Abstract
Introduction
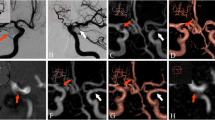
Nexus coils are a type of bioactive coil used to embolize intracranial aneurysms. The purpose of this study was to test the feasibility of the noninvasive follow-up of aneurysms treated with Nexus coils by means of magnetic resonance angiography (MRA).
Methods
Three-dimensional (3D) time-of-flight (TOF) MRA images of patients treated with Nexus coils (the Nexus coil group) or bare platinum coils (the control group) were compared for the severity and frequency of artifacts. The reviewers were unaware of the coil types used. In the Nexus coil group, 17 MRA examinations were performed in 14 patients harboring 15 aneurysms treated with Nexus coils using 3-T (n = 11) and 1.5-T (n = 6) MR units. The findings of these examinations were compared to those of 28 MRA studies conducted on 24 control patients (bare platinum coils). Conventional angiograms, maximum intensity projections, and source data of 3D-TOF MRA were reviewed in terms of residual flow within aneurysms and parent arterial patencies. The qualities of the MRA images were rated from grade 0 (no significant signal loss) to grade 2 (complete segmental signal loss of the parent artery). The normalized ratio, defined as the diameter of signal loss on MRA axial source images (in mm) divided by that of coil mass on plain radiographs (in mm), was calculated to compare the sizes of coil-related artifacts in the two groups.
Results
The quality of the MRA image for the Nexus coil group was significantly poorer than that for the control group (p < 0.0001) due to signal loss caused by the presence of artifacts in the former. In particular, the interpretation of aneurysmal status was impossible in all cases of coiled aneurysms due to segmental signal loss. The sizes of the MRA artifacts were also significantly larger in the Nexus coil group (normalized ratio 1.61 ± 0.22 vs. 1.15 ± 0.20; p < 0.0001).
Conclusion
Follow-up evaluations by 3D-TOF MRA of aneurysms treated with Nexus coils are severely limited.



Similar content being viewed by others
References
Bendszus M, Solymosi L (2006) Cerecyte coils in the treatment of intracranial aneurysms: a preliminary clinical study. AJNR Am J Neuroradiol 27:2053–2057
Connor SE, West RJ, Yates DA (2001) The ability of plain radiography to predict intracranial aneurysm occlusion instability during follow-up of endosaccular treatment with Guglielmi detachable coils. Neuroradiology 43:680–686
Costalat V, Lebars E, Sarry L, Defasque A, Barbotte E, Brunel H, Bourbotte G, Bonafe A (2006) In vitro evaluation of 2D-digital subtraction angiography versus 3D-time-of-flight in assessment of intracranial cerebral aneurysm filling after endovascular therapy. AJNR Am J Neuroradiol 27:177–184
Cottier JP, Bleuzen-Couthon A, Gallas S, Vinikoff-Sonier CB, Bertrand P, Domengie F, Barantin L, Herbreteau D (2003) Intracranial aneurysms treated with Guglielmi detachable coils: is contrast material necessary in the follow-up with 3D time-of-flight MR angiography? AJNR Am J Neuroradiol 24:1797–1803
Cottier JP, Bleuzen-Couthon A, Gallas S, Vinikoff-Sonier CB, Bertrand P, Domengie F, Barantin L, Herbreteau D (2003) Follow-up of intracranial aