Abstract
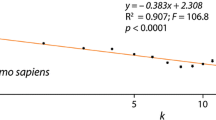
Diverse studies have shown that the content of genes present in sequenced genomes does not seem to correlate with the complexity of the organisms. However, various studies have shown that organism complexity and the size of the proteome has, indeed, a significant correlation. This characteristic allows us to postulate that some molecular mechanisms have permitted a greater functional diversity to some proteins to increase their participation in developing organisms with higher complexity. Among those mechanisms, the domain promiscuity, defined as the ability of the domains to organize in combination with other distinct domains, is of great importance for the evolution of organisms. Previous works have analyzed the degree of domain promiscuity of the proteomes showing how it seems to have paralleled the evolution of eukaryotic organisms. The latter has motivated the present study, where we analyzed the domain promiscuity in a collection of 84 eukaryotic proteomes representative of all the taxonomy groups of the tree of life. Using a grammar definition approach, we determined the architecture of 1,223,227 proteins, conformed by 2,296,371 domains, which established 839,184 bigram types. The phylogenetic reconstructions based on differences in the content of information from measures of proteome promiscuity confirm that the evolution of the promiscuity of domains in eukaryotic organisms resembles the evolutionary history of the species. However, a close analysis of the PHD and RING domains, the most promiscuous domains found in fungi and functional components of chromatin remodeling enzymes and important expression regulators, suggests an evolution according to their function.





Similar content being viewed by others
References
Amoutzias GD, Robertson DL, Oliver SG, Bornberg-Bauer E (2004) Convergent evolution of gene networks by single-gene duplications in higher eukaryotes. EMBO Rep 5:274
Apic G, Huber W, Teichmann SA (2003) Multi-domain protein families and domain pairs: comparison with known structures and a random model of domain recombination. J Struct Funct Genomics 4:67
Barrera A, Alastruey-Izquierdo A, Martin MJ, Cuesta I, Vizcaino JA (2014) Analysis of the protein domain and domain architecture content in fungi and its application in the search of new antifungal targets. PLoS Comput Biol 10:e1003733
Basu MK, Carmel L, Rogozin IB, Koonin EV (2008) Evolution of protein domain promiscuity in eukaryotes. Genome Res 18:449
Basu MK, Poliakov E, Rogozin IB (2009) Domain mobility in proteins: functional and evolutionary implications. Brief Bioinform 10:205
Blanc G, Wolfe KH (2004) Widespread paleopolyploidy in model plant species inferred from age distributions of duplicate genes. Plant Cell 16:1667
Brites D, Brena C, Ebert D, Du Pasquier L (2013) More than one way to produce protein diversity: duplication and limited alternative splicing of an adhesion molecule gene in basal arthropods. Evolution 67:2999
Buljan M, Frankish A, Bateman A (2010) Quantifying the mechanisms of domain gain in animal proteins. Genome Biol 11:R74
Canovas D, Marcos AT, Gacek A, Ramos MS, Gutierrez G, Reyes-Dominguez Y, Strauss J (2014) The histone acetyltransferase GcnE (GCN5) plays a central role in the regulation of Aspergillus asexual development. Genetics 197:1175
Cavalier-Smith T, Chao EE, Snell EA, Berney C, Fiore-Donno AM, Lewis R (2014) Multigene eukaryote phylogeny reveals the likely protozoan ancestors of opisthokonts (animals, fungi, choanozoans) and Amoebozoa. Mol Phylogenet Evol 81:71
Choi J, Kim SH (2020) Whole-proteome tree of life suggests a deep burst of organism diversity. Proc Natl Acad Sci USA 117:3678
Choi IY, Kwon EC, Kim NS (2020) The C- and G-value paradox with polyploidy, repeatomes, introns, phenomes and cell economy. Genes Genomics 42:699
Clarke AS, Lowell JE, Jacobson SJ, Pillus L (1999) Esa1p is an essential histone acetyltransferase required for cell cycle progression. Mol Cell Biol 19:2515
