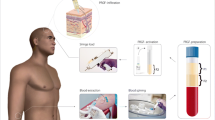
Abstract
Background
Androgenetic alopecia (AGA) is a common hair disorder. Finasteride and minoxidil have been approved by the Food and Drug Administration (FDA) for the treatment of AGA. However, some side effects have been related to the intake of these drugs. Recently, plasma rich in growth factors has emerged as a safe alternative for the management of pattern hair loss. The purposes of the present study are to evaluate and compare the efficacy and safety of plasma rich in growth factors (PRGFs) versus topical minoxidil treatment in AGA-affected patients.
Methods
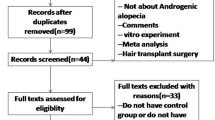
Following a retrospective case control design, 379 patients treated with either PRGF or minoxidil were included in this study. Diagnostic trichograms at baseline and 4 months after treatment were performed with the aim of analyzing the anagen/telogen hair change improvement.
Results
PRGF-treated patients showed higher anagen hair increase improvement compared to minoxidil-treated ones (6.9 ± 0.4 and 4.6 ± 0.5 %, respectively) (p < 0.05). Telogen hair decrease improvement was also higher in PRGF group (5.7 ± 0.3 and 2.6 ± 0.5 %, respectively) (p < 0.05). Global photographs showed an overall volume and quality hair improvement for both treatments. No adverse effects were reported.
Conclusions
This retrospective study shows that PRGF is a safe and effective treatment for AGA and evidences that PRGF exerts improved anagen/telogen results than topical minoxidil.
Level of evidence: Level IV, therapeutic study.




Similar content being viewed by others
References
Birch MP, Lalla SC, Messenger AG (2002) Female pattern hair loss. Clin Exp Dermatol 27(5):383–388
Rhodes T, Girman CJ, Savin RC, Kaufman KD, Guo S, Lilly FR, Siervogel RM, Chumlea WC (1998) Prevalence of male pattern hair loss in 18–49 year old men. Dermatol Surg 24(12):1330–1332
de Koning EB, Passchier J, Dekker FW (1990) Psychological problems with hair loss in general practice and the treatment policies of general practitioners. Psychol Rep 67(3 Pt 1):775–778
Hadshiew IM, Foitzik K, Arck PC, Paus R (2004) Burden of hair loss: stress and the underestimated psychosocial impact of telogen effluvium and androgenetic alopecia. J Invest Dermatol 123(3):455–457
Price VH (2003) Androgenetic alopecia in women. J Investig Dermatol Symp Proc 8(1):24–27
Nieves A, Garza LA (2014) Does prostaglandin D2 hold the cure to male pattern baldness? Exp Dermatol 23(4):224–227
Sasaki S, Hozumi Y, Kondo S (2005) Influence of prostaglandin F2α and its analogues on hair regrowth and follicular melanogenesis in a murine model. Exp Dermatol 14(5):323–328
Michelet J-F, Commo S, Billoni N, Mahé YF, Bernard BA (1997) Activation of cytoprotective prostaglandin synthase-1 by minoxidil as a possible explanation for its hair growth-stimulating effect. J Invest Dermatol 108(2):205–209
Olsen EA, Whiting D, Bergfeld W, Miller J, Hordinsky M, Wanser R, Zhang P, Kohut B (2007) A multicenter, randomized, placebo-controlled, double-blind clinical trial of a novel formulation of 5% minoxidil topical foam versus placebo in the treatment of androgenetic alopecia in men. J Am Acad Dermatol 57(5):767–774
Wood AJ, Price VH (1999) Treatment of hair loss. N Engl J Med 341(13):964–973
Friedman ES, Friedman PM, Cohen DE, Washenik K (2002) Allergic contact dermatitis to topical minoxidil solution: etiology and treatment. J Am Acad Dermatol 46(2):309–312
Rossi A, Cantisani C, Melis L, Iorio A, Scali E, Calvieri S (2012) Minoxidil use in dermatology, side effects and recent patents. Recent Pat Inflamm Allergy Drug Discov 6(2):130–136
Georgala S, Befon A, Maniatopoulou E, Georgala C (2007) Topical use of minoxidil in children and systemic side effects. Dermatology 214(1):101–102
Gnedeva K, Vorotelyak E, Cimadamore F, Cattarossi G, Giusto E, Terskikh VV, Terskikh AV (2015) Derivation of hair-inducing cell from human pluripotent stem cells. PLoS One 10(1):e0116892
Gay D, Kwon O, Zhang Z, Spata M, Plikus MV, Holler PD, Ito M, Yang Z, Treffeisen E, Kim CD (2013) Fgf9 from dermal [gamma][delta] T cells induces hair follicle neogenesis after wounding. Nat Med 19(7):916–923
Kang JS, Zheng Z, Choi MJ, Lee SH, Kim DY, Cho SB (2014) The effect of CD34+ cell-containing autologous platelet-rich plasma injection on pattern hair loss: a preliminary study. J Eur Acad Dermatol Venereol 28(1):72–79
