Abstract
Purpose
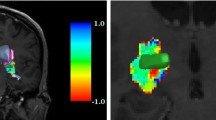
For safe deep brain stimulation (DBS) planning, an accurate visualization and localization of vessels is mandatory. Contrast enhanced (ce) MRI depicts both arteries and veins. Computed tomography angiography (CTA) detects arteries with high geometric accuracy. We routinely combine both modalities for DBS planning.
Methods
A total of 222 trajectories in a consecutive series of 113 patients who underwent DBS operations were included. In all trajectories, the number of veins and arteries in a 10-mm diameter around the planned trajectory were counted in a ceMRI and a CTA. If a vessel was visible in both modalities, the distance was measured.
Results
A total of 370 vessels were counted. Two hundred forty vessels (65%) were visible in both modalities. With 134 of the vessels, we detected a difference of the vessel’s location with an average distance of 1.24 mm (SD 0.58). Eighty vessels (22%) were visible only in the ceMRI, 50 vessels (13%) only in the CTA. We had four bleedings (1.8% per lead) of which one was symptomatic (0.45%).
Conclusion
The majority of vessels were visible in both modalities; however, in more than half of these cases, the location was not identical. Here, the location in the CTA can be regarded as the ground truth. Moreover, both the CTA and the ceMRI depicted vessels not seen in the other imaging modality. We therefore assume that the combination of both imaging modalities for DBS planning increases the chance to detect vascular conflicts along the trajectory, thus reducing the risk of intracranial bleeding.




Similar content being viewed by others
References
Schuepbach WMM, Rau J, Knudsen K, Volkmann J, Krack P, Timmermann L, Hälbig TD, Hesekamp H, Navarro SM, Meier N, Falk D, Mehdorn M, Paschen S, Maarouf M, Barbe MT, Fink GR, Kupsch A, Gruber D, Schneider GH, Seigneuret E, Kistner A, Chaynes P, Ory-Magne F, Brefel Courbon C, Vesper J, Schnitzler A, Wojtecki L, Houeto JL, Bataille B, Maltête D, Damier P, Raoul S, Sixel-Doering F, Hellwig D, Gharabaghi A, Krüger R, Pinsker MO, Amtage F, Régis JM, Witjas T, Thobois S, Mertens P, Kloss M, Hartmann A, Oertel WH, Post B, Speelman H, Agid Y, Schade-Brittinger C, Deuschl G, EARLYSTIM Study Group (2013) Neurostimulation for Parkinson’s disease with early motor complications. N Engl J Med 368:610–622. https://doi.org/10.1056/NEJMoa1205158
Schlaepfer TE, Bewernick BH, Kayser S, Hurlemann R, Coenen VA (2014) Deep brain stimulation of the human reward system for major depression—rationale, outcomes and outlook. Neuropsychopharmacology 39:1303–1314. https://doi.org/10.1038/npp.2014.28
Ben-Haim S, Asaad WF, Gale JT, Eskandar EN (2009) Risk factors for hemorrhage during microelectrode-guided deep brain stimulation and the introduction of an improved microelectrode design. Neurosurgery 64:754–763. https://doi.org/10.1227/01.NEU.0000339173.77240.34
Binder DK, Rau G, Starr PA (2004) Hemorrhagic complications of microelectrode-guided deep brain stimulation. Stereotact Funct Neurosurg 80:28–31. https://doi.org/10.1159/000075156
Binder DK, Rau GM, Starr PA (2005) Risk factors for hemorrhage during microelectrode-guided deep brain stimulator implantation for movement disorders. Neurosurgery 56:722–732 discussion 722–732
Seijo FJ, Alvarez-Vega MA, Gutierrez JC, Fdez-Glez F, Lozano B (2007) Complications in subthalamic nucleus stimulation surgery for treatment of Parkinson’s disease. Review of 272 procedures. Acta Neurochir 149:867–876. https://doi.org/10.1007/s00701-007-1267-1
Terao T, Takahashi H, Yokochi F, Taniguchi M, Okiyama R, Hamada I (2003) Hemorrhagic complication of stereotactic surgery in patients with movement disorders. J Neurosurg 98:1241–1246. https://doi.org/10.3171/jns.2003.98.6.1241
Voges J, Hilker R, Bötzel K, Kiening KL, Kloss M, Kupsch A, Schnitzler A, Schneider GH, Steude U, Deuschl G, Pinsker MO (2007) Thirty days complication rate following surgery performed for deep-brain-stimulation. Mov Disord 22:1486–1489. https://doi.org/10.1002/mds.21481
Patel DM, Walker HC, Brooks R, Omar N, Ditty B, Guthrie BL (2015) Adverse events associated with deep brain stimulation for movement disorders analysis of 510 consecutive cases. Oper Neurosurg 11:190–199. https://doi.org/10.1227/NEU.0000000000000659