Clarke AS, Samal E, Pillus L (2006) Distinct roles for the essential MYST family HAT Esa1p in transcriptional silencing. Mol Biol Cell 17:1744
Cohen-Gihon I, Fong JH, Sharan R, Nussinov R, Przytycka TM, Panchenko AR (2011) Evolution of domain promiscuity in eukaryotic genomes—a perspective from the inferred ancestral domain architectures. Mol Biosyst 7:784
Cromar G, Wong KC, Loughran N, On T, Song H, Xiong X, Zhang Z, Parkinson J (2014) New tricks for “old” domains: how novel architectures and promiscuous hubs contributed to the organization and evolution of the ECM. Genome Biol Evol 6:2897
Darriba D, Taboada GL, Doallo R, Posada D (2011) ProtTest 3: fast selection of best-fit models of protein evolution. Bioinformatics 27:1164
Dohmen E, Klasberg S, Bornberg-Bauer E, Perrey S, Kemena C (2020) The modular nature of protein evolution: domain rearrangement rates across eukaryotic life. BMC Evol Biol 20:30
Eddy SR (2011) Accelerated profile HMM searches. PLoS Comput Biol 7:e1002195
El-Gebali S, Mistry J, Bateman A, Eddy SR, Luciani A, Potter SC, Qureshi M, Richardson LJ, Salazar GA, Smart A, Sonnhammer ELL, Hirsh L, Paladin L, Piovesan D, Tosatto SCE, Finn RD (2019) The Pfam protein families database in 2019. Nucleic Acids Res 47:D427
Forslund K, Henricson A, Hollich V, Sonnhammer EL (2008) Domain tree-based analysis of protein architecture evolution. Mol Biol Evol 25:254
Grant JR, Katz LA (2014) Building a phylogenomic pipeline for the eukaryotic tree of life—addressing deep phylogenies with genome-scale data. PLoS Curr. https://doi.org/10.1371/currents.tol.c24b6054aebf3602748ac042ccc8f2e9
Hill-Burns EM, Debelius JW, Morton JT, Wissemann WT, Lewis MR, Wallen ZD, Peddada SD, Factor SA, Molho E, Zabetian CP, Knight R, Payami H (2017) Parkinson’s disease and Parkinson’s disease medications have distinct signatures of the gut microbiome. Mov Disord 32:739
Hobohm U, Scharf M, Schneider R, Sander C (1992) Selection of representative protein data sets. Protein Sci 1:409
Kondrashov FA, Koonin EV (2001) Origin of alternative splicing by tandem exon duplication. Hum Mol Genet 10:2661
Lan H, Sun R, Fan K, Yang K, Zhang F, Nie XY, Wang X, Zhuang Z, Wang S (2016) The Aspergillus flavus histone acetyltransferase AflGcnE regulates morphogenesis, aflatoxin biosynthesis, and pathogenicity. Front Microbiol 7:1324
Lance GN, Williams WT (1966) Computer programs for hierarchical polythetic classification (“similarity analyses”). Comput J 9:60
Le SQ, Gascuel O (2008) An improved general amino acid replacement matrix. Mol Biol Evol 25:1307
Lee KK, Workman JL (2007) Histone acetyltransferase complexes: one size doesn’t fit all. Nat Rev Mol Cell Biol 8:284
Letunic I, Bork P (2016) Interactive tree of life (iTOL) v3: an online tool for the display and annotation of phylogenetic and other trees. Nucleic Acids Res 44:W242
Lynch M (2007) The frailty of adaptive hypotheses for the origins of organismal complexity. Proc Natl Acad Sci USA 104(Suppl 1):8597
Marcotte EM, Pellegrini M, Ng HL, Rice DW, Yeates TO, Eisenberg D (1999) Detecting protein function and protein-protein interactions from genome sequences. Science 285:751
Marsh JA, Teichmann SA (2010) How do proteins gain new domains? Genome Biol 11:126
Matthews JM, Sunde M (2002) Zinc fingers–folds for many occasions. IUBMB Life 54:351
Meng Y, Fan Y, Liao W, Lin X (2018) Plant Homeodomain genes play important roles in cryptococcal yeast-hypha transition. Appl Environ Microbiol. https://doi.org/10.1128/AEM.01732-17
Metzger MB, Pruneda JN, Klevit RE, Weissman AM (2014) RING-type E3 ligases: master manipulators of E2 ubiquitin-conjugating enzymes and ubiquitination. Biochim Biophys Acta 1843:47
Montell DJ (2003) The P160 family of steroid hormone receptor coactivators. In: Crews ST (ed) PAS Proteins: regulators and sensors of development and physiology. Springer, Boston, pp 133–147
