Abstract
Introduction
We investigated the correlation between age and the fractional anisotropy (FA) values of peripheral nerves in healthy adults and compared the age-corrected FA values of peripheral nerves in healthy subjects and patients with polyneuropathy.
Methods
The institutional review board approved this study and informed consent was obtained from all participants before entry into the study. We optimized diffusion tensor imaging using a 3-T magnetic resonance scanner and an extremity coil for scanning tibial nerves. The effect of age and sex on the FA values of tibial nerves in healthy volunteers was investigated and the age-corrected FA values of tibial nerves in healthy volunteers and patients with polyneuropathy were compared.
Results
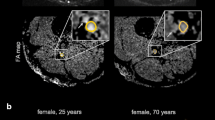
The maximum FA values of the tibial nerves remained constant until age 45 (approximately 0.516); they subsequently decreased by 0.004/year in healthy volunteers. After removing the effect of age with an age-adjusted equation, the median maximum FA values in the volunteers and patients were 0.518 (range, 0.406–0.616) and 0.442 (range, 0.376–0.530), respectively. The age-corrected FA values were significantly lower in the patients than the healthy volunteers (p < 0.001). There was no significant gender-related difference in the maximum FA values of the tibial nerves (p = 0.416).
Conclusion
The age-corrected FA value of the peripheral nerves helps to differentiate between age-related peripheral nerve degeneration and polyneuropathies.





Similar content being viewed by others
References
Stein D, Neufeld A, Pasternak O, Graif M, Patish H, Schwimmer E, Ziv E, Assaf Y (2009) Diffusion tensor imaging of the median nerve in healthy and carpal tunnel syndrome subjects. J Magn Reson Imaging 29:657–662
Khalil C, Hancart C, Le Thuc V, Chantelot C, Chechin D, Cotten A (2008) Diffusion tensor imaging and tractography of the median nerve in carpal tunnel syndrome: preliminary results. Eur Radiol 18:2283–2291
Yao L, Gai N (2009) Median nerve cross-sectional area and MRI diffusion characteristics: normative values at the carpal tunnel. Skeletal Radiol 38:355–361
Sheikh KA (2010) Non-invasive imaging of nerve regeneration. Exp Neurol 223:72–76
Kakuda T, Fukuda H, Tanitame K, Takasu M, Date S, Ochi K, Ohshita T, Kohriyama T, Ito K, Matsumoto M, Awai K (2011) Diffusion tensor imaging of peripheral nerve in patients with chronic inflammatory demyelinating polyradiculoneuropathy: a feasibility study. Neuroradiology (in press)
Vargas MI, Viallon M, Nguyen D, Delavelle J, Becker M (2010) Diffusion tensor imaging (DTI) and tractography of the brachial plexus: feasibility and initial experience in neoplastic conditions. Neuroradiology 52:237–245
Basser PJ, Pierpaoli C (1996) Microstructural and physiological features of tissues elucidated by quantitative-diffusion-tensor MRI. J Magn Reson B 111:209–219
Beaulieu C (2002) The basis of anisotropic water diffusion in the nervous system—a technical review. NMR Biomed 15:435–455
Wimberger DM, Roberts TP, Barkovich AJ, Prayer LM, Moseley ME, Kucharczyk J (1995) Identification of premyelination by diffusion-weighted MRI. J Comput Assist Tomogr 19:28–33
Takagi T, Nakamura M, Yamada M, Hikishima K, Momoshima S, Fujiyoshi K, Shibata S, Okano HJ, Toyama Y, Okano H (2009) Visualization of peripheral nerve degeneration and regeneration: monitoring with diffusion tensor tractography. Neuroimage 44:884–892
Morisaki S, Kawai Y, Umeda M, Nishi M, Oda R, Fujiwara H, Yamada K, Higuchi T, Tanaka C, Kawata M, Kubo T (2011) In vivo assessment of peripheral nerve regeneration by diffusion tensor imaging. J Magn Reson Imaging 33:535–542
Pfefferbaum A, Sullivan EV (2003) Increased brain white matter diffusivity in normal adult aging: relationship to anisotropy and partial voluming. Magn Reson Med 49:953–961
Pfefferbaum A, Sullivan EV, Hedehus M, Lim KO, Adalsteinsson E, Moseley M (2000) Age-related decline in brain white matter anisotropy measured with spatially corrected echo-planar diffusion tensor imaging. Magn Reson Med 44:259–268
Salat DH, Smith EE, Tuch DS, Benner T, Pappu V, Schwab KM, Gurol ME, Rosas D, Rosand J, Greenberg SM (2006) White matter alterations in cerebral amyloid angiopathy measured by diffusion tensor imaging. Stroke 37:1759–1764
Van Hecke W, Leemans A, Sijbers J, Vandervliet E, Van Goethem J, Parizel PM (2008) A tracking-based diffusion tensor imaging segmentation method for the detection of diffusion-related changes of the cervical spinal cord with aging. J Magn Reson Imaging 27:978–991
Maier SE, Mamata H (2005) Diffusion tensor imaging of the spinal cord. Ann NY Acad Sci 1064:50–60
Tong HC, Werner RA, Franzblau A (2004) Effect of aging on sensory nerve conduction study parameters. Muscle Nerve 29:716–720