Sansur CA, Frysinger RC, Pouratian N, Fu KM, Bittl M, Oskouian RJ, Laws ER, Elias WJ (2007) Incidence of symptomatic hemorrhage after stereotactic electrode placement. J Neurosurg 107:998–1003. https://doi.org/10.3171/JNS-07/11/0998
Chen T, Mirzadeh Z, Chapple K, Lambert M, Ponce FA (2016) Complication rates, lengths of stay, and readmission rates in “awake” and “asleep” deep brain simulation. J Neurosurg 127:1–10. https://doi.org/10.3171/2016.6.JNS152946
Wang X, Wang J, Zhao H, Li N, Ge S, Chen L, Li J, Jing J, Su M, Zheng Z, Zhang J, Gao G, Wang X (2016) Clinical analysis and treatment of symptomatic intracranial hemorrhage after deep brain stimulation surgery. Br J Neurosurg 0:1–6. doi: https://doi.org/10.1080/02688697.2016.1244252, 31
Nowell M, Rodionov R, Diehl B, Wehner T, Zombori G, Kinghorn J, Ourselin S, Duncan J, Miserocchi A, McEvoy A (2014) A novel method for implementation of frameless StereoEEG in epilepsy surgery. Neurosurgery 10:525–534. https://doi.org/10.1227/NEU.0000000000000544
Gilard V, Proust F, Gerardin E, Lebas A, Chastan N, Fréger P, Parain D, Derrey S (2016) Usefulness of multidetector-row computerized tomographic angiography for the surgical planning in stereoelectroencephalography. Diagn Interv Imaging 97:333–337. https://doi.org/10.1016/j.diii.2015.10.001
Coenen VA, Rijntjes M, Prokop T, Piroth T, Amtage F, Urbach H, Reinacher PC (2016) One-pass deep brain stimulation of dentato-rubro-thalamic tract and subthalamic nucleus for tremor-dominant or equivalent type Parkinson’s disease. Acta Neurochir 158:773–781. https://doi.org/10.1007/s00701-016-2725-4
Gonzalez-Martinez J, Mullin J, Vadera S, Bulacio J, Hughes G, Jones S, Enatsu R, Najm I (2014) Stereotactic placement of depth electrodes in medically intractable epilepsy. J Neurosurg 120:639–644. https://doi.org/10.3171/2013.11.JNS13635
Blatt DR, Roper SN, Friedman WA (1997) Invasive monitoring of limbic epilepsy using stereotactic depth and subdural strip electrodes: surgical technique. Surg Neurol 48:74–79. https://doi.org/10.1016/S0090-3019(96)00277-7
Burneo JG, Steven DA, McLachlan RS, Parrent AG (2006) Morbidity associated with the use of intracranial electrodes for epilepsy surgery. Can J Neurol Sci J Can Sci Neurol 33:223–227
Guenot M, Isnard J, Ryvlin P, Fischer C, Ostrowsky K, Mauguiere F, Sindou M (2001) Neurophysiological monitoring for epilepsy surgery: the Talairach SEEG method. Stereotact Funct Neurosurg 77:29–32. https://doi.org/10.1159/000064595
Behrens E, Schramm J, Zentner J, König R (1997) Surgical and neurological complications in a series of 708 epilepsy surgery procedures. Neurosurgery 41:1–9 discussion 9–10
Østergaard K, Sunde N, Dupont E (2002) Effects of bilateral stimulation of the subthalamic nucleus in patients with severe Parkinson’s disease and motor fluctuations. Mov Disord 17:693–700. https://doi.org/10.1002/mds.10188
Starr PA, Christine CW, Theodosopoulos PV, Lindsey N, Byrd D, Mosley A, Marks WJ Jr (2002) Implantation of deep brain stimulators into subthalmic nucleus: technical approach and magnetic imaging—verified electrode locations. J Neurosurg 97:370–387. https://doi.org/10.3171/jns.2002.97.2.0370
Sterio D, Zonenshayn M, Mogilner AY et al (2002) Neurophysiological refinement of subthalamic nucleus targeting. Neurosurgery 50:58–67 discussion 67–69
Voges J, Volkmann J, Allert N, Lehrke R, Koulousakis A, Freund HJ, Sturm V (2002) Bilateral high-frequency stimulation in the subthalamic nucleus for the treatment of Parkinson disease: correlation of therapeutic effect with anatomical electrode position. J Neurosurg 96:269–279. https://doi.org/10.3171/jns.2002.96.2.0269
Rezai AR, Machado AG, Deogaonkar M, Azmi H, Kubu C, Boulis NM (2008) Surgery for movement disorders. Neurosurgery 62:SHC809–SHC839. https://doi.org/10.1227/01.neu.0000316285.52865.53
Acknowledgments
We thank Olivia Chorny for reviewing the manuscript and providing language support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Informed consent
For this type of retrospective study formal consent is not required.
Rights and permissions
About this article
Cite this article
Krüger, M.T., Coenen, V.A., Jenkner, C. et al. Combination of CT angiography and MRI in surgical planning of deep brain stimulation. Neuroradiology 60, 1151–1158 (2018). https://doi.org/10.1007/s00234-018-2079-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-018-2079-0