Abstract
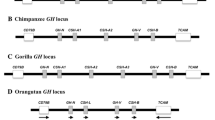
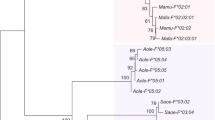
The GH gene cluster in marmoset, Callithrix jacchus, comprises eight GH-like genes and pseudogenes and appears to have arisen as a consequence of gene duplications occurring independently of those leading to the human GH gene cluster. We report here the complete sequence of the marmoset GH gene locus, including the intergenic regions and 5′ and 3′ flanking sequence, and a study of the multiple GH-like genes of an additional New World monkey (NWM), the white-fronted capuchin, Cebus albifrons. The marmoset sequence includes 945 nucleotides (nt) of 5′ flanking sequence and 1596 nt of 3′ flanking sequence that are “unique”; between these are eight repeat units, including the eight GH genes/pseudogenes. The breakpoints between these repeats are very similar, indicating a regular pattern of gene duplication. These breakpoints do not correspond to those found in the much less regular human GH gene cluster. This and phylogenetic analysis of the repeat units within the marmoset gene cluster strongly support the independent origin of these gene clusters, and the idea that the episode of rapid evolution that occurred during GH evolution in primates preceded the gene duplications. The marmoset GH gene cluster also differs from that of human in having fewer and more evenly distributed Alu sequences (a single pair in each repeat unit) and a “P-element” upstream of every gene/pseudogene. In human there is no P-element upstream of the gene encoding pituitary GH, and these elements have been implicated in placental expression of the other genes of the cluster. The GH gene clusters in marmoset and capuchin appear to have arisen as the consequence of a single-gene duplication event, but in capuchin there was then a remarkable expansion of the GH locus, giving at least 40 GH-like genes and pseudogenes. Thus even among NWMs the GH gene cluster is very variable.





Similar content being viewed by others
References
Adkins RM, Nekrutenko A, Li W-H (2001) Bushbaby growth hormone is much more similar to nonprimate growth hormones than to rhesus monkey and human growth hormones. Mol Biol Evol 18:55–60
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic Local Alignment Search Tool. J Mol Biol 215:403–410
Brown RJ, Adams JJ, Pelekanos RA, Wan Y, McKinstry WJ, Palethorpe K, Seeber RM, Monks TA, Eidne KA, Parker MW, Waters MJ (2005) Model for growth hormone receptor activation based on subunit rotation within a receptor dimer. Nature Struct Mol Biol 12:814–821
Chen EY, Liao Y-C, Smith DH, Barrera-Saldana HA, Gelinas RE, Seeburg PH (1989) The human growth hormone locus: nucleotide sequence, biology, and evolution. Genomics 4:479–497
De Vos AM, Ultsch M, Kossiakoff AA (1992) Human growth hormone and extracellular domain of its receptor: crystal structure of the complex. Science 255:306–312
Drouin G, Prat F, Ell M, Clarke GDP (1999) Detecting and characterizing gene conversions between multigene family members. Mol Biol Evol 16:1369–1390
Elefant F, Su Y, Liebhaber SA, Cooke NE (2000) Patterns of histone acetylation suggest dual pathways for gene activation by a bifunctional locus control region. EMBO J 19:6814–6822
Force A, Lynch M, Pickett FB, Amores A, Yan Y, Postlethwait J (1999) Preservation of duplicate genes by complementary, degenerative mutations. Genetics 151:1531–1545
Forsyth IA, Wallis M (2002) Growth hormone and prolactin—molecular and functional evolution. J Mammary Gland Biol Neoplasia 7:291–312
Giordano M, Marchetti C, Chiorboli E, Bona G, Richiardi PM (1997) Evidence for gene conversion in the generation of extensive polymorphism in the promoter of the growth hormone gene. Hum Genet 100:249–255
Goffin V, Shiverick KT, Kelly PA, Martial JA (1996) Sequence-function relationships within the expanding family of prolactin, growth hormone, placental lactogen, and related proteins in mammals. Endocr Rev 17:385–410
Golos TG, Durning M, Fisher JM, Fowler PD (1993) Cloning of four growth hormone/chorionic somatomammotropin-related complementary deoxyribonucleic acids differentially expressed during pregnancy in the rhesus monkey placenta. Endocrinology 133:1744–1752
Hancock JM (2005) Gene factories, microfunctionalization and the evolution of gene families. Trends Geneti 21:591–595
Harvey S, Azumaya Y, Hull KL (2000) Pituitary and extrapituitary growth hormone: Pit-1 dependence? Can J Physiol Pharmacol 78:1013–1028
Higgins DG, Sharp PM (1988) CLUSTAL: a package for performing multiple sequence alignment on a microcomputer. Gene 73:237–244
