Abstract
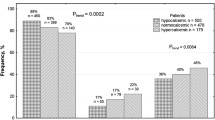
We examined whether single nucleotide polymorphisms (SNPs) in SPP1 gene are associated with risk of calcium oxalate urolithiasis (COU). We genotyped nine known SNPs in SPP1 gene (rs11739060, rs28357094, rs2728127, rs11730582, rs1126772, rs9138, rs2853744, rs4754=p.Asp80Asp, and rs1126616=p.Ala236Ala). Genomic DNA from 1,026 individuals (n = 342 patients with first episode COU, and n = 684 healthy unrelated controls) was analyzed for nine SPP1 SNPs using polymerase chain reaction and melting curve analysis by means of a pair of fluorescence resonance energy transfer probes. Serum and urine osteopontin (OPN) levels were also measured using enzyme-linked immunosorbent assay kits. rs9138 AA genotype was protective (OR 0.62, 95 % CI 0.47–0.81; P = 0.004). rs28357094 TT genotype (OR 2.52, 95 % CI 1.74–3.79; P = 0.021), rs2728127 GG genotype (OR 2.64, 95 % CI 1.42–4.81; P = 0.002), and rs2853744 GG genotype (OR 1.68, 95 % CI 1.22–3.87; P = 0.003) were predisposing. None of the other examined SPP1 SNPs was associated with COU susceptibility. Subjects with protective and predisposing polymorphisms had increased and decreased serum levels of OPN, respectively. Urinary calcium/OPN ratios were higher and lower in subjects with predisposing and protective SNPs of SPP1 gene, respectively. Of 28 constructed haplotypes, 6 demonstrated significant association with COU risk. There was no sex difference in the obtained results. The SPP1 gene polymorphisms are associated with the COU susceptibility.

Similar content being viewed by others
References
Lee YH, Huang WC, Tsai JY, Lu CM, Chen WC, Lee MH, Hsu HS, Huang JK, Chang LS (2002) Epidemiological studies on the prevalence of upper urinary calculi in Taiwan. Urol Int 68:172–177
Safarinejad MR (2007) Adult urolithiasis in a population-based study in Iran: prevalence, incidence, and associated risk factors. Urol Res 35:73–82
Goodman HO, Holmes RP, Assimos DG (1995) Genetic factors in calcium oxalate stone disease. J Urol 153:301–307
Yamate T, Umekawa T, Iguchi M, Kohri K, Kurita T (1998) Molecular biological diagnosis of urolithiasis. Rinsho Hinyokika 52:213–221
Suzuki K, Kikuchi N, Nagata K (1994) Isolation and partial characterization of crystal matrix protein as a potent inhibitor of calcium oxalate crystal aggregation: evidence of activation peptide of human prothrombin. Urol Res 22:45–50
Wesson JA, Ganne V, Beshensky AM, Kleinman JG (2005) Regulation by macromolecules of calcium oxalate crystal aggregation in stone formers. Urol Res 33:206–212
Atmani F, Glenton PA, Khan SR (1998) Identification of proteins extracted from calcium oxalate and calcium phosphate crystals induced in the urine of healthy and stone forming subjects. Urol Res 26:201–207
Worcester EM, Beshensky AM (1995) Osteopontin inhibits nucleation of calcium oxalate crystals. Ann N Y Acad Sci 760:375–377
Yagisawa T, Chandhoke PS, Fan J, Lucia S (1998) Renal osteopontin expression in experimental urolithiasis. J Endourol 12:171–176
Young MF, Kerr JM, Termine JD, Wewer UM, Wang MG, McBride OW, Fisher LW (1990) cDNA cloning, mRNA distribution and heterogeneity, chromosomal location, and RFLP analysis of human osteopontin (OPN). Genomics 7:491–502
El-Tanani MK, Campbell FC, Kurisetty V, Jin D, McCann M, Rudland PS (2006) The regulation and role of osteopontin in malignant transformation and cancer. Cytokine Growth Factor Rev 17:463–474
