Abstract
Purpose
To comprehensively summarize the clinical data and CT/MRI characteristics of thyroid-like low-grade nasopharyngeal papillary adenocarcinoma (TL-LGNPPA).
Methods
Twenty-seven lesions from 25 study articles identified through a systematic review and three lesions from our institution associated with TL-LGNPPA were evaluated.
Results
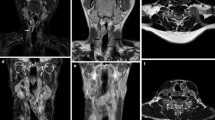
The mean age of the patients at diagnosis was 35.7 years, and the male-to-female ratio was nearly half. The chief complaint was nasal obstruction, followed by epistaxis. All patients underwent excision. None of the patients had neck nodes or distant metastases. All patients survived with no locoregional/distant recurrence during 3–93 months of follow-up. All lesions were located at the posterior edge of the nasal septum, attached to the nasopharyngeal parietal wall, and showed no laterality. The mean lesion diameter was 1.7 cm. The margins of lesions were well-defined and lobulated, followed by well-defined smooth margins. None of lesions were associated with parapharyngeal space or skull base destruction. All lesions were iso- and low-density on non-contrast CT. Adjacent skull base sclerosis was detected in 63.6% of lesions. High signal intensity on T2-weighted imaging and mostly iso-signal intensity on T1-weighted imaging compared to muscle tissue. Most lesions were heterogeneous and exhibited moderate contrast enhancement. Relatively large lesions (≥1.4 cm) tended to be more lobulated than smooth margins compared to relatively small lesions (<1.4 cm) (p = 0.016).
Conclusion
We summarized the clinical and radiological features of TL-LGNPPA to facilitate accurate diagnosis and appropriate management.




Similar content being viewed by others
References
Pineda-Daboin K, Neto A, Ochoa-Perez V, Luna MA (2006) Nasopharyngeal adenocarcinomas: a clinicopathologic study of 44 cases including immunohistochemical features of 18 papillary phenotypes. Ann Diagn Pathol 10:215–221. https://doi.org/10.1016/j.anndiagpath.2005.11.002
Oishi N, Kondo T, Nakazawa T et al (2014) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: case report and literature review. Pathol - Res Pract 210:1142–1145. https://doi.org/10.1016/j.prp.2014.04.020
Lai Y, Li W, Zhai C et al (2021) Low-grade nasopharyngeal papillary adenocarcinoma: a review of 28 patients in a single institution. Cancer Manag Res 13:1271–1278. https://doi.org/10.2147/CMAR.S288007
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Murad MH, Sultan S, Haffar S, Bazerbachi F (2018) Methodological quality and synthesis of case series and case reports. BMJ Evidence-Based Med 23:60 LP. https://doi.org/10.1136/bmjebm-2017-110853
Kurokawa R, Kurokawa M, Baba A et al (2021) Radiological features of head and neck mammary analogue secretory carcinoma: 11 new cases with a systematic review of 29 cases reported in 28 publications. Neuroradiology 63:1901–1911. https://doi.org/10.1007/s00234-021-02796-7
Baba A, Kurokawa R, Fukuda T et al (2022) Comprehensive radiological features of laryngeal sarcoidosis: cases series and systematic review. Neuroradiology 64:1239–1248. https://doi.org/10.1007/s00234-022-02922-z
Baba A, Kurokawa R, Fukuda T et al (2022) Radiological features of human papillomavirus-related multiphenotypic sinonasal carcinoma: systematic review and case series. Neuroradiology 64:2049–2058. https://doi.org/10.1007/s00234-022-03009-5
Baba A, Kurokawa R, Kurokawa M et al (2022) Imaging features of laryngeal chondrosarcomas: a case series and systematic review. J Neuroimaging 32:213–222. https://doi.org/10.1111/jon.12963
Baba A, Kurokawa R, Kurokawa M, Srinivasan A (2023) MRI features of sinonasal tract angiofibroma/juvenile nasopharyngeal angiofibroma: case series and systematic review. J Neuroimaging 33:675–687. https://doi.org/10.1111/jon.13116