Ho Y, Liebhaber SA, Cooke NE (2004) Activation of the human GH gene cluster:roles for targeted chromatin modification. Trends Endocrinol Metab 15:40–45
Hugot JP (1998) Phylogeny of neotropical monkeys: the interplay of morphological, molecular, and parasitological data. Mol Phylogenet Evol 9:408–413
Kimura AP, Liebhaber SA, Cooke NE (2004) Epigenetic modifications at the human growth hormone locus predict distinct roles for histone acetylation and methylation in placental gene activation. Mol Endocrinol 18:1018–1032
Li W-H (1997) Molecular evolution. Sinauer, Sunderland, MA
Li Y, Ye C, Shi P, Zou X-J, Xiao R, Gong Y-Y, Zhang Y-P (2005) Independent origin of the growth hormone gene family in New World monkeys and Old World monkeys and hominoids. J Mol Endocrinol 35:399–409
Liu J-C, Makova KD, Adkins RM, Gibson S, Li W-H (2001) Episodic evolution of growth hormone in primates and emergence of the species specificity of human growth hormone receptor. Mol Biol Evol 18:945–953
Martial JA, Hallewell RA, Baxter JD, Goodman HM (1979) Human growth hormone complementary DNA cloning and expression in bacteria. Science 205:602–607
Ohta T (1993) Pattern of nucleotide substitutions in growth hormone-prolactin gene family: a paradigm for evolution by gene duplication. Genetics 134:1271–1276
Purvis A (1995) A composite estimate of primate phylogeny. Phil Trans Roy Soc London (Ser B) 348:405–421
Rand-Weaver M, Kawauchi H, Ono M (1993) Evolution of the structure of the growth hormone and prolactin family. In: Schreibman MP, Scanes CG, Pang PKT (eds) The endocrinology of growth, development, and metabolism in vertebrates . Academic Press, San Diego, CA, pp 13–42
Revol de Mendoza A, Esquivel Escobedo D, Santiago Alarcon D, Barrera Saldana H (2001) Independent duplication of the growth hormone gene in three anthropoidean lineages. J Endocr Genet 2:151–159
Revol de Mendoza A, Esquivel Escobedo D, Martinez Davila I, Barrera Saldana H (2004) Expansion and divergence of the GH locus between spider monkey and chimpanzee. Gene 336:185–193
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sawyer SA (1999) GENECONV: a computer package for the statistical detection of gene conversion. Distributed by the author, Department of Mathematics, Washington University, St. Louis, MO; available at: http://www.math.wustl.edu/∼sawyer
Swofford DL (1998) PAUP*. Phylogenetic Analysis Using Parsimony (*and other methods), version 4. Sinauer, Sunderland, MA
Wallis M (1981) The molecular evolution of pituitary growth hormone, prolactin and placental lactogen: a protein family showing variable rates of evolution. J Mol Evol 17:10–18
Wallis M (1994) Variable evolutionary rates in the molecular evolution of mammalian growth hormones. J Mol Evol 38:619–627
Wallis M, (1996) The molecular evolution of vertebrate growth hormones: a pattern of near-stasis interrupted by sustained bursts of rapid change. J Mol Evol 43:93–100
Wallis M (1997) Function switching as a basis for bursts of rapid change during the evolution of pituitary growth hormone. J Mol Evol 44:348–350
Wallis OC, Wallis M (2002) Characterisation of the GH gene cluster in a new-world monkey, the marmoset (Callithrix jacchus). J Mol Endocrinol 29:89–97
Wallis OC, Zhang Y-P, Wallis M (2001) Molecular evolution of GH in primates: characterisation of the GH genes from slow loris and marmoset defines an episode of rapid evolutionary change. J Mol Endocrinol 26:249–258
Wallis OC, Mac-Kwashie AO, Makri G, Wallis M (2005) Molecular evolution of prolactin in primates. J Mol Evol 60:606–614
Ye C, Li Y, Shi P, Zhang Y-P (2005) Molecular evolution of growth hormone gene family in old world monkeys and hominoids. Gene 350:183–192
Yi S, Bernat B, Pal G, Kossiakoff A, Li W-H (2002) Functional promiscuity of squirrel monkey growth hormone receptor toward both primate and nonprimate growth hormones. Mol Biol Evol 19:1083–1092
Acknowledgments
We thank Drs. Jeremy Slightom, Nick Mundy, Joseph Martial, and David Hunt for gifts of genomic libraries and DNA.
Author information
Authors and Affiliations
Corresponding author
Additional information
[Reviewing Editor: Nicolas Galtier]
Rights and permissions
About this article
Cite this article
Wallis, O.C., Wallis, M. Evolution of Growth Hormone in Primates: The GH Gene Clusters of the New World Monkeys Marmoset (Callithrix jacchus) and White-Fronted Capuchin (Cebus albifrons). J Mol Evol 63, 591–601 (2006). https://doi.org/10.1007/s00239-006-0039-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-006-0039-5