 6 months from VN in those who received ASA monotherapy (n = 113). Median age was 1.8 years. Common indications for ASA included cardiac anomalies or dysfunction (74.8%) and ischemic stroke (22.6%). Median ASA dose before VN was 4.6 mg/kg/day. Mean VN was 471 ARU. ASA biochemical resistance was detected in 14.4% (n = 28). Of 113 patients receiving ASA monotherapy, 14 (12.4%) had a thrombotic event and 2 (1.8%) had a bleeding event. Mean VN was significantly higher at initial testing in patients experiencing thrombotic event compared to those without thrombosis (516 vs 465 ARU, [95% CI: 9.8, 92.2], p = 0.02). Multivariable analysis identified initial VN ASA result ≥ 500 ARU at initial testing as the only significant independent risk factor for thrombosis (p < 0.01). VN testing identifies ASA biochemical resistance in 14.4% of children. VN ASA ≥ 500 ARU rather than ≥ 550 ARU at initial testing was independently associated with increased odds of thrombosis. Designated cut-off of 550 ARU for detecting platelet dysfunction by ASA may need reconsideration in children.
Similar content being viewed by others
References
Monagle P, Chan AKC, Goldenberg NA, Ichord RN, Journeycake JM, Nowak-Göttl U, Vesely SK (2012) Antithrombotic therapy in neonates and children: antithrombotic therapy and prevention of thrombosis, 9th ed: American college of chest physicians evidence-based clinical practice guidelines. Chest 141(2 Suppl):e737S–e801S. https://doi.org/10.1378/chest.11-2308. Erratum in: Chest. 2014 Dec;146(6):1694. Dosage error in article text. Erratum in: Chest. 2014 Nov;146(5):1422.
Giglia TM, Massicotte MP, Tweddell JS, Barst RJ, Bauman M, Erickson CC, Feltes TF, Foster E, Hinoki K, Ichord RN, Kreutzer J, McCrindle BW, Newburger JW, Tabbutt S, Todd JL, Webb CL (2013) American heart association congenital heart defects committee of the council on cardiovascular disease in the young, council on cardiovascular and stroke nursing, council on epidemiology and prevention, and stroke council. prevention and treatment of thrombosis in pediatric and congenital heart disease: a scientific statement from the american heart association. Circulation 128(24):2622–703. https://doi.org/10.1161/01.cir.0000436140.77832.7a. Erratum in: Circulation. 2014 Jan 14;129(2):e23.
Anton N, Cox PN, Massicotte MP, Chait P, Yasui Y, Dinyari PM, Marzinotto V, Mitchell LG (2009) Heparin-bonded central venous catheters do not reduce thrombosis in infants with congenital heart disease: a blinded randomized, controlled trial. Pediatr 123(3):e453–e458. https://doi.org/10.1542/peds.2008-1508
Eikelboom JW, Hirsh J, Spencer FA, Baglin TP, Weitz JI (2012) Antiplatelet drugs: antithrombotic therapy and prevention of thrombosis, 9th ed: American college of chest physicians evidence-based clinical practice guidelines. Chest 141(2 Suppl):e89S-e119S. https://doi.org/10.1378/chest.11-2293
Sivakumar K (2021) PDA stenting in duct-dependent pulmonary circulation. In: Butera G, Chessa M, Eicken A, Thomson J (eds) Cardiac catheterization for congenital heart disease. Springer, Cham, pp 449–478. https://doi.org/10.1007/978-3-030-69856-0_28
Li JS, Yow E, Berezny KY, Rhodes JF, Bokesch PM, Charpie JR, Forbus GA, Mahony L, Boshkov L, Lambert V, Bonnet D, Michel-Behnke I, Graham TP, Takahashi M, Jaggers J, Califf RM, Rakhit A, Fontecave S, Sanders SP (2007) Clinical outcomes of palliative surgery including a systemic-to-pulmonary artery shunt in infants with cyanotic congenital heart disease: does aspirin make a difference? Circ 116(3):293–297
Huang JY, Soeharto DA, Damara I, Ignjatovic V, Linden MD, Monagle P (2019) Antiplatelet therapy monitoring in neonates and children. Semin Thromb Hemost 45(1):73–85. https://doi.org/10.1055/s-0038-1676375
Erdem S, Akay E, Akbas T, Demir F, Ozbarlas N (2021) Aspirin resistance in children with congenital heart disease and its relationship with laboratory and clinical parameters. Iran J Pediatr 31(2):e109797