Umekawa T, Yamate T, Amasaki N, Kohri K, Kurita T (1995) Osteopontin mRNA in the kidney on an experimental rat model of renal stone formation without renal failure. Urol Int 55:6–10
Yamate T, Kohri K, Umekawa T, Iguchi M, Kurita T (1998) Osteopontin antisense oligonucleotide inhibits adhesion of calcium oxalate crystals in Madin–Darby canine kidney cell. J Urol 160:1506–1512
Umekawa T (1999) Structural characteristics of osteopontin for calcium oxalate crystal. Jpn J Urol 90:436–444
Mazzali M, Kipari T, Ophascharoensuk V, Wesson JA, Johnson R, Hughes J (2002) Osteopontin—a molecule for all seasons. Q J Med 95:3–13
Konya E, Umekawa T, Iguchi M, Kurita T (2003) The role of osteopontin on calcium oxalate crystal formation. Eur Urol 43:564–571
Chen WC, Lu HF, Chen HY, Hsu CD, Tsai FJ (2001) Arginine form of p21 gene codon 31 is less prominent in patients with calcium oxalate stone. Urol Res 29:94–97
Giacopelli F, Marciano R, Pistorio A, Catarsi P, Canini S, Karsenty G, Ravazzolo R (2004) Polymorphisms in the osteopontin promoter affect its transcriptional activity. Physiol Genomics 20:87–96
D’Alfonso S, Barizzone N, Giordano M, Chiocchetti A, Magnani C, Castelli L, Indelicato M, Giacopelli F, Marchini M, Scorza R, Danieli MG, Cappelli M, Migliaresi S, Bigliardo B, Sabbadini MG, Baldissera E, Galeazzi M, Sebastiani GD, Minisola G, Ravazzolo R, Dianzani U, Momigliano-Richiardi P (2005) Two single-nucleotide polymorphisms in the 5′ and 3′ ends of the osteopontin gene contribute to susceptibility to systemic lupus erythematosus. Arthritis Rheum 52:539–547
Caillier S, Barcellos LF, Baranzini SE, Swerdlin A, Lincoln RR, Steinman L, Martin E, Haines JL, Pericak-Vance M, Hauser SL, Oksenberg JR, Multiple Sclerosis Genetics Group (2003) Osteopontin polymorphisms and disease course in multiple sclerosis. Genes Immun 4:312–315
Chiocchetti A, Indelicato M, Bensi T, Mesturini R, Giordano M, Sametti S, Castelli L, Bottarel F, Mazzarino MC, Garbarini L, Giacopelli F, Valesini G, Santoro C, Dianzani I, Ramenghi U, Dianzani U (2004) High levels of osteopontin associated with polymorphisms in its gene are a risk factor for development of autoimmunity/lymphoproliferation. Blood 103:1376–1382
Marciano R, D’Annunzio G, Minuto N, Pasquali L, Santamaria A, Di Duca M, Ravazzolo R, Lorini R (2009) Association of alleles at polymorphic sites in the osteopontin encoding gene in young type 1 diabetic patients. Clin Immunol 131:84–91
Gao B, Yasui T, Itoh Y, Li Z, Okada A, Tozawa K, Hayashi Y, Kohri K (2007) Association of osteopontin gene haplotypes with nephrolithiasis. Kidney Int 72:592–598
Gögebakan B, Igci YZ, Arslan A, Igci M, Erturhan S, Oztuzcu S, Sen H, Demiryürek S, Arikoglu H, Cengiz B, Bayraktar R, Yurtseven C, Sarıca K, Demiryürek AT (2010) Association between the T-593A and C6982T polymorphisms of the osteopontin gene and risk of developing nephrolithiasis. Arch Med Res 41:442–448
Liu CC, Huang SP, Tsai LY, Wu WJ, Juo SH, Chou YH, Huang CH, Wu MT (2010) The impact of osteopontin promoter polymorphisms on the risk of calcium urolithiasis. Clin Chim Acta 411:739–743
Glas J, Seiderer J, Nagy M, Fries C, Beigel F, Weidinger M, Pfennig S, Klein W, Epplen JT, Lohse P, Folwaczny M, Göke B, Ochsenkühn T, Diegelmann J, Müller-Myhsok B, Roeske D, Brand S (2010) Evidence for STAT4 as a common autoimmune gene: rs7574865 is associated with colonic Crohn’s disease and early disease onset. PLoS ONE 5:e10373
