Abstract
Introduction
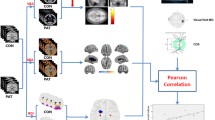
Glaucoma is the second leading cause of blindness worldwide. The purpose of this study is to identify areas of neurodegeneration in glaucoma utilizing 3 T magnetic resonance (MR) diffusion tensor imaging (DTI) parameters with whole-brain voxel-based analysis (VBA) and determine whether these parameters correlate with disease severity.
Methods
Twenty-five glaucoma patients and 25 age-matched healthy volunteers were prospectively examined. Clinical glaucoma severity was assessed utilizing static threshold visual field parameters. All subjects underwent 3 T MRI utilizing a DTI sequence (repetition time/echo time 13,000/68.9 ms, maximal b value 800 s/mm2 along 30 directions) and an anatomic sequence to provide structural information. All data sets were processed by VBA. Brain fractional anisotropy, mean diffusivity (MD), radial diffusivity (RD), and axial diffusivity (AD) were compared in the two groups. Correlation between DTI parameters and glaucoma stage were determined.
Results
The bilateral optic radiations and chiasma of glaucoma patients demonstrated statistically significantly lower fractional anisotropy (p < 0.05). Optic radiation RD was similarly decreased in glaucoma patients (p < 0.05). There were no statistically significant differences noted in MD or AD between the two groups (p > 0.05). Optic chiasm fractional anisotropy values were negatively correlated with glaucoma stage (r = −0.53, p < 0.05) and optic radiation RD values positively correlated (left r = 0.45, p < 0.05; right = 0.38, p = 0.06).
Conclusion
DTI parameters fractional anisotropy and RD are altered in the optic chiasm and radiations of glaucoma patients. As fractional anisotropy and RD also correlate with glaucoma stage, these values could serve as potential noninvasive markers of disease severity.





Similar content being viewed by others
References
Weinreb RN, Khaw PT (2004) Primary open-angle glaucoma. Lancet 363:1711–1720
Boland MV, Quigley HA (2007) Risk factors and open-angle glaucoma: classification and application. J Glaucoma 16:406–418
Gupta N, Yucel YH (2007) Glaucoma as a neurodegenerative disease. Curr Opin Ophthalmol 18:110–114
Dai H, Mu KT, Qi JP, Wang CY, Zhu WZ, Xia LM, Chen ZQ, Zhang H, Ai F, Morelli JN (2011) Assessment of lateral geniculate nucleus atrophy with 3 T MR imaging and correlation with clinical stage of glaucoma. AJNR Am J Neuroradiol 32:1347–1353
Kitsos G, Zikou AK, Bagli E, Kosta P, Argyropoulou MI (2009) Conventional MRI and magnetisation transfer imaging of the brain and optic pathway in primary open-angle glaucoma. Br J Radiol 82:896–900
Gupta N, Ang LC, de Tilly LN, Bidaisee L, Yucel YH (2006) Human glaucoma and neural degeneration in intracranial optic nerve, lateral geniculate nucleus, and visual cortex. Br J Ophthalmol 90:674–678
Gupta N, Yucel YH (2007) What changes can we expect in the brain of glaucoma patients? Surv Ophthalmol 52(Suppl 2):S122–126
Ito Y, Shimazawa M, Chen YN, Tsuruma K, Yamashima T, Araie M, Hara H (2009) Morphological changes in the visual pathway induced by experimental glaucoma in Japanese monkeys. Exp Eye Res 89:246–255
Yucel YH, Zhang Q, Weinreb RN, Kaufman PL, Gupta N (2003) Effects of retinal ganglion cell loss on magno-, parvo-, koniocellular pathways in the lateral geniculate nucleus and visual cortex in glaucoma. Prog Retin Eye Res 22:465–481
Qing G, Zhang S, Wang B, Wang N (2010) Functional MRI signal changes in primary visual cortex corresponding to the central normal visual field of patients with primary open-angle glaucoma. Invest Ophthalmol Vis Sci 51:4627–4634
Dai H, Morelli JN, Ai F, Yin D, Hu C, Xu D, Li Y (2012) Resting-state functional MRI: functional connectivity analysis of the visual cortex in primary open-angle glaucoma patients Hum Brain Mapp.
Beaulieu C (2002) The basis of anisotropic water diffusion in the nervous system—a technical review. NMR Biomed 15:435–455
Augustinack JC, Helmer K, Huber KE, Kakunoori S, Zollei L, Fischl B. Direct visualization of the perforant pathway in the human brain with ex vivo diffusion tensor imaging (Front Hum Neurosci 4: 42.
Tao XF, Wang ZQ, Gong WQ, Jiang QJ, Shi ZR (2009) A new study on diffusion tensor imaging of the whole visual pathway fiber bundle and clinical application. Chin Med J (Engl) 122:178–182
Westlye LT, Walhovd KB, Dale AM, Bjornerud A, Due-Tonnessen P, Engvig A, Grydeland H, Tamnes CK, Ostby Y, Fjell AM. Life-span changes of the human brain white matter: diffusion tensor imaging (DTI) and volumetry (Cereb Cortex 20: 2055–2068.