Taylor PK (1984) Non-linear effects of age on nerve-conduction in adults. J Neurol Sci 66:223–234
Gorson KC, Allam G, Ropper AH (1997) Chronic inflammatory demyelinating polyneuropathy: clinical features and response to treatment in 67 consecutive patients with and without a monoclonal gammopathy. Neurology 48:321–328
Skre H (1974) Genetic and clinical aspects of Charcot–Marie–Tooth’s disease. Clin Genet 6:98–118
Hartung HP, van der Meche FGA, Pollard JD (1998) Guillain–Barre syndrome, CIDP and other chronic immune-mediated neuropathies. Curr Opin Neurol 11:497–513
Hattori N, Ichimura M, Nagamatsu M, Li M, Yamamoto K, Kumazawa K, Mitsuma T, Sobue G (1999) Clinicopathological features of Churg–Strauss syndrome-associated neuropathy. Brain 122:427–439
Akaike H (1973) Information theory and an extension of the maximum likelihood principle. In: Petrov BN, Csaki F (eds) Proceedings of the 2nd International Symposium on Information Theory. Akademiai Kiado, Budapest, pp 267–281
Kabakci N, Gurses B, Firat Z, Bayram A, Ulug AM, Kovanlikaya A, Kovanlikaya I (2007) Diffusion tensor imaging and tractography of median nerve: normative diffusion values. AJR Am J Roentgenol 189:923–927
Kimura J (1984) Principles and pitfalls of nerve-conduction studies. Ann Neurol 16:415–429
Matsuo K, Mizuno T, Yamada K, Akazawa K, Kasai T, Kondo M, Mori S, Nishimura T, Nakagawa M (2008) Cerebral white matter damage in frontotemporal dementia assessed by diffusion tensor tractography. Neuroradiology 50:605–611
Kara B, Celik A, Karadereler S, Ulusoy L, Ganiyusufoglu K, Onat L, Mutlu A, Ornek I, Sirvanci M, Hamzaoglu A (2011) The role of DTI in early detection of cervical spondylotic myelopathy: a preliminary study with 3-T MRI. Neuroradiology 53:609–616
Hiltunen J, Suortti T, Arvela S, Seppa M, Joensuu R, Hari R (2005) Diffusion tensor imaging and tractography of distal peripheral nerves at 3T. Clin Neurophysiol 116:2315–2323
Bammer R, Keeling SL, Augustin M, Pruessmann KP, Wolf R, Stollberger R, Hartung HP, Fazekas F (2001) Improved diffusion-weighted single-shot echo-planar imaging (EPI) in stroke using sensitivity encoding (SENSE). Magn Reson Med 46:548–554
Yang QX, Wang JL, Smith’ MB, Meadowcroft M, Sun XY, Eslinger PJ, Golay X (2004) Reduction of magnetic field inhomogeneity artifacts in echo planar imaging with SENSE and GESEPI at high field. Magn Reson Med 52:1418–1423
Kuhl CK, Traber F, Gieseke J, Drahanowsky W, Morakkabati-Spitz N, Willinek W, Von Falkenhausen M, Manka C, Schild HH (2008) Whole-body high-field-strength (3.0-T) MR imaging in clinical practice. Part II. Technical considerations and clinical applications. Radiology 247:16–35
Rosen Y, Bloch BN, Lenkinski RE, Greenman RL, Marquis RP, Rofsky NM (2007) 3T MR of the prostate: reducing susceptibility gradients by inflating the endorectal coil with a barium sulfate suspension. Magn Reson Med 57:898–904
Rohde GK, Barnett AS, Basser PJ, Marenco S, Pierpaoli C (2004) Comprehensive approach for correction of motion and distortion in diffusion-weighted MRI. Magn Reson Med 51:103–114
Reese TG, Heid O, Weisskoff RM, Wedeen VJ (2003) Reduction of eddy-current-induced distortion in diffusion MRI using a twice-refocused spin echo. Magn Reson Med 49:177–182
Mangin JF, Poupon C, Clark C, Le Bihan D, Bloch I (2002) Distortion correction and robust tensor estimation for MR diffusion imaging. Med Image Anal 6:191–198
Rosenberg NR, Portegies P, de Visser M, Vermeulen M (2001) Diagnostic investigation of patients with chronic polyneuropathy: evaluation of a clinical guideline. J Neurol Neurosurg Psychiatry 71:205–209
Berger AR, Bradley WG, Brannagan TH, Busis NA, Cros DP, Dalakas MC, Danon MJ, Donofrio PD, Engel WK, England JD, Feldman EL, Freeman RL, Kinsella LJ, Lacomis D, Latov N, Menkes DL, Sander HW, Thomas FP, Triggs WJ, Windebank AJ, Wolfe GI, Assoc N, Comm MA (2003) Guidelines for the diagnosis and treatment of chronic inflammatory demyelinating polyneuropathy. J Peripher Nerv Syst 8:282–284
Molenaar DSM, Vermeulen M, de Haan R (1998) Diagnostic value of sural nerve biopsy in chronic inflammatory demyelinating polyneuropathy. J Neurol Neurosurg Psychiatry 64:84–89
Paus T (2010) Growth of white matter in the adolescent brain: myelin or axon? Brain Cogn 72:26–35
Barnea-Goraly N, Menon V, Eckert M, Tamm L, Bammer R, Karchemskiy A, Dant CC, Reiss AL (2005) White matter development during childhood and adolescence: a cross-sectional diffusion tensor imaging study. Cereb Cortex 15:1848–1854
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tanitame, K., Iwakado, Y., Akiyama, Y. et al. Effect of age on the fractional anisotropy (FA) value of peripheral nerves and clinical significance of the age-corrected FA value for evaluating polyneuropathies. Neuroradiology 54, 815–821 (2012). https://doi.org/10.1007/s00234-011-0981-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-011-0981-9