Abstract
Introduction
Sensory neuronopathy (SNN) is a distinctive subtype of peripheral neuropathies, specifically targeting dorsal root ganglion (DRG). We utilized MRI to demonstrate the imaging characteristics of DRG, spinal cord (SC), and brachial plexus at C7 level in SNN.
Methods
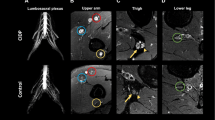
We attempted multiple-echo data image combination (MEDIC) and turbo inversion recovery magnitude (TIRM) methods in nine patients with sensory neuronopathy and compared with those in 16 disease controls and 20 healthy volunteers. All participants underwent MRI for the measurement of DRG, posterior column (PC), lateral column, and spinal cord area (SCA) at C7 level. DRG diameters were obtained through its largest cross section, standardized by dividing sagittal diameter of mid-C7 vertebral canal. We also made comparisons of standardized anteroposterior diameter (APD) and left–right diameters of SC and PC in these groups. Signal intensity and diameter of C7 spinal nerve were assessed on TIRM.
Results
Compared to control groups, signal intensities of DRG and PC were higher in SNN patients when using MEDIC, but the standardized diameters were shorter in either DRG or PC. Abnormal PC signal intensities were identified in eight out of nine SNN patients (89 %) with MEDIC and five out of nine (56 %) with T2-weighted images. SCA, assessed with MEDIC, was smaller in SNN patients than in the other groups, with significant reduction of its standardized APD. C7 nerve root diameters, assessed with TIRM, were decreased in SNN patients.
Conclusion
MEDIC and TIRM sequences demonstrate increased signal intensities and decreased area of DRG and PC, and decreased diameter of nerve roots in patients with SNN, which can play a significant role in early diagnosis.




Similar content being viewed by others
References
Martinez AR, Nunes MB et al (2012) Sensory neuronopathy and autoimmune diseases. Autoimmune Dis 2012:873587
Damasceno A, Franca MC Jr et al (2008) Chronic acquired sensory neuron diseases. Eur J Neurol 15(12):1400–1405
Lauria G, Pareyson D et al (2003) Neurophysiological diagnosis of acquired sensory ganglionopathies. Eur Neurol 50(3):146–152
Denny-Brown D (1948) Primary sensory neuropathy with muscular changes associated with carcinoma. J Neurol Neurosurg Psychiatry 11(2):73–87
Kuntzer T, Antoine J-C et al (2004) Clinical features and pathophysiological basis of sensory neuronopathies (ganglionopathies). Muscle Nerve 30(3):255–268
Sghirlanzoni A, Pareyson D et al (2005) Sensory neuron diseases. Lancet Neurol 4(6):349–361
Krarup-Hansen A, Helweg-Larsen S et al (2007) Neuronal involvement in cisplatin neuropathy: prospective clinical and neurophysiological studies. Brain 130(Pt 4):1076–1088
Sheikh SI, Amato AA (2010) The dorsal root ganglion under attack: the acquired sensory ganglionopathies. Pract Neurol 10(6):326–334
Pavlakis PP, Alexopoulos H et al (2012) Peripheral neuropathies in Sjogren's syndrome: a critical update on clinical features and pathogenetic mechanisms. J Autoimmun 39(1–2):27–33
Mori K, Iijima M et al (2005) The wide spectrum of clinical manifestations in Sjogren's syndrome-associated neuropathy. Brain 128:2518–2534
Franca MC Jr, D'Abreu A et al (2008) MRI shows dorsal lesions and spinal cord atrophy in chronic sensory neuronopathies. J Neuroimaging 18(2):168–172
Walk D (2009) Role of skin biopsy in the diagnosis of peripheral neuropathic pain. Curr Pain Headache Rep 13(3):191–196
Lauria G, Sghirlanzoni A et al (2001) Epidermal nerve fiber density in sensory ganglionopathies: clinical and neurophysiologic correlations. Muscle Nerve 24(8):1034–1039
Lauria G, Lombardi R (2012) Small fiber neuropathy: is skin biopsy the holy grail? Curr Diab Rep 12(4):384–392
Colli BO, Carlotti CG et al (2008) Dorsal root ganglionectomy for the diagnosis of sensory neuropathies. Surgical technique and results. Surg Neurol 69(3):266–273