Guo H, Zhang Z, Huang C et al (2023) Immunohistochemical characteristics of thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: a case report and review. Medicine (Baltimore) 102:e32655
Chen F, Zhang Q, Tian X et al (2022) Efficacy of endoscopic resection and intensity-modulated radiotherapy for thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: a case report. Transl Cancer Res 11:2408–2415
Lee J-H, Kown J-H (2020) Nasopharyngeal papillary adenocarcinoma: a cotton Swab appearance on computer tomography. Ear, Nose Throat J 101:28–30. https://doi.org/10.1177/0145561320942684
Karamitsou P, Poutoglidis A, Skliris JP et al (2022) An extremely rare nasopharyngeal malignant tumor: a case report. Cureus 14:e31444. https://doi.org/10.7759/cureus.31444
Jibril YN, Salisu AD, Mustapha MI, Oluwafemi OOA (2022) A rare case of low-grade nasopharyngeal papillary adenocarcinoma in a young adult woman: a case report from a tertiary hospital in Northwestern Nigeria. J West Afr Coll Surg 12:107–110
Yi L, Liu H (2021) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma with squamous differentiation in the posterior nasal septum: a rare case report. Medicine (Baltimore) 100:e28349
Compagnoni IM, Lamounier LA, Fontanini L et al (2021) Complete endoscopic resection of low-grade nasopharyngeal papillary adenocarcinoma: a case report. Braz J Otorhinolaryngol 87:237–240. https://doi.org/10.1016/j.bjorl.2020.10.004
Wang J, Luo S, Li Y, Zheng H (2021) Nasopharyngeal papillary adenocarcinoma harboring a fusion of ROS1 with GOPC: a case report. Medicine (Baltimore) 100:e24377
Theresia E, Dwianingsih EK, Rinonce HT et al (2020) Primary thyroid-like low-grade nasopharyngeal papillary adenocarcinoma in a 11-year-old male patient: the first case report in Indonesia. Hum Pathol Case Reports 21:200392. https://doi.org/10.1016/j.ehpc.2020.200392
Maocai L, Fuxing L, Lianqing L et al (2020) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: a case report. Medicine (Baltimore) 99:e21599
Wang J, Chen Z-Z, Lian C-L et al (2020) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: a case report and literature review. Transl Cancer Res 9:4457–4463
Lee SH, Kim H, Kim MJ et al (2020) Biphasic thyroid-like low-grade nasopharyngeal papillary adenocarcinoma with a prominent spindle cell component: a case report. Diagnostics 10:323
Takakura H, Hamashima T, Tachino H et al (2020) Clinicopathological features of thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: a case report and review of the literature. Front. Surg. 19:596796
Baumann KB, Betz SJ (2019) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma. Head Neck Pathol 13:661–663. https://doi.org/10.1007/s12105-018-0947-x
Huang F, Xiang X, Hong B et al (2019) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: a clinicopathologic study of five cases and a literature review. Am J Clin Pathol 152:582–589. https://doi.org/10.1093/ajcp/aqz082
Sourati A, Malekzadeh M, Rakhshan A (2019) Thyroid-like low-grade papillary adenocarcinoma of nasopharynx. BMJ Case Rep 12:e226949. https://doi.org/10.1136/bcr-2018-226949
Yokoi H, Terado Y, Fujiwara M et al (2018) Biphasic low-grade nasopharyngeal papillary adenocarcinoma: a case report and literature review. BMC Clin Pathol 18:10. https://doi.org/10.1186/s12907-018-0076-1
Ünsaler S, Başaran B, Aslan I, Yilmazbayhan D (2018) Endonasal endoscopic nasopharyngectomy for the treatment of nasopharyngeal papillary adenocarcinoma: report of a rare case. Int J Pediatr Otorhinolaryngol 104:51–53. https://doi.org/10.1016/j.ijporl.2017.10.041
Chen B-N, Wey S-L (2018) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma originating from the nasal septum. Int J Clin Exp Med 11:11326–11329
Oide T, Kadosono O, Matsushima J et al (2017) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma with squamous differentiation: a novel histological finding. Hum Pathol 70:43–48. https://doi.org/10.1016/j.humpath.2017.05.020
