Abstract
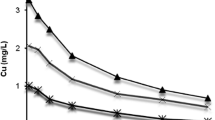
Dynamic changes in Cu speciation and its binding to fish gills were investigated by exposing common carp (Cyprinus carpio) to a 1 mg/1 Cu solution for 7 hours. Cu speciation in the bulk solution and fish gill microenvironment was calculated using general chemical equilibrium modeling. A sequential extraction procedure using distilled water, magnesium dichloride (1.0 mol/1), and acetic acid (10%) was used to characterize the Cu associated with the fish gills. Cu residual in the gill tissue was measured after the sequential extraction. Changes in total Cu concentration, pH, and dissolved organic carbon (Doc) in the bulk solution were recorded during the experimental period and calculated for the fish gill microenvironment. Cu-hydroxide species and Cu2 were dominant Cu species in both bulk solution and the fish gill microenvironment, whereas increased Cu–mucus was found in the fish gill microenvironment. DOC in the exposure medium, assumed to arise from mucus release, also increased and complexed Cu in solution. Forty-three percent of the Cu associated with the gills was readily water extractable, with an additional 22% exchangeable with Mg2+ or protons. Only 35% of the Cu accumulated within the gill tissues. The binding of Cu to the fish gills reached apparent equilibrium after 3 hours of exposure. Furthermore, the amount of water-extractable Cu within the gills showed significant correlation to the concentration of Cu predicted to be complexed with calculated free mucus in the gill microenvironment.







Similar content being viewed by others
References
Allison JD, Brown DS, Novo-Gradac KJ (1991) MINTEQA2/PRODEFA2, a geochemical assessment model for environmental systems: version 3.0 User’s Manual. Environ Res Lab United States Environmental Protection Agency Washington, D.C.
Allison JD, Brown DS (1995) MINTEQA2/PRODEFA2—A geochemical speciation model and interactive preprocessor. In: Loeppert RH, Schwab AP, Goldberg S, (eds) Chemical equilibrium and reaction models. SSSA Publication no. H2. SSSA, Madison, WI, pp 241–252
Toro DM, Alien HE, Bergman HL, Meyer JS, Paquin PR, Santore RC (2001) Biotic ligand model of the acute toxicity of metals, 1. Technical basis. Environ Toxicol Chem 20:2382–2396
Eddy FB (1982) Osmotic and ionic regulation in captive fish with particular reference to salmonids. Comp Biochem Physiol B. 73:125–141
Filgueiras AV, Lavilla I, Bendicho C (2002) Chemical sequential extraction for metal partitioning in environmental solid samples. J Environ Monit 4:823–857
Gleyzes C, Tellier S, Astruc M (2002) Fractionation studies of trace elements in contaminated soils and sediments: A review of sequential extraction procedures. T Trends Analyst Chem 21:451–467
Handy RD (1989) The ionic composition of rainbow trout body mucus. Comp Biochem Physiol 93A:571–576
Handy RD, Eddy FB, Baines R (2002) Sodium-dependent copper uptake across epithelia; a review of rationale with experimental evidence from gill and intestine. Biochim Biophys Acta l566:104–115
Hudson RJM (1998) Which aqueous species control the rates of trace metal uptake by aquatic biota? Observations and predictions of non-equilibrium effects. Sci Total Environ 219:95–115
Janes N, Playle RC (1995) Modeling silver-binding to gills of rainbow-trout (Oncorhynchus mykiss). Environ Toxicol Chem l4:1847–1858
Kirschner LB (1978) External charged layer and Na+ regulation. In: Jorgensen CB, Skadhauge E (eds) Osmotic and volume regulation. Academic Press, New York, NY, p. 310–324
MacRae RK, Smith DE, Swoboda-Colberg N, Meyer JS, Bergman HL (1999) Copper binding affinity of rainbow trout (Oncorhynchus mykiss) and brook trout (Salvelinus fontinalis) gills; Implications for assessing bioavailable metal, Environ Toxicol Chem 18:1180–1189
McGeer JC, Playle RC, Wood CM, Galvez F (2000) A physiologically based biotic ligand model for predicting the acute toxicity of waterborne silver to rainbow trout in freshwaters. Environ Sci TechnoL 34:4199–14207
McKim JM (1994) Physiological and biochemical mechanisms that regulate the accumulation and toxicity of environmental chemicals in fish. In: Hamelink JL, Landrum PF, Bergman HL, Benson WH (eds) Bioavailability: physical, chemical, and biological interactions. CRC, Boca Raton, FL, pp 179–201
