Abstract
Introduction
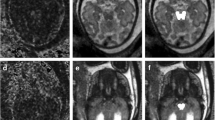
Forebrain malformations include some of the most severe developmental anomalies and require early diagnosis. The proof of normal or abnormal prosencephalic development may have an influence on further management in the event of a suspected fetal malformation. The purpose of this retrospective study was to evaluate the detectability of anatomical landmarks of forebrain development using in vivo fetal magnetic resonance imaging (MRI) before gestational week (gw) 27.
Methods
MRI studies of 83 singleton fetuses (gw 16-26, average ± sd: gw 22 ± 2) performed at 1.5 Tesla were assessed. T2-weighted (w) fast spin echo, T1w gradient-echo and diffusion-weighted sequences were screened for the detectability of anatomical landmarks as listed below.
Results
The interhemispheric fissure, ocular bulbs, corpus callosum, infundibulum, chiasm, septum pellucidum (SP), profile, and palate were detectable in 95%, 95%, 89%, 87%, 82%, 81%, 78%, 78% of cases. Olfactory tracts were more easily delineated than bulbs and sulci (37% versus 18% and 8%), with significantly higher detection rates in the coronal plane. The pituitary gland could be detected on T1w images in 60% with an increasing diameter with gestational age (p = 0.041). The delineation of olfactory tracts (coronal plane), chiasm, SP and pituitary gland were significantly increased after week 21 (p < 0.05). Pathologies were found in 28% of cases.
Conclusion
This study provides detection rates for anatomical landmarks of forebrain development with fetal MRI before gw 27. Several anatomical structures are readily detectable with routine fetal MRI sequences; thus, if these landmarks are not delineable, it should raise the suspicion of a pathology. Recommendations regarding favorable sequences/planes are provided.









Similar content being viewed by others
References
Demyer W, Zeman W, Palmer CG (1964) The face predicts the brain: diagnostic significance of median facial anomalies for holoprosencephaly (arhinencephaly). Pediatrics 34:256–263
Belhocine O, Andre C, Kalifa G, Adamsbaum C (2005) Does asymptomatic septal agenesis exist? A review of 34 cases. Pediatr Radiol 35:410–418
Chadie A, Radi S, Trestard L, Charollais A, Eurin D, Verspyck E, Marret S (2008) Neurodevelopmental outcome in prenatally diagnosed isolated agenesis of the corpus callosum. Acta Paediatr 97:420–424
Moutard ML, Kieffer V, Feingold J, Kieffer F, Lewin F, Adamsbaum C, Gelot A, Campistol IPJ, van Bogaert P, Andre M, Ponsot G (2003) Agenesis of corpus callosum: prenatal diagnosis and prognosis. Childs Nerv Syst 19:471–476
Gupta JK, Lilford RJ (1995) Assessment and management of fetal agenesis of the corpus callosum. Prenat Diagn 15:301–312
Shevell MI (2002) Clinical and diagnostic profile of agenesis of the corpus callosum. J Child Neurol 17:896–900
Francesco P, Maria-Edgarda B, Giovanni P, Dandolo G, Giulio B (2006) Prenatal diagnosis of agenesis of corpus callosum: what is the neurodevelopmental outcome? Pediatr Int 48:298–304
Moore KL (2008) Development of the brain. The developing human: clinically oriented embryology. Saunders Elselvier, Philadelphia, pp 392–404
Prayer D, Brugger PC, Krampl E, Prayer L (2006) Indications for fetal magnetic resonance imaging. Radiologe 46:98–104
Azoulay R, Fallet-Bianco C, Garel C, Grabar S, Kalifa G, Adamsbaum C (2006) MRI of the olfactory bulbs and sulci in human fetuses. Pediatr Radiol 36:97–107
Brugger PC, Stuhr F, Lindner C, Prayer D (2006) Methods of fetal MR: beyond T2-weighted imaging. Eur J Radiol 57:172–181
Scoffings DJ, Kurian KM (2008) Congenital and acquired lesions of the septum pellucidum. Clin Radiol 63:210–219