Yang S, Huang Y, Lu X et al (2017) Thyroid-like low-grade nasopharyngeal papillary carcinoma with a “biphasic” morphology: report of 3 cases and literature review. Int J Clin Exp Pathol 10:6038–6046
Horino T, Ichii O, Hamada-Ode K et al (2016) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: a case report. Mol Clin Oncol 5:693–696. https://doi.org/10.3892/mco.2016.1056
Borsetto D, Cazzador D, Prosenikliev V et al (2016) Nasopharyngeal thyroid-like low-grade papillary adenocarcinoma. B-ENT 12:235–240
Ryu J, Park WS, Jung Y-S (2013) Exclusive endoscopic resection of nasopharyngeal papillary adenocarcinoma via combined transnasal and transoral approach. Clin Exp Otorhinolaryngol 6:48–51. https://doi.org/10.3342/ceo.2013.6.1.48
Ohe C, Sakaida N, Tadokoro C et al (2010) Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: report of two cases: case report. Pathol Int 60:107–111. https://doi.org/10.1111/j.1440-1827.2009.02480.x
Wenig BM, Hyams VJ, Heffner DK (1988) Nasopharyngeal papillary adenocarcinoma: a clinicopathologic study of a low-grade carcinoma. Am J Surg Pathol 12:946–953
Carrizo F, Luna MA (2005) Thyroid transcription factor–1 expression in thyroid-like nasopharyngeal papillary adenocarcinoma: report of 2 cases. Ann Diagn Pathol 9:189–192. https://doi.org/10.1016/j.anndiagpath.2005.04.019
Bai R, Sun J, Xu Y et al (2022) Incidence and mortality trends of nasopharynx cancer from 1990 to 2019 in China: an age-period-cohort analysis. BMC Public Health 22:1351. https://doi.org/10.1186/s12889-022-13688-7
Kamran SC, Riaz N, Lee N (2015) Nasopharyngeal carcinoma. Surg Oncol Clin N Am 24:547–561. https://doi.org/10.1016/j.soc.2015.03.008
Ho FCH, Tham IWK, Earnest A et al (2012) Patterns of regional lymph node metastasis of nasopharyngeal carcinoma: a meta-analysis of clinical evidence. BMC Cancer 12:98. https://doi.org/10.1186/1471-2407-12-98
Qu W, Li S, Zhang M, Qiao Q (2020) Pattern and prognosis of distant metastases in nasopharyngeal carcinoma: a large-population retrospective analysis. Cancer Med 9:6147–6158. https://doi.org/10.1002/cam4.3301
Chen S, Yang D, Liao X et al (2022) Failure patterns of recurrence and metastasis after intensity-modulated radiotherapy in patients with nasopharyngeal carcinoma: results of a multicentric clinical study. Front. Oncol. 11:693199
American Cancer Society Survival Rates for Nasopharyngeal Cancer. https://www.cancer.org/cancer/types/nasopharyngeal-cancer/detection-diagnosis-staging/survival-rates.html. Accessed 15 Sept 2023
Razek AAKA, King A (2012) MRI and CT of nasopharyngeal carcinoma. Am J Roentgenol 198:11–18. https://doi.org/10.2214/AJR.11.6954
King AD, Wong LYS, Law BKH et al (2018) MR imaging criteria for the detection of nasopharyngeal carcinoma: discrimination of early-stage primary tumors from benign hyperplasia. Am J Neuroradiol 39:515 LP–515523. https://doi.org/10.3174/ajnr.A5493
Sireci F, Dispenza F, Lorusso F et al (2022) Tumours of nasal septum: a retrospective study of 32 patients. Int J Environ Res Public Health 19:1713
Yang BT, Li SP, Wang YZ et al (2013) Routine and dynamic MR imaging study of lobular capillary hemangioma of the nasal cavity with comparison to inverting papilloma. Am J Neuroradiol 34:2202 LP–2202207. https://doi.org/10.3174/ajnr.A3523
Lee DG, Lee SK, Chang HW et al (2010) CT features of lobular capillary hemangioma of the nasal cavity. Am J Neuroradiol 31:749 LP–749754. https://doi.org/10.3174/ajnr.A1908
Motoori K, Takano H, Nakano K et al (2000) Pleomorphic adenoma of the nasal septum: MR features. Am J Neuroradiol 21:1948 LP–1941950
Baron S, Koka V, El Chater P et al (2014) Pleomorphic adenoma of the nasal septum. Eur Ann Otorhinolaryngol Head Neck Dis 131:139–141. https://doi.org/10.1016/j.anorl.2013.03.007