Watts RW (2005) Idiopathic urinary stone disease: possible polygenic aetiological factors. QJM 98:241–246
Yasui T, Fujita K, Hayashi Y, Ueda K, Kon S, Maeda M, Uede T, Kohri K (1999) Quantification of osteopontin in the urine of healthy and stone-forming men. Urol Res 27:225–230
Huang HS, Ma MC, Chen CF, Chen J (2003) Lipid peroxidation and its correlations with urinary levels of oxalate, citric acid, and osteopontin in patients with renal calcium oxalate stones. Urology 62:1123–1128
Asplin JR, Parks JH, Chen MS, Lieske JC, Toback FG, Pillay SN, Nakagawa Y, Coe FL (1999) Reduced crystallization inhibition by urine from men with nephrolithiasis. Kidney Int 56:1505–1516
Wesson JA, Johnson RJ, Mazzali M, Beshensky AM, Stietz S, Giachelli C, Liaw L, Alpers CE, Couser WG, Kleinman JG, Hughes J (2003) Osteopontin is a critical inhibitor of calcium oxalate crystal formation and retention in renal tubules. J Am Soc Nephrol 14:139–147
Gao B, Yasui T, Okada A, Tozawa K, Hayashi Y, Kohri K (2005) A polymorphism of the osteopontin gene is related to urinary calcium stones. J Urol 174:1472–1476
Yamate T, Tsuji H, Amasaki N, Iguchi M, Kurita T, Kohri K (2000) Analysis of osteopontin DNA in patients with urolithiasis. Urol Res 28:159–166
Iguchi M, Umekawa T, Katoh Y, Kohri K, Kurita T (1996) Prevalence of urolithiasis in Kaizuka City, Japan—an epidemiologic study of urinary stones. Int J Urol 3:175–179
Curhan GC, Rimm EB, Willett WC, Stampfer MJ (1994) Regional variation in nephrolithiasis incidence and prevalence among United States men. J Urol 151:838–841
Hesse A, Brandle E, Wilbert D, Kohrmann KU, Alken P (2003) Study on the prevalence and incidence of urolithiasis in Germany comparing the years 1979 vs. 2000. Eur Urol 44:709–713
Kim H, Jo MK, Kwak C, Park SK, Yoo KY, Kang D, Lee C (2003) Prevalence and epidemiologic characteristics of urolithiasis in Seoul, Korea. Urology 59:517–521
Nyholt DR (2001) Genetic case–control association studies—correcting for multiple testing. Hum Genet 109:564–567
Wang S, Wang X, Wu J, Lin Y, Chen H, Zheng X, Zhou C, Xie L (2012) Association of vitamin D receptor gene polymorphism and calcium urolithiasis in the Chinese Han population. Urol Res 40:277–284
Takaoka N, Takayama T, Miyazaki M, Nagata M, Ozono S (2008) Modification of primers for GRHPR genotyping: avoiding allele dropout by single nucleotide polymorphisms and homology sequence. Urol Res 36:297–302
Tugcu V, Ozbek E, Aras B, Arisan S, Caskurlu T, Tasci AI (2007) Manganese superoxide dismutase (Mn-SOD) gene polymorphisms in urolithiasis. Urol Res 35:219–224
Acknowledgments
We thank the participants, project managers, and coordinators involved in this study, without whom this study would not be possible. We also thank Saba Safarinejad for her technical assistance and statistical analysis. This work was supported by the nationwide financial support of the Iranian people.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Safarinejad, M.R., Shafiei, N. & Safarinejad, S. Association between polymorphisms in osteopontin gene (SPP1) and first episode calcium oxalate urolithiasis. Urolithiasis 41, 303–313 (2013). https://doi.org/10.1007/s00240-013-0582-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00240-013-0582-7