Isapof A, Kieffer V, Sacco S, Billette de Villemeur T, Gelot A, Garel C, Adamsbaum C, Lewin F, Jouannic JM, Raffo E, Moutard ML. Impact of prenatal corpus callosum agenesis diagnosis on pregnancy outcome. Evaluation of 155 cases between 2000 and 2006. Arch Pediatr 17:226-232
Rapp B, Perrotin F, Marret H, Sembely-Taveau C, Lansac J, Body G (2002) Value of fetal cerebral magnetic resonance imaging for the prenatal diagnosis and prognosis of corpus callosum agenesis. J Gynécol Obstét Biol Reprod 31:173–182
Volpe JJ (2000) Overview: normal and abnormal human brain development. Ment Retard Dev Disabil Res Rev 6:1–5
Volpe P, Campobasso G, De Robertis V, Rembouskos G (2009) Disorders of prosencephalic development. Prenat Diagn 29:340–354
Ren T, Anderson A, Shen WB, Huang H, Plachez C, Zhang J, Mori S, Kinsman SL, Richards LJ (2006) Imaging, anatomical, and molecular analysis of callosal formation in the developing human fetal brain. Anat Rec A Discov Mol Cell Evol Biol 288:191–204
Huang H, Xue R, Zhang J, Ren T, Richards LJ, Yarowsky P, Miller MI, Mori S (2009) Anatomical characterization of human fetal brain development with diffusion tensor magnetic resonance imaging. J Neurosci 29:4263–4273
Kasprian G, Brugger PC, Weber M, Krssak M, Krampl E, Herold C, Prayer D (2008) In utero tractography of fetal white matter development. Neuroimage 43:213–224
Glenn OA, Goldstein RB, Li KC, Young SJ, Norton ME, Busse RF, Goldberg JD, Barkovich AJ (2005) Fetal magnetic resonance imaging in the evaluation of fetuses referred for sonographically suspected abnormalities of the corpus callosum. J Ultrasound Med 24:791–804
Lin AE, Siebert JR, Graham JM Jr (1990) Central nervous system malformations in the CHARGE association. Am J Med Genet 37:304–310
Li C, Yousem DM, Doty RL, Kennedy DW (1994) Neuroimaging in patients with olfactory dysfunction. AJR 162:411–418
Cervantes LF, Altman NR, Medina LS (2006) Case 102: pituitary aplasia. Radiology 241:936–938
Bonneville F, Cattin F, Marsot-Dupuch K, Dormont D, Bonneville JF, Chiras J (2006) T1 signal hyperintensity in the sellar region: spectrum of findings. Radiographics 26:93–113
Colombo N, Berry I, Kucharczyk J, Kucharczyk W, de Groot J, Larson T, Norman D, Newton TH (1987) Posterior pituitary gland: appearance on MR images in normal and pathologic states. Radiology 165:481–485
Lee MH, Choi HY, Sung YA, Lee JK (2001) High signal intensity of the posterior pituitary gland on T1-weighted MR images. Correlation with plasma vasopressin concentration to water deprivation. Acta Radiol 42:129–134
Kurokawa H, Fujisawa I, Nakano Y, Kimura H, Akagi K, Ikeda K, Uokawa K, Tanaka Y (1998) Posterior lobe of the pituitary gland: correlation between signal intensity on T1-weighted MR images and vasopressin concentration. Radiology 207:79–83
Cox TD, Elster AD (1991) Normal pituitary gland: changes in shape, size, and signal intensity during the 1st year of life at MR imaging. Radiology 179:721–724
Dietrich RB, Lis LE, Greensite FS, Pitt D (1995) Normal MR appearance of the pituitary gland in the first 2 years of life. AJNR 16:1413–1419
Argyropoulou MI, Xydis V, Kiortsis DN, Pantou K, Zikou A, Efremidis SC, Andronikou S (2004) Pituitary gland signal in pre-term infants during the first year of life: an MRI study. Neuroradiology 46:1031–1035
Acknowledgments
The authors would like to thank the radiographer team of this institution for their commitment and skillfull work, Sheida Mehrain, M.D., for proofreading and Mary Mc. Alister for her help in editing the manuscript.
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schmook, M.T., Brugger, P.C., Weber, M. et al. Forebrain development in fetal MRI: evaluation of anatomical landmarks before gestational week 27. Neuroradiology 52, 495–504 (2010). https://doi.org/10.1007/s00234-010-0701-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-010-0701-x