Kelly JH, Joseph M, Carroll E et al (1980) Inverted papilloma of the nasal septum. Arch Otolaryngol 106:767–771. https://doi.org/10.1001/archotol.1980.00790360045012
Park I-H, Lee HC, Lee H-M (2013) Respiratory epithelial adenomatoid hamartoma originating from nasal septum. Clin Exp Otorhinolaryngol 6:45–47. https://doi.org/10.3342/ceo.2013.6.1.45
Tong KN, Serra RM, Shih RY, Foss RD (2019) Seromucinous hamartoma of the nasal cavity. Head Neck Pathol 13:239–242. https://doi.org/10.1007/s12105-018-0914-6
King AD (2022) MR Imaging of nasopharyngeal carcinoma. Magn Reson Imaging Clin N Am 30:19–33. https://doi.org/10.1016/j.mric.2021.06.015
Siebert SM, Gomez AJ, Liang T et al (2018) Diagnostic performance of margin features in thyroid nodules in prediction of malignancy. Am J Roentgenol 210:860–865. https://doi.org/10.2214/AJR.17.18787
Slone HW, Blake JJ, Shah R et al (2005) CT and MRI findings of intracranial lymphoma. Am J Roentgenol 184:1679–1685. https://doi.org/10.2214/ajr.184.5.01841679
Horiuchi D, Shimono T, Tatekawa H et al (2022) Frequency and imaging features of the adjacent osseous changes of salivary gland carcinomas in the head and neck region. Neuroradiology 64:1869–1877. https://doi.org/10.1007/s00234-022-02972-3
Koyasu S, Sakurada H, Nakamoto Y (2023) Letter regarding the article “Frequency and imaging features of adjacent osseous changes of salivary gland carcinomas in the head and neck region” by Horiuchi and Shimono et al. Neuroradiology 65:1315. https://doi.org/10.1007/s00234-023-03199-6
Srinivasan A, Dvorak R, Perni K et al (2008) Differentiation of benign and malignant pathology in the head and neck using 3T apparent diffusion coefficient values: early experience. Am J Neuroradiol 29:40 LP – 44. https://doi.org/10.3174/ajnr.A0743
Baba A, Kurokawa R, Kurokawa M et al (2022) ADC for differentiation between posttreatment changes and recurrence in head and neck cancer: a systematic review and meta-analysis. Am J Neuroradiol 43:442 LP – 447. https://doi.org/10.3174/ajnr.A7431
Baba A, Kurokawa R, Rawie E et al (2022) Normalized parameters of dynamic contrast-enhanced perfusion MRI and DWI-ADC for differentiation between posttreatment changes and recurrence in head and neck cancer. Am J Neuroradiol 43:1184–1189. https://doi.org/10.3174/ajnr.A7567
Baba A, Kurokawa R, Kurokawa M et al (2023) Dynamic contrast-enhanced MRI parameters and normalized ADC values could aid differentiation of skull base osteomyelitis from nasopharyngeal cancer. Am J Neuroradiol 44:84–78
Yan D-F, Zhang W-B, Ke S-B et al (2017) The prognostic value of pretreatment tumor apparent diffusion coefficient values in nasopharyngeal carcinoma. BMC Cancer 17:678. https://doi.org/10.1186/s12885-017-3658-x
Razek AAKA, Sadek AG, Kombar OR et al (2008) Role of apparent diffusion coefficient values in differentiation between malignant and benign solitary thyroid nodules. Am J Neuroradiol 29:563–568. https://doi.org/10.3174/ajnr.A0849
Acknowledgments
None.
Funding
No funding was received for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None to declare.
Ethics approval
We obtained our institutional review board approval for including cases to this systematic review study and performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Informed consent was waived of due to the retrospective nature of the study.
Consent for publication
All authors consent to the publication of the manuscript in Neuroradiology.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Baba, A., Matsushima, S., Kessoku, H. et al. Radiological features of thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: case series and systematic review. Neuroradiology 66, 249–259 (2024). https://doi.org/10.1007/s00234-023-03254-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-023-03254-2