Valente Duarte de Sousa IC, Tosti A (2013) New investigational drugs for androgenetic alopecia. Expert Opin Investig Drugs 22(5):573–589
Anitua E, Prado R, Azkargorta M, Rodriguez-Suarez E, Iloro I, Casado-Vela J, Elortza F, Orive G (2015) High-throughput proteomic characterization of plasma rich in growth factors (PRGF-Endoret)-derived fibrin clot interactome. J Tissue Eng Regen Med 9(11):E1–E12
Sanchez M, Fiz N, Azofra J, Usabiaga J, Aduriz Recalde E, Garcia Gutierrez A, Albillos J, Garate R, Aguirre JJ, Padilla S, Orive G, Anitua E (2012) A randomized clinical trial evaluating plasma rich in growth factors (PRGF-Endoret) versus hyaluronic acid in the short-term treatment of symptomatic knee osteoarthritis. Arthroscopy 28(8):1070–1078
Anitua E, Sanchez M, Nurden AT, Zalduendo M, de la Fuente M, Orive G, Azofra J, Andia I (2006) Autologous fibrin matrices: a potential source of biological mediators that modulate tendon cell activities. J Biomed Mater Res A 77(2):285–293
Panda A, Jain M, Vanathi M, Velpandian T, Khokhar S, Dada T (2012) Topical autologous platelet-rich plasma eyedrops for acute corneal chemical injury. Cornea 31(9):989–993
Anitua E, Pascual C, Pérez-Gonzalez R, Antequera D, Padilla S, Orive G, Carro E (2013) Intranasal delivery of plasma and platelet growth factors using PRGF-Endoret system enhances neurogenesis in a mouse model of Alzheimer’s disease. PLoS One 8(9):e73118
Anitua E, Begona L, Orive G (2013) Treatment of hemimandibular paresthesia in a patient with bisphosphonate-related osteonecrosis of the jaw (BRONJ) by combining surgical resection and PRGF-Endoret. Br J Oral Maxillofac Surg 51(8):e272–e274
Anitua E, Aguirre JJ, Algorta J, Ayerdi E, Cabezas AI, Orive G, Andia I (2008) Effectiveness of autologous preparation rich in growth factors for the treatment of chronic cutaneous ulcers. J Biomed Mater Res B Appl Biomater 84(2):415–421
Anitua E, Sánchez M, Sarabia R, Sanz J, Aguirre JJ, Orive G (2011) Eficacia y seguridad del PRGF (Plasma Rico en Factores de Crecimiento) en la regeneración cutánea facial. Ensayo clínico, randomizado y controlado con ácido hialurónico. AECEP 23–33
Orcajo B, Muruzabal F, Isasmendi MC, Gutierrez N, Sánchez M, Orive G, Anitua E (2011) The use of plasma rich in growth factors (PRGF-Endoret) in the treatment of a severe mal perforant ulcer in the foot of a person with diabetes. Diabetes Res Clin Pract 93(2):e65–e67
Diaz-Ley B, Cuevast J, Alonso-Castro L, Calvo MI, Rios-Buceta L, Orive G, Anitua E, Jaen P (2015) Benefits of plasma rich in growth factors (PRGF) in skin photodamage: clinical response and histological assessment. Dermatol Ther 28(4):258–263
Anitua E, Sanchez M, Zalduendo MM, de la Fuente M, Prado R, Orive G, Andia I (2009) Fibroblastic response to treatment with different preparations rich in growth factors. Cell Prolif 42(2):162–170
Sinclair R (1998) Fortnightly review: male pattern androgenetic alopecia. Br Med J 317(7162):865
García‐Rodiño S, Espasandín‐Arias M, Suárez‐Peñaranda JM, Rodríguez‐Granados MT, Vázquez‐Veiga H, Fernández‐Redondo V (2015) Persisting allergic patch test reaction to minoxidil manifested as cutaneous lymphoid hyperplasia. Contact Dermatitis 72(6):413–416
Hagemann T, Schlütter‐Böhmer B, Allam JP, Bieber T, Novak N (2005) Positive lymphocyte transformation test in a patient with allergic contact dermatitis of the scalp after short‐term use of topical minoxidil solution. Contact Dermatitis 53(1):53–55
Blumeyer A, Tosti A, Messenger A, Reygagne P, Del Marmol V, Spuls PI, Trakatelli M, Finner A, Kiesewetter F, Trueb R, Rzany B, Blume-Peytavi U, European Dermatology F (2011) Evidence-based (S3) guideline for the treatment of androgenetic alopecia in women and in men. J Dtsch Dermatol Ges 9 Suppl 6(s6):S1–S57
Hsu C-L, Liu J-S, Lin A-C, Yang C-H, Chung W-H, Wu W-G (2014) Minoxidil may suppress androgen receptor-related functions. Oncotarget 5(8):2187
Anitua E, Zalduendo MM, Prado R, Alkhraisat MH, Orive G (2015) Morphogen and proinflammatory cytokine release kinetics from PRGF-Endoret fibrin scaffolds: evaluation of the effect of leukocyte inclusion. J Biomed Mater Res A 103(3):1011–1020
Trink A, Sorbellini E, Bezzola P, Rodella L, Rezzani R, Ramot Y, Rinaldi F (2013) A randomized, double-blind, placebo- and active-controlled, half-head study to evaluate the effects of platelet-rich plasma on alopecia areata. Br J Dermatol 169(3):690–694
