Abstract
Introduction
Acute or subacute cerebellar inflammation is mainly caused by postinfectious, toxic, neoplastic, vascular, or idiopathic processes and can result in cerebellar ataxia. Previous magnetic resonance (MR) studies in single patients who developed acute or subacute ataxia showed varying imaging features.
Methods
Eighteen patients presenting with acute and subacute onset of ataxia were included in this study. Cases of chronic-progressive/hereditary and noncerebellar causes (ischemia, multiple sclerosis lesions, metastasis, bleedings) were excluded. MR imaging findings were then matched with the clinical history of the patient.
Results
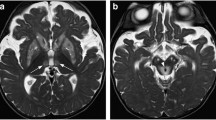
An underlying etiology for ataxic symptoms were found in 14/18 patients (postinfectious/infectious, paraneoplastic, autoimmune, drug-induced). In two of five patients without MR imaging findings and three of eight patients with minimal imaging features (cerebellar atrophy, slight signal alterations, and small areas of restricted diffusion), adverse clinical outcomes were documented. Of the five patients with prominent MR findings (cerebellar swelling, contrast enhancement, or broad signal abnormalities), two were lost to follow-up and two showed long-term sequelae.
Conclusion
No correlation was found between the presence of initial MRI findings in subacute or acute ataxia patients and their long-term clinical outcome. MR imaging was more flagrantly positive in cases due to encephalitis.




Similar content being viewed by others
References
Martinez-Gonzalez MJ, Martinez-Gonzalez S, Garcia-Ribes A, Mintegi-Raso S, Benito-Fernandez J, Prats-Vinas JM (2006) Acute onset ataxia in infancy: its aetiology, treatment and follow-up. Rev Neurol 42(6):321–324
Gieron-Korthals MA, Westberry KR, Emmanuel PJ (1994) Acute childhood ataxia: 10-year experience. J Child Neurol 9(4):381–384
Moses H, Nagel MA, Gilden DH (2006) Acute cerebellar ataxia in a 41 year old woman. Lancet Neurol 5(11):984–988. doi:10.1016/S1474-4422(06)70601-9
Cho TA, Schmahmann JD, Cunnane ME (2013) Case records of the Massachusetts General Hospital. Case 30-2013. A 19-year-old man with otalgia, slurred speech, and ataxia. N Engl J Med 369(13):1253–1261. doi:10.1056/NEJMcpc1214218
Whelan HT, Verma S, Guo Y, Thabet F, Bozarth X, Nwosu M, Katyayan A, Parachuri V, Spangler K, Ruggeri BE, Srivatsal S, Zhang G, Ashwal S (2013) Evaluation of the child with acute ataxia: a systematic review. Pediatr Neurol 49(1):15–24. doi:10.1016/j.pediatrneurol.2012.12.005
Desai J, Mitchell WG (2012) Acute cerebellar ataxia, acute cerebellitis, and opsoclonus-myoclonus syndrome. J Child Neurol 27(11):1482–1488. doi:10.1177/0883073812450318
Liu Y, Nuutinen J, Laakso MP, Karonen JO, Kononen M, Vanninen E, Kuikka JT, Vanninen RL (2010) Cerebellar apparent diffusion coefficient changes in patients with supratentorial ischemic stroke. Acta Neurol Scand 122(5):316–322. doi:10.1111/j.1600-0404.2009.01289.x
Reiss KA, Bailey JR, Pham PA, Gallant JE (2010) Raltegravir-induced cerebellar ataxia. Aids 24(17):2757. doi:10.1097/QAD.0b013e32833fca42
De Bruecker Y, Claus F, Demaerel P, Ballaux F, Sciot R, Lagae L, Buyse G, Wilms G (2004) MRI findings in acute cerebellitis. Eur Radiol 14(8):1478–1483. doi:10.1007/s00330-004-2247-y
Carceller Lechon F, Duat Rodriguez A, Sirvent Cerda SI, Khabra K, de Prada I, Garcia-Penas JJ, Madero Lopez L (2013) Hemicerebellitis: report of three paediatric cases and review of the literature. Eur J Paediatr Neurol. doi:10.1016/j.ejpn.2013.12.004
Hennes E, Zotter S, Dorninger L, Hartmann H, Hausler M, Huppke P, Jacobs J, Kraus V, Makowski C, Schlachter K, Ulmer H, van Baalen A, Koch J, Gotwald T, Rostasy K (2012) Long-term outcome of children with acute cerebellitis. Neuropediatrics 43(5):240–248. doi:10.1055/s-0032-1324732
Dogulu F, Onk A, Kaymaz M, Kardes O, Baykaner MK (2003) Acute cerebellitis with hydrocephalus. Neurology 60(10):1717
Lee KY, Lee YJ, Kim TH, Cheon DS, Nam SO (2014) Clinico-radiological spectrum in enterovirus 71 infection involving the central nervous system in children. J Clin Neurosci 21(3):416–420. doi:10.1016/j.jocn.2013.04.032
Takanashi J, Miyamoto T, Ando N, Kubota T, Oka M, Kato Z, Hamano S, Hirabayashi S, Kikuchi M, Barkovich AJ (2010) Clinical and radiological features of rotavirus cerebellitis. AJNR Am J Neuroradiol 31(9):1591–1595. doi:10.3174/ajnr.A2131
