Abstract
Purpose
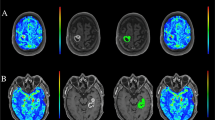
Gadolinium-based contrast agent (GBCA) effect on automated segmentation algorithms of subcortical gray matter (GM) is not fully known. The aim of this study is to determine gadolinium effect on the segmentation of the thalamus and whole brain tissue using different automated segmentation techniques.
Methods
Eighty-four multiple sclerosis (MS) patients underwent an MRI acquisition of two 3DT1-weighted sequences with and without gadolinium injection among which 10 were excluded after image quality check. Manual thalamic segmentation considered as gold standard was performed on unenhanced T1 images. volBrain and FSL-Anat were used to automatically segment the thalamus on both enhanced and unenhanced T1 and the degree of similitude (DICE) values were compared between manual and automatic segmentations. Whole brain tissue segmentation (GM, white matter (WM), and lateral ventricles (LV)) was also performed using SIENAX. A paired samples t test was applied to test the significance of DICE value differences between the thalamic manual and automatic segmentations of both enhanced and unenhanced T1 images.
Results
Significant differences (FSL-Anat 1.474% p < 0.001 and volBrain 1.990% p < 0.001) in DICE between thalamic manual and automatic segmentations on both enhanced and unenhanced images were observed. Automatic tissue segmentation showed a mean DICE of 81.5%, with LV having the lowest DICE value (74.2%). When compared to tissue segmentations, automatic thalamic segmentations by FSL-Anat or volBrain demonstrated a higher degree of similitude (FSL-Anat = 91.7% and volBrain = 90.7%).
Conclusion
Gadolinium has a significant effect on subcortical GM segmentation. Although significant, the observed subtle changes could be considered acceptable when used for region-based analysis in perfusion or diffusion imaging.


Similar content being viewed by others
Abbreviations
- BBB:
-
Brain blood barrier
- BPF:
-
Brain parenchymal fraction
- CSF:
-
Cerebro-spinal fluid
- DICE:
-
Degree of similitude
- DTI:
-
Diffusion tensor imaging
- DWI:
-
Diffusion weighted imaging
- FLAIR:
-
Fluid-attenuated inversion recovery
- GBCA:
-
Gadolinium-based contrast agent
- GM:
-
Gray matter
- LV:
-
Lateral ventricles
- MRI:
-
Magnetic resonance imaging
- MS:
-
Multiple sclerosis
- WM:
-
White matter
References
Durand-Dubief F, Belaroussi B, Armspach JPP et al (2012) Reliability of longitudinal brain volume loss measurements between 2 sites in patients with multiple sclerosis: comparison of 7 quantification techniques. Am J Neuroradiol 33:1918–1924. https://doi.org/10.3174/ajnr.A3107
Zhou Z, Lu Z-R (2013) Gadolinium-based contrast agents for magnetic resonance cancer imaging. Wiley Interdiscip Rev Nanomedicine Nanobiotechnology 5:1–18. https://doi.org/10.1002/wnan.1198
Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, Fujihara K, Havrdova E, Hutchinson M, Kappos L, Lublin FD, Montalban X, O'Connor P, Sandberg-Wollheim M, Thompson AJ, Waubant E, Weinshenker B, Wolinsky JS (2011) Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 69:292–302. https://doi.org/10.1002/ana.22366
McDonald WI, Compston A, Edan G et al (2001) Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol 50:121–127
Calabrese M, Agosta F, Rinaldi F, Mattisi I, Grossi P, Favaretto A, Atzori M, Bernardi V, Barachino L, Rinaldi L, Perini P, Gallo P, Filippi M (2009) Cortical lesions and atrophy associated with cognitive impairment in relapsing-remitting multiple sclerosis. Arch Neurol 66:1144–1150. https://doi.org/10.1001/archneurol.2009.174
Štecková T, Hluštík P, Sládková V, Odstrčil F, Mareš J, Kaňovský P (2014) Thalamic atrophy and cognitive impairment in clinically isolated syndrome and multiple sclerosis. J Neurol Sci 342:62–68. https://doi.org/10.1016/j.jns.2014.04.026
Riccitelli G, Rocca MA, Pagani E, Rodegher ME, Rossi P, Falini A, Comi G, Filippi M (2011) Cognitive impairment in multiple sclerosis is associated to different patterns of gray matter atrophy according to clinical phenotype. Hum Brain Mapp 32:1535–1543. https://doi.org/10.1002/hbm.21125
Jovicich J, Czanner S, Han X, Salat D, van der Kouwe A, Quinn B, Pacheco J, Albert M, Killiany R, Blacker D, Maguire P, Rosas D, Makris N, Gollub R, Dale A, Dickerson BC, Fischl B (2009) MRI-derived measurements of human subcortical, ventricular and intracranial brain volumes: reliability effects of scan sessions, acquisition sequences, data analyses, scanner upgrade, scanner vendors and field strengths. Neuroimage 46:177–192. https://doi.org/10.1016/j.neuroimage.2009.02.010