Uebel CO, da Silva JB, Cantarelli D, Martins P (2006) The role of platelet plasma growth factors in male pattern baldness surgery. Plast Reconstr Surg 118(6):1458–1466, discussion 1467
Paus R, Cotsarelis G (1999) The biology of hair follicles. N Engl J Med 341(7):491–497
Serrano-Falcón C, Fernández-Pugnaire M, Serrano-Ortega S (2013) Evaluación del pelo y cuero cabelludo: tricograma. Actas Dermosifiliogr 104(10):867–876
Cervelli V, Garcovich S, Bielli A, Cervelli G, Curcio BC, Scioli MG, Orlandi A, Gentile P (2014) The effect of autologous activated platelet rich plasma (AA-PRP) injection on pattern hair loss: clinical and histomorphometric evaluation. Biomed Res Int 2014:760709
Schiavone G, Raskovic D, Greco J, Abeni D (2014) Platelet-rich plasma for androgenetic alopecia: a pilot study. Dermatol Surg 40(9):1010–1019
Sclafani AP (2014) Platelet-rich fibrin matrix (PRFM) for androgenetic alopecia. Facial Plast Surg 30(2):219–224
Betsi E-E, Germain E, Kalbermatten DF, Tremp M, Emmenegger V (2013) Platelet-rich plasma injection is effective and safe for the treatment of alopecia. Eur J Plast Surg 36(7):407–412
Gkini M-A, Kouskoukis A-E, Tripsianis G, Rigopoulos D, Kouskoukis K (2014) Study of platelet-rich plasma injections in the treatment of androgenetic alopecia through an one-year period. J Cutan Aesthet Surg 7(4):213
Rastegar H, Ahmadi Ashtiani H, Aghaei M, Ehsani A, Barikbin B (2013) Combination of herbal extracts and platelet-rich plasma induced dermal papilla cell proliferation: involvement of ERK and Akt pathways. J Cosmet Dermatol 12(2):116–122
Miao Y, Sun YB, Sun XJ, Du BJ, Jiang JD, Hu ZQ (2013) Promotional effect of platelet-rich plasma on hair follicle reconstitution in vivo. Dermatol Surg 39(12):1868–1876
Lee SH, Zheng Z, Kang JS, Kim DY, Oh SH, Cho SB (2015) Therapeutic efficacy of autologous platelet-rich plasma and polydeoxyribonucleotide on female pattern hair loss. Wound Repair Regen 23(1):30–36
Takikawa M, Nakamura S, Nakamura S, Ishirara M, Kishimoto S, Sasaki K, Yanagibayashi S, Azuma R, Yamamoto N, Kiyosawa T (2011) Enhanced effect of platelet-rich plasma containing a new carrier on hair growth. Dermatol Surg 37(12):1721–1729
Anitua E, Sánchez M, Prado R, Orive G (2014) The type of platelet-rich plasma may influence the safety of the approach. Knee Surg Sports Traumatol Arthrosc 22(7):1708–1709
Jang JH (2005) Stimulation of human hair growth by the recombinant human keratinocyte growth factor-2 (KGF-2). Biotechnol Lett 27(11):749–752
Akiyama M, Smith LT, Holbrook KA (1996) Growth factor and growth factor receptor localization in the hair follicle bulge and associated tissue in human fetus. J Invest Dermatol 106(3):391–396
Takakura N, Yoshida H, Kunisada T, Nishikawa S, Nishikawa SI (1996) Involvement of platelet-derived growth factor receptor-alpha in hair canal formation. J Invest Dermatol 107(5):770–777
Li ZJ, Choi HI, Choi DK, Sohn KC, Im M, Seo YJ, Lee YH, Lee JH, Lee Y (2012) Autologous platelet-rich plasma: a potential therapeutic tool for promoting hair growth. Dermatol Surg 38(7 Pt 1):1040–1046
Yano K, Brown LF, Detmar M (2001) Control of hair growth and follicle size by VEGF-mediated angiogenesis. J Clin Invest 107(4):409–417
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Eduardo Anitua and Ander Pino are scientists at BTI Biotechnology Institute. This biotechnology company has developed the technology of plasma rich in growth factors. Navarro MR, Asín M, Martínez AM, Molina C, Navarro V, and Orive G declare that they have no conflict of interest.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This is a retrospective study, so for this type of study, formal consent is not required.
Funding
All authors state that this study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Patient consent
Patients provided written consent before their inclusion in the study. Additional consent was obtained for the use of their images.
Rights and permissions
About this article
Cite this article
Navarro, M.R., Asín, M., Martínez, M.A. et al. Management of androgenetic alopecia: a comparative clinical study between plasma rich in growth factors and topical minoxidil. Eur J Plast Surg 39, 173–180 (2016). https://doi.org/10.1007/s00238-015-1175-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00238-015-1175-1