Kobayashi S, Negishi Y, Ando N, Ito T, Nakano M, Togari H, Wakuda M, Taniguchi K (2010) Two patients with acute rotavirus encephalitis associated with cerebellar signs and symptoms. Eur J Pediatr 169(10):1287–1291. doi:10.1007/s00431-010-1202-y
Hata A, Fujita M, Morishima T, Kumakura A, Hata D (2008) Acute cerebellar ataxia associated with primary human herpesvirus-6 infection: a report of two cases. J Paediatr Child Health 44(10):607–609. doi:10.1111/j.1440-1754.2008.01395.x
Burton JM, Kern RZ, Halliday W, Mikulis D, Brunton J, Fearon M, Pepperell C, Jaigobin C (2004) Neurological manifestations of West Nile virus infection. Can J Neurol Sci 31(2):185–193
Fellgiebel A, Siessmeier T, Winterer G, Luddens H, Mann K, Schmidt LG, Bartenstein P (2004) Increased cerebellar PET glucose metabolism corresponds to ataxia in Wernicke-Korsakoff syndrome. Alcohol Alcohol 39(2):150–153
Nalini A, Kovoor JM, Dawn R, Kallur KG (2009) Marchiafava-Bignami disease: two cases with magnetic resonance imaging and positron emission tomography scan findings. Neurol India 57(5):644–648. doi:10.4103/0028-3886.57813
Appenzeller S, Cendes F, Costallat LT (2008) Cerebellar ataxia in systemic lupus erythematosus. Lupus 17(12):1122–1126. doi:10.1177/0961203308093071
Demir E, Aksoy A, Anlar B, Sonmez FM (2007) Atypical presentations of SSPE: a clinical study in four cases. Turk J Pediatr 49(3):295–300
Niethammer M, Ford B (2007) Permanent lithium-induced cerebellar toxicity: three cases and review of literature. Mov Disord 22(4):570–573. doi:10.1002/mds.21318
Baizabal-Carvallo JF, Stocco A, Muscal E, Jankovic J (2013) The spectrum of movement disorders in children with anti-NMDA receptor encephalitis. Mov Disord 28(4):543–547. doi:10.1002/mds.25354
Sawaishi Y, Takada G (2002) Acute cerebellitis. Cerebellum 1(3):223–228. doi:10.1080/14734220260418457
Hoftberger R, Sabater L, Ortega A, Dalmau J, Graus F (2013) Patient with homer-3 antibodies and cerebellitis. JAMA Neurol 70(4):506–509. doi:10.1001/jamaneurol.2013.1955
Marignier R, Chenevier F, Rogemond V, Sillevis Smitt P, Renoux C, Cavillon G, Androdias G, Vukusic S, Graus F, Honnorat J, Confavreux C (2010) Metabotropic glutamate receptor type 1 autoantibody-associated cerebellitis: a primary autoimmune disease? Arch Neurol 67(5):627–630. doi:10.1001/archneurol.2010.51
Zuliani L, Sabater L, Saiz A, Baiges JJ, Giometto B, Graus F (2007) Homer 3 autoimmunity in subacute idiopathic cerebellar ataxia. Neurology 68(3):239–240. doi:10.1212/01.wnl.0000251308.79366.f9
Gruis KL, Moretti P, Gebarski SS, Mikol DD (2003) Cerebellitis in an adult with abnormal magnetic resonance imaging findings prior to the onset of ataxia. Arch Neurol 60(6):877–880. doi:10.1001/archneur.60.6.877
Donmez FY, Agildere AM, Tore HG, Ure S, Benli S (2008) Abnormal diffusion-weighted imaging findings in an adult patient with acute cerebellitis presenting with a normal magnetic resonance imaging. J Comput Assist Tomogr 32(1):156–158. doi:10.1097/RCT.0b013e3180653c8a
Sener RN (2002) Diffusion MRI in Rasmussen's encephalitis, herpes simplex encephalitis, and bacterial meningoencephalitis. Comput Med Imaging Graph 26(5):327–332
Guimaraes RP, D'Abreu A, Yasuda CL, Franca MC Jr, Silva BH, Cappabianco FA, Bergo FP, Lopes-Cendes IT, Cendes F (2013) A multimodal evaluation of microstructural white matter damage in spinocerebellar ataxia type 3. Mov Disord 28(8):1125–1132. doi:10.1002/mds.25451
Alcauter S, Barrios FA, Diaz R, Fernandez-Ruiz J (2011) Gray and white matter alterations in spinocerebellar ataxia type 7: an in vivo DTI and VBM study. NeuroImage 55(1):1–7. doi:10.1016/j.neuroimage.2010.12.014
Kitamura K, Nakayama K, Kosaka S, Yamada E, Shimada H, Miki T, Inoue Y (2008) Diffusion tensor imaging of the cortico-ponto-cerebellar pathway in patients with adult-onset ataxic neurodegenerative disease. Neuroradiology 50(4):285–292. doi:10.1007/s00234-007-0351-9
Ethical standards and patient consent
We declare that all human and animal studies have been approved by the appropriate Ethics Committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. We declare that this manuscript does not contain clinical studies or patient data.
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 143 kb)
Rights and permissions
About this article
Cite this article
Schneider, T., Thomalla, G., Goebell, E. et al. Magnetic resonance imaging findings in patients presenting with (sub)acute cerebellar ataxia. Neuroradiology 57, 551–559 (2015). https://doi.org/10.1007/s00234-015-1496-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-015-1496-6