Abstract
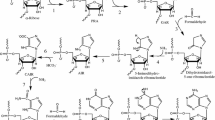
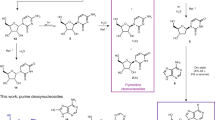
Systematic investigation into the chemical etiology of ribose has led to the discovery of glycerol nucleic acid (GNA) and threose nucleic acid (TNA) as possible progenitor candidates of RNA in the origins of life. Coupled with their chemical simplicity, polymers for both systems are capable of forming stable Watson-Crick antiparallel duplex structures with themselves and RNA, thereby providing a mechanism for the transfer of genetic information between successive genetic systems. Investigation into whether both polymers arose independently or descended from a common evolutionary pathway would provide additional constraints on models that describe the emergence of a hypothetical RNA world. Here we show by thermal denaturation that complementary GNA and TNA mixed sequence polymers are unable, even after prolonged incubation times, to adopt stable helical structures by intersystem cross-pairing. This experimental observation suggests that GNA and TNA, whose structures derive from one another, were not consecutive polymers in the same evolutionary pathway to RNA.





Similar content being viewed by others
References
Acevedo OL, Andrews RS (1996) Synthesis of propane-2,3-diol combinatorial monomers. Tetrahedron Lett 37:3931–3139
Ban N, Nissen P, Hansen J, Moore PB, Steitz TA (2000) The complete atomic structure of the large ribosomal subunit at 2.4 Å resolution. Science 289:905–920
Chaput JC, Switzer C (2000) Nonenzymatic oligomerization on tempaltes containing phosphodiester-linked acyclic glycerol nucleic acid analogues. J Mol Evol 51:464–470
Cooper G, Kimmich N, Belisle W, Sarinana J, Brabham K, Garrel L (2001) Carbonaceous meteorites as a source of sugar-related organic compounds for the early earth. Nature 414:879–883
Eschenmoser A (1999) Chemical etiology of nucleic acid structure. Science 284:2118–2124
Gilbert W (1986) The RNA world. Nature 319:618
Holý A (1975) Aliphatic analogues of nucleosides, nucleotides, and oligonucleotides. Collect Czech Chem Commun 40:187–214
Horhota A, Zou K, Ichida JK, Yu B, McLaughlin LW, Szostak JW, Chaput JC (2005) Kinetic analysis of an efficient DNA-dependent TNA polymerase. J Am Chem Soc 127:7427–7434
Horhota AT, Szostak JW, McLaughlin LW (2006) Glycerol nucleoside triphosphates: Synthesis and polymerase substrate activities. Organ Lett 8:5345–5347
Ichida JK, Zou K, Horhota AT, Yu B, McLaughlin LW, Szostak JW (2005) An in vitro selection system for TNA. J Am Chem Soc 127:2802–2803
Joyce GF (1989) RNA evolution and the origins of life. Nature 338:217–224
Joyce GF (2002) The antiquity of RNA-based evolution. Nature 418:214–221
Joyce GF, Schwartz AW, Miller SL, Orgel LE (1987) The case for an ancestral genetic system involving simple analogues of the nucleotides. Proc Natl Acad Sci USA 84:4398–4402
Kruger K, Grabowski PJ, Zaug AJ, Cech TR (1982) Self-splicing RNA: autoexcision and autocyclization of the ribosomal RNA intervening sequence by Tetrahymena. Cell 31:147–157
Marky LA, Breslauer KJ (1987) Calculating thermodynamic data for transitions of any molecularity from equilibrium melting curves. Biopolymers 26:1601–1620
Schlonvogt I, Pitsch S, Lesuer C, Eschenmoser A, Jaun B, Wolf RM (1996) Pyranosyl-RNA (‘p-RNA’): NMR and molecular dynamics study of the duplex formed by self-pairing of ribopyranosyl-(C-G-A-A-T-T-C-G). Helv Chim Acta 79:2316–2345
Schneider KC, Benner SA (1990) Oligonucleotides containing flexible nucleoside analogues. J Am Chem Soc 112:453–455
Schöning K-U, Scholz P, Guntha S, Wu X, Krishnamurthy R, Eschenmoser A (2000) Chemical etiology of nucleic acid structure: The α-threofuranosyl-(3′→2′) oligonucleotide system. Science 290:1347–1351
Schöning K-U, Scholz P, Wu X, Guntha S, Guillermo D, Krishnamurthy R, Eschenmoser A (2002) The α-L-threofuranosyl-(3′→2′)-oligonucleotide system(‘TNA’): Synthesis and pairing properties. Helv Chim Acta 85:4111–4153
Wilds CJ, Wawrzak Z, Krishnamurthy R, Eschenmoser A, Egli M (2002) Crystal structure of a B-form DNA duplex containing (L)-a-threofuranosyl (3′-2′) nucleosides: a four-carbon sugar is easily accommodated into the backbone of DNA. J Am Chem Soc 124:13716–13721
Wimberly BT, Brodersen DE, Clemons WM, Morgan-Warren RJ, Carter AP, Vonrhein C, Hartsch T, Ramakrishnan V (2000) Structure of the 30S ribosomal subunit. Nature 407:327–339
Yusupov MM, Yusupova GZ, Baucom A, Lieberman K, Earnest TN, Cate JHD, Noller HF (2001) Crystal structure of the ribosome at 5.5 Å resolution. Nature 292:883–896
Zhang L, Peritz A, Meggers E (2005) A simple glycol nucleic acid. J Am Chem Soc 127:4174–4175
Acknowledgment
This work was supported by new laboratory start-up funds from The Biodesign Institute at Arizona State University to J.C.C.
Author information
Authors and Affiliations
Corresponding author
Additional information
Reviewing Editor: Dr. Niles Lehman
Rights and permissions
About this article
Cite this article
Yang, YW., Zhang, S., McCullum, E.O. et al. Experimental Evidence That GNA and TNA Were Not Sequential Polymers in the Prebiotic Evolution of RNA. J Mol Evol 65, 289–295 (2007). https://doi.org/10.1007/s00239-007-9017-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-007-9017-9