Warntjes JBM, Tisell A, Landtblom AM, Lundberg P (2014) Effects of gadolinium contrast agent administration on automatic brain tissue classification of patients with multiple sclerosis. Am J Neuroradiol 35:1330–1336. https://doi.org/10.3174/ajnr.A3890
Cotton F, Kremer S, Hannoun S, Vukusic S, Dousset V (2015) OFSEP, a nationwide cohort of people with multiple sclerosis: consensus minimal MRI protocol. J Neuroradiol 42:133–140. https://doi.org/10.1016/j.neurad.2014.12.001
Manjón JV, Coupé P (2016) volBrain: an online MRI brain volumetry system. Front Neuroinform 10:30. https://doi.org/10.3389/fninf.2016.00030
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TEJ, Johansen-Berg H, Bannister PR, de Luca M, Drobnjak I, Flitney DE, Niazy RK, Saunders J, Vickers J, Zhang Y, de Stefano N, Brady JM, Matthews PM (2004) Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23:S208–S219. https://doi.org/10.1016/j.neuroimage.2004.07.051
Smith SM, Zhang Y, Jenkinson M, Chen J, Matthews PM, Federico A, de Stefano N (2002) Accurate, robust, and automated longitudinal and cross-sectional brain change analysis. Neuroimage 17:479–489. https://doi.org/10.1006/nimg.2002.1040
Dice LR (1945) Measures of the amount of ecologic association between species. Ecology 26:297–302. https://doi.org/10.2307/1932409
Rotge J-Y, Guehl D, Dilharreguy B, Tignol J, Bioulac B, Allard M, Burbaud P, Aouizerate B (2009) Meta-analysis of brain volume changes in obsessive-compulsive disorder. Biol Psychiatry 65:75–83. https://doi.org/10.1016/j.biopsych.2008.06.019
González-Villà S, Valverde S, Cabezas M, Pareto D, Vilanova JC, Ramió-Torrentà L, Rovira À, Oliver A, Lladó X (2017) Evaluating the effect of multiple sclerosis lesions on automatic brain structure segmentation. NeuroImage Clin 15:228–238. https://doi.org/10.1016/j.nicl.2017.05.003
Zivadinov R, Cox JL (2007) Neuroimaging in multiple sclerosis. Int Rev Neurobiol 79:449–474. https://doi.org/10.1016/S0074-7742(07)79020-7
Valverde S, Oliver A, Lladó X (2014) A white matter lesion-filling approach to improve brain tissue volume measurements. NeuroImage Clin 6:86–92. https://doi.org/10.1016/j.nicl.2014.08.016
Bishop CA, Newbould RD, Lee JS et al (2017) Analysis of ageing-associated grey matter volume in patients with multiple sclerosis shows excess atrophy in subcortical regions. NeuroImage Clin 13:9–15. https://doi.org/10.1016/j.nicl.2016.11.005
Doche E, Lecocq A, Maarouf A, Duhamel G, Soulier E, Confort-Gouny S, Rico A, Guye M, Audoin B, Pelletier J, Ranjeva JP, Zaaraoui W (2017) Hypoperfusion of the thalamus is associated with disability in relapsing remitting multiple sclerosis. J Neuroradiol 44:158–164. https://doi.org/10.1016/j.neurad.2016.10.001
Hannoun S, Durand-Dubief F, Confavreux C, Ibarrola D, Streichenberger N, Cotton F, Guttmann CRG, Sappey-Marinier D (2012) Diffusion tensor-MRI evidence for extra-axonal neuronal degeneration in caudate and thalamic nuclei of patients with multiple sclerosis. AJNR Am J Neuroradiol 33:1363–1368. https://doi.org/10.3174/ajnr.A2983
Vågberg M, Lindqvist T, Ambarki K, Warntjes JBM, Sundström P, Birgander R, Svenningsson A (2013) Automated determination of brain parenchymal fraction in multiple sclerosis. AJNR Am J Neuroradiol 34:498–504. https://doi.org/10.3174/ajnr.A3262
Battaglini M, Jenkinson M, De Stefano N (2012) Evaluating and reducing the impact of white matter lesions on brain volume measurements. Hum Brain Mapp 33:2062–2071. https://doi.org/10.1002/hbm.21344
Montagne A, Toga AW, Zlokovic BV (2016) Blood-brain barrier permeability and gadolinium: benefits and potential pitfalls in research. JAMA Neurol 73:13–14. https://doi.org/10.1001/jamaneurol.2015.2960
Khonsary S (2017) Guyton and hall: textbook of medical physiology. Surg Neurol Int 8:275. https://doi.org/10.4103/sni.sni_327_17
Acknowledgements
The authors would like to thank the Nehme and Therese Tohme Multiple Sclerosis Center at the American University of Beirut Medical Center for funding this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the Nehme and Therese Thome Multiple Sclerosis Center, American University of Beirut Medical Center.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Informed consent
For this type of retrospective study formal consent is not required.
Electronic supplementary material
Supplementary Table 1
(DOCX 31 kb)
Rights and permissions
About this article
Cite this article
Hannoun, S., Baalbaki, M., Haddad, R. et al. Gadolinium effect on thalamus and whole brain tissue segmentation. Neuroradiology 60, 1167–1173 (2018). https://doi.org/10.1007/s00234-018-2082-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-018-2082-5