Cholette JM, Mamikonian L, Alfieris GM, Blumberg N, Lerner NB (2010) Aspirin resistance following pediatric cardiac surgery. Thromb Res. 126(3):200–6
Heistein LC, Scott WA, Zellers TM, Fixler DE, Ramaciotti C, Journeycake JM, Lemler MS (2008) Aspirin resistance in children with heart disease at risk for thromboembolism: prevalence and possible mechanisms. Pediatr Cardiol. 29(2):285–291
Emani S, Zurakowski D, Mulone M, DiNardo JA, Trenor CC 3rd, Emani SM (2017) Platelet testing to guide aspirin dose adjustment in pediatric patients after cardiac surgery. J Thorac Cardiovasc Surg 154(5):1723–1730
Emani S, Trainor B, Zurakowski D, Baird CW, Fynn-Thompson FE, Pigula FA, Emani SM (2014) Aspirin unresponsiveness predicts thrombosis in high-risk pediatric patients after cardiac surgery. J Thorac Cardiovasc Surg. 148(3):814–816
Koh W, Rodts M, Nebbia A, Sawyer J, Henry B, Cooper DS (2022) Aspirin resistance in infants with shunt-dependent congenital heart disease. Cardiol Young 32(5):705–710
VerifyNow Aspirin Test [package insert]. San Diego, CA; Accumetrics®; Revised April 2011.
Oh MS, Yu KH, Lee JH, Jung S, Kim C, Jang MU, Lee J, Lee BC (2016) Aspirin resistance is associated with increased stroke severity and infarct volume. Neurology 86(19):1808–1817. https://doi.org/10.1212/WNL.0000000000002657
Zheng AS, Churilov L, Colley RE, Goh C, Davis SM, Yan B (2013) Association of aspirin resistance with increased stroke severity and infarct size. JAMA Neurol 70(2):208–213. https://doi.org/10.1001/jamaneurol.2013.601
Pasala T, Hoo JS, Lockhart MK, Waheed R, Sengodan P, Alexander J, Gandhi S (2016) Aspirin resistance predicts adverse cardiovascular events in patients with symptomatic peripheral artery disease. Tex Heart Inst J 43(6):482–487. https://doi.org/10.14503/THIJ-14-4986
Nielsen HL, Kristensen SD (2007) Anne-mette hvas; is the new point-of-care test verifynow® aspirin able to identify aspirin resistance using the recommended cut-off? Blood 110(11):3896. https://doi.org/10.1182/blood.V110.11.3896.3896
Addonizio VP (1990) Platelet function in cardiopulmonary bypass and artificial organs. Hematol Oncol Clin North Am 4(1):145–155
Guthikonda S, Lev EI, Patel R, DeLao T, Bergeron AL, Dong JF, Kleiman NS (2007) Reticulated platelets and uninhibited COX-1 and COX-2 decrease the antiplatelet effects of aspirin. J Thromb Haemost 5(3):490–496
Schmugge M, Speer O, Kroiss S, Knirsch W, Kretschmar O, Rand ML, Albisetti M (2015) Monitoring aspirin therapy in children after interventional cardiac catheterization: laboratory measures, dose response, and clinical outcomes. Eur J Pediatr 174(7):933–941
Eikelboom JW, Hirsh J, Weitz JI, Johnston M, Yi Q, Yusuf S (2002) Aspirin-resistant thromboxane biosynthesis and the risk of myocardial infarction, stroke, or cardiovascular death in patients at high risk for cardiovascular events. Circ 105(14):1650–1655
Funding
No funds, grants, or other support were received.
Author information
Authors and Affiliations
Contributions
DMN, MMP, and MSK involved in study design, data acquisition, data analysis, interpretation of data, drafting, and revision of manuscript. LRK, JLP, ELA, JFL, BJH, YML, and KLS contributed to interpretation of data and revision of manuscript. All authors have approved the final article.
Corresponding author
Ethics declarations
Conflict of interest
All authors listed in this manuscript have no conflicts of interest to disclose and have approved the final article.
Informed Consent
The need for informed consent was waived for this study of de-identified data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Newland, D.M., Palmer, M.M., Knorr, L.R. et al. Analysis of Platelet Function Testing in Children Receiving Aspirin for Antiplatelet Effects. Pediatr Cardiol 45, 614–622 (2024). https://doi.org/10.1007/s00246-023-03377-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-023-03377-6