Muñoz-Ramirez ZY, Mendez-Tenorio A, Kato I, Bravo MM, Rizzato C, Thorell K, Torres R, Aviles-Jimenez F, Camorlinga M, Canzian F, Torres J (2017) Whole genome sequence and phylogenetic analysis show Helicobacter pylori strains from Latin America have followed a unique evolution pathway. Front Cell Infect Microbiol 7:50
Ortiz-Alvarez J, Becerra-Bracho A, Mendez-Tenorio A, Murcia-Garzon J, Villa-Tanaca L, Hernandez-Rodriguez C (2020) Phylogeny, evolution, and potential ecological relationship of cytochrome CYP52 enzymes in Saccharomycetales yeasts. Sci Rep 10:10269
Panchy N, Lehti-Shiu M, Shiu SH (2016) Evolution of gene duplication in plants. Plant Physiol 171:2294
Pang E, Tan T, Lin K (2012) Promiscuous domains: facilitating stability of the yeast protein-protein interaction network. Mol Biosyst 8:766
Patthy L (2003) Modular assembly of genes and the evolution of new functions. Genetica 118:217
Retief JD (2000) Phylogenetic analysis using PHYLIP. Methods Mol Biol 132:243
Roth SY, Denu JM, Allis CD (2001) Histone acetyltransferases. Annu Rev Biochem 70:81
Sanchez R, Zhou MM (2011) The PHD finger: a versatile epigenome reader. Trends Biochem Sci 36:364
Schad E, Tompa P, Hegyi H (2011) The relationship between proteome size, structural disorder and organism complexity. Genome Biol 12:R120
Searls DB (2002) The language of genes. Nature 420:211
Seidl MF, Van den Ackerveken G, Govers F, Snel B (2011) A domain-centric analysis of oomycete plant pathogen genomes reveals unique protein organization. Plant Physiol 155:628
Smith ER, Eisen A, Gu W, Sattah M, Pannuti A, Zhou J, Cook RG, Lucchesi JC, Allis CD (1998) ESA1 is a histone acetyltransferase that is essential for growth in yeast. Proc Natl Acad Sci USA 95:3561
Spratt DE, Walden H, Shaw GS (2014) RBR E3 ubiquitin ligases: new structures, new insights, new questions. Biochem J 458:421
Sterner DE, Berger SL (2000) Acetylation of histones and transcription-related factors. Microbiol Mol Biol Rev 64:435
Suganuma T, Workman JL (2008) Crosstalk among histone modifications. Cell 135:604
Thomsen MC, Nielsen M (2012) Seq2Logo: a method for construction and visualization of amino acid binding motifs and sequence profiles including sequence weighting, pseudo counts and two-sided representation of amino acid enrichment and depletion. Nucleic Acids Res 40:W281
Todd AE, Orengo CA, Thornton JM (2001) Evolution of function in protein superfamilies, from a structural perspective. J Mol Biol 307:1113
Tordai H, Nagy A, Farkas K, Banyai L, Patthy L (2005) Modules, multidomain proteins and organismic complexity. FEBS J 272:5064
Treangen TJ, Rocha EP (2011) Horizontal transfer, not duplication, drives the expansion of protein families in prokaryotes. PLoS Genet 7:e1001284
Weiner J 3rd, Moore AD, Bornberg-Bauer E (2008) Just how versatile are domains? BMC Evol Biol 8:285
Wuchty S (2001) Scale-free behavior in protein domain networks. Mol Biol Evol 18:1694
Yang S, Bourne PE (2009) The evolutionary history of protein domains viewed by species phylogeny. PLoS ONE 4:e8378
Yu L, Tanwar DK, Penha EDS, Wolf YI, Koonin EV, Basu MK (2019) Grammar of protein domain architectures. Proc Natl Acad Sci USA 116:3636
Zheng N, Shabek N (2017) Ubiquitin ligases: structure, function, and regulation. Annu Rev Biochem 86:129
Zhuang J, Huo Q, Yang F, Xie N (2020) Perspectives on the role of histone modification in breast cancer progression and the advanced technological tools to study epigenetic determinants of metastasis. Front Genet 11:603552
Acknowledgements
JMG is grateful to the "Consejo Nacional de Ciencia y Tecnología" (CONACYT) and PIFI/BEIFI-IPN for the scholarships. AMT is fellow of SNI-CONACYT. To the memory of Ph. D. Juan Manuel González Prieto.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling editor: Erich Bornberg-Bauer.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Murcia-Garzón, J., Méndez-Tenorio, A. Promiscuous Domains in Eukaryotes and HAT Proteins in FUNGI Have Followed Different Evolutionary Paths. J Mol Evol 90, 124–138 (2022). https://doi.org/10.1007/s00239-021-10046-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-021-10046-w