Asbury AK (1987) Sensory neuronopathy. Semin Neurol 7(1):58–66
Asbury AK, Brown MJ (1990) Sensory neuronopathy and pure sensory neuropathy. Curr Opin Neurol Neurosurg 3:708–711
Camdessanche JP, Jousserand G et al (2009) The pattern and diagnostic criteria of sensory neuronopathy: a case-control study. Brain 132(Pt 7):1723–1733
Lauria G, Pareyson D et al (2000) Clinical and magnetic resonance imaging findings in chronic sensory ganglionopathies. Ann Neurol 47(1):104–109
Sobue G, Yasuda T et al (1995) MRI demonstrates dorsal column involvement of the spinal cord in Sjogren's syndrome-associated neuropathy. Neurology 45(3 Pt 1):592–593
Grant GA, Goodkin R et al (2004) MR neurography: diagnostic utility in the surgical treatment of peripheral nerve disorders. Neuroimaging Clin N Am 14(1):115–133
Schmid MR, Pfirrmann CW et al (2005) Imaging of patellar cartilage with a 2D multiple-echo data image combination sequence. AJR Am J Roentgenol 184(6):1744–1748
Vertinsky AT, Krasnokutsky MV et al (2007) Cutting-edge imaging of the spine. Neuroimaging Clin N Am 17(1):117–136
Viallon M, Vargas MI et al (2008) High-resolution and functional magnetic resonance imaging of the brachial plexus using an isotropic 3D T2 STIR (Short Term Inversion Recovery) SPACE sequence and diffusion tensor imaging. Eur Radiol 18(5):1018–1023
Mori K, Koike H et al (2001) Spinal cord magnetic resonance imaging demonstrates sensory neuronal involvement and clinical severity in neuronopathy associated with Sjogren's syndrome. J Neurol Neurosurg Psychiatry 71(4):488–492
Ko HY, Park JH et al (2004) Gross quantitative measurements of spinal cord segments in human. Spinal Cord 42(1):35–40
West CA, McKay Hart A et al (2011) Sensory neurons of the human brachial plexus: a quantitative study employing optical fractionation and in-vivo volumetric magnetic resonance imaging. Neurosurgery 70(5):1183–1194, discussion 1194
Okumura R, Asato R et al (1992) Degeneration of the posterior columns of the spinal cord: postmortem MRI and histopathology. J Comput Assist Tomogr 16(6):865–867
Lin CC, Chiu MJ (2008) Teaching neuroimage: cervical cord atrophy with dorsal root ganglionopathy in Sjogren syndrome. Neurology 70(7):e27
Koike H, Atsuta N et al (2010) Clinicopathological features of acute autonomic and sensory neuropathy. Brain 133:2881–2896
Tavee J, Zhou L (2009) Small fiber neuropathy: a burning problem. Cleve Clin J Med 76(5):297–305
Koike H, Sobue G (2008) Small neurons may be preferentially affected in ganglionopathy. J Neurol Neurosurg Psychiatry 79(2):113
Kastrup O, Timman D et al (2010) Isolated degeneration of the posterior column as a distinct entity—a clinical and electrophysiologic follow-up study. Clin Neurol Neurosurg 112(3):209–212
Ishikawa M, Matsumoto M et al (2003) Changes of cervical spinal cord and cervical spinal canal with age in asymptomatic subjects. Spinal Cord 41(3):159–163
Zivadinov R, Banas AC et al (2008) Comparison of three different methods for measurement of cervical cord atrophy in multiple sclerosis. AJNR Am J Neuroradiol 29(2):319–325
Lundell H, Barthelemy D et al (2011) Independent spinal cord atrophy measures correlate to motor and sensory deficits in individuals with spinal cord injury. Spinal Cord 49(1):70–75
Acknowledgments
The authors thank Hui Zhang PhD., from the Department of Computer Science and Centre for Medical Image Computing, University College London, UK, for critical comments and helpful suggestions. We also would like to thank Huan Xu for her kind help in revising the manuscript.
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Bao, YF., Tang, WJ., Zhu, DQ. et al. Sensory neuronopathy involves the spinal cord and brachial plexus: a quantitative study employing multiple-echo data image combination (MEDIC) and turbo inversion recovery magnitude (TIRM). Neuroradiology 55, 41–48 (2013). https://doi.org/10.1007/s00234-012-1085-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-012-1085-x