Meyer JS, Santore RC, Bobbitt JP, Debrey LD, Boese CJ, Paquin PR, Alien HE, et al. (1999) Binding of nickel and copper to fish gills predicts toxicity when water hardness varies, but free-ion activity does not. Environ Sci TechnoL 33:913–916
Newman MC, Jagoe CH (1994) Ligands and the bioavailability of metals in aquatic environments. In: Hamelink JL, Landrum PF, Bergman HL, Benson WH (eds) Bioavailability: Physical, chemical, and biological interactions. CRC, Boca Raton, FL pp 39–62
Pagenkopf GK (1983) Gill surface interaction model for trace-metal toxicity to fishes: Role of complexation, pH and water hardness. Environ Sci Technol 111:342–347
Paquin PR, Santore RC, Wu KB, Kavvadas CD, Di Toro DM (2000) The biotic ligand model:A model of the acute toxicity of metals to aquatic life. Environ Sci Policy, 3(Suppl.):S175–S182
Pan B, Cao J, Liu GJ, Tao S (2002) Characteristics of common carp gill mucus complexed with copper. Environ Cheim 21:245–249
Part P, Lock RAC (1983) Diffusion of calcium, cadmium and mercury in a mucous solution from rainbow trout. Comp Biochem Physiol C 76:259–263
Playle RC, Gensemer RW, Dixon DG (1992) Copper accumulation on gills of fathead minnows: Influence of water hardness, complexation and pH of gill microenvironment. Environ ToxicolChem 11:381–391
Playle RC, Dixon DQ Burnison K (1993a) Copper and cadmium binding to fish gills: Modification by dissolved organic carbon and synthetic ligands, Can J Fish Aquat ScL 50:2667–2677
Playle RC, Dixon DQ Burnison K (1993b) Copper and cadmium binding to fish gills: Estimates of metal-gill stability constants and modelling of metal accumulation. Can J Fish Aquat Sci 50:2678–2687
Randall D, Lin H, Wright PA (1998) Gill water flow and chemistry of the boundary layer, Physiol Zool 64:26–38
Richards JG, Playle RC (1998) Cobalt binding to gills of rainbow trout (Oncorhynchus mykiss): An equilibrium model. Comp Biochem Physiol C 119C:185–197
Santore RC, Di Toro DM, Paquin PR, Alien HE, Meyer JS (2001) Biotic ligand model of the acute toxicity of metals. 2. Application to acute copper toxicity in freshwater fish and Daphnia. Environ Toxicol Chem 20:2397–2402
Shephard KL (1989) The effects of mucus and mucilaginous materials on ion-distributions at 17 epithelial surface. In: Chantler E, Ratcliffe NA (eds) Mucus and related topics, Co. Biol. Ltd., Cambridge UK pp 123–130
Shephard KL (1992) Studies on the fish gill microclimate. J Comp Physiol 162B:231–240
Tao S, Long AM, Liu CF, Dawson R (2000) The Influence of mucus on copper speciation in the gill microenvironment of carp (Cypriirus carpio). Ecotoxicol Environ Saf 47:59–64
Tao S, Wen Y, Long AM, Dawson R, Cao J, Xu FL (2001) Simulation of acid-base condition and copper speciation in the fish gill microenvironment. Compute Chem 25:215–222
Tao S, Long AM, Xu FL, Dawson R (2002a) Copper speciation in the gill microenvironment of carp (Cyprinus carpio) at various levels of pPL. Ecotoxicol Environ Saf 52:221–226
Tao S, Long AM, Pan B, Xu FL, Dawson R (2002b) Uptake of copper complexed to EDTA, diaminoethane, oxalic acid, or tartatic acid by neon tetra. Ecotoxicol Environ Saf 53:317–322
Tao S, Guo LQ, Wang X, Liu WX, Ju TZ, Dawson R, et al. (2004) Use of sequential ASE extraction to evaluate the bioavailability of DDT and its metabolites to wheat roots in soils with various organic carbon contents. Sci Total Environ. 320:1–9
Wright PA, Randall DJ, Perry II SF (1989) Fish gill water boundary layer: A site of linkage between carbon dioxide and ammonia excretion. J Comp Physiol B 158:627–635
Acknowledgments
Funding was provided by International Copper Association (Grant No. E-AS-02-03) and The National Scientific Foundation of China (Grants No. 400332015 and 40021101).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tao, S., Liu, W.X., Liu, G.J. et al. Short-Term Dynamic Change of Gill Copper in Common Carp, Cyprinus carpio, Evaluated by a Sequential Extraction. Arch Environ Contam Toxicol 51, 408–415 (2006). https://doi.org/10.1007/s00244-005-0170-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-005-0170-0