Song SK, Sun SW, Ramsbottom MJ, Chang C, Russell J, Cross AH (2002) Dysmyelination revealed through MRI as increased radial (but unchanged axial) diffusion of water. NeuroImage 17:1429–1436
Song SK, Sun SW, Ju WK, Lin SJ, Cross AH, Neufeld AH (2003) Diffusion tensor imaging detects and differentiates axon and myelin degeneration in mouse optic nerve after retinal ischemia. NeuroImage 20:1714–1722
Huang J, Friedland RP, Auchus AP (2007) Diffusion tensor imaging of normal-appearing white matter in mild cognitive impairment and early Alzheimer disease: preliminary evidence of axonal degeneration in the temporal lobe. AJNR Am J Neuroradiol 28:1943–1948
Bozzali M, Falini A, Franceschi M, Cercignani M, Zuffi M, Scotti G, Comi G, Filippi M (2002) White matter damage in Alzheimer's disease assessed in vivo using diffusion tensor magnetic resonance imaging. J Neurol Neurosurg Psychiatr 72:742–746
Naismith RT, Xu J, Tutlam NT, Trinkaus K, Cross AH, Song SK (2010) Radial diffusivity in remote optic neuritis discriminates visual outcomes. Neurology 74:1702–1710
Engelhorn T, Michelson G, Waerntges S, Struffert T, Haider S, Doerfler A. Diffusion tensor imaging detects rarefaction of optic radiation in glaucoma patients (Acad Radiol 18: 764–769.
Garaci FG, Bolacchi F, Cerulli A, Melis M, Spano A, Cedrone C, Floris R, Simonetti G, Nucci C (2009) Optic nerve and optic radiation neurodegeneration in patients with glaucoma: in vivo analysis with 3-T diffusion-tensor MR imaging. Radiology 252:496–501
El-Rafei A, Engelhorn T, Warntges S, Dorfler A, Hornegger J, Michelson G (2011) A framework for voxel-based morphometric analysis of the optic radiation using diffusion tensor imaging in glaucoma. Magn Reson Imaging 29:1076–1087
Michelson G, Engelhorn T, Warntges S, El Rafei A, Hornegger J, Doerfler A. (2012) DTI parameters of axonal integrity and demyelination of the optic radiation correlate with glaucoma indices Graefes Arch Clin Exp Ophthalmol.
Zikou AK, Kitsos G, Tzarouchi LC, Astrakas L, Alexiou GA, Argyropoulou MI. Voxel-based morphometry and diffusion tensor imaging of the optic pathway in primary open-angle glaucoma: a preliminary study (AJNR Am J Neuroradiol.
Melonakos ED, Shenton ME, Rathi Y, Terry DP, Bouix S, Kubicki M. Voxel-based morphometry (VBM) studies in schizophrenia-can white matter changes be reliably detected with VBM? (Psychiatry Res 193: 65–70.
Smith SA, Williams ZR, Ratchford JN, Newsome SD, Farrell SK, Farrell JA, Gifford A, Miller NR, van Zijl PC, Calabresi PA, Reich DS (2011) Diffusion tensor imaging of the optic nerve in multiple sclerosis: association with retinal damage and visual disability. AJNR Am J Neuroradiol 32:1662–1668
Wheeler-Kingshott CA, Cercignani M (2009) About “axial” and “radial” diffusivities. Magn Reson Med 61:1255–1260
Jones DK, Symms MR, Cercignani M, Howard RJ (2005) The effect of filter size on VBM analyses of DT-MRI data. NeuroImage 26:546–554
Smith SM, Jenkinson M, Johansen-Berg H, Rueckert D, Nichols TE, Mackay CE, Watkins KE, Ciccarelli O, Cader MZ, Matthews PM, Behrens TE (2006) Tract-based spatial statistics: voxelwise analysis of multi-subject diffusion data. NeuroImage 31:1487–1505
Bodini B, Khaleeli Z, Cercignani M, Miller DH, Thompson AJ, Ciccarelli O (2009) Exploring the relationship between white matter and gray matter damage in early primary progressive multiple sclerosis: an in vivo study with TBSS and VBM. Hum Brain Mapp 30:2852–2861
Della Nave R, Ginestroni A, Tessa C, Salvatore E, Bartolomei I, Salvi F, Dotti MT, De Michele G, Piacentini S, Mascalchi M (2008) Brain white matter tracts degeneration in Friedreich ataxia. An in vivo MRI study using tract-based spatial statistics and voxel-based morphometry. NeuroImage 40:19–25
Haller S, Xekardaki A, Delaloye C, Canuto A, Lovblad KO, Gold G, Giannakopoulos P (2011) Combined analysis of grey matter voxel-based morphometry and white matter tract-based spatial statistics in late-life bipolar disorder. J Psychiatry Neurosci 36:391–401
Newlander S (2011) Methodological improvements in voxel based analysis of diffusion tensor images: aplications to study the impact of APOE on white matter integrity. San Diego State University, Dissertation
Dai H, Deng X, Li Y, Morelli J, Ai F, Guo L, Wang C. (2011) Dorsal visual pathway neurodegeneration in patients with primary open angle glaucoma: in vivo analysis with 3 T diffusion-tensor MR imaging 97th Scientific Assembly and Annual meeting of the Radiological Society of North America. Chicago, IL
Acknowledgments
This work was supported by the Natural Science Fund of China (81201079 /81171393), a grant from the Shanghai Commission of Science and Technology #10440710200, and a grant from NIH/NIMH P50 MH090966-01.
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dai, H., Yin, D., Hu, C. et al. Whole-brain voxel-based analysis of diffusion tensor MRI parameters in patients with primary open angle glaucoma and correlation with clinical glaucoma stage. Neuroradiology 55, 233–243 (2013). https://doi.org/10.1007/s00234-012-1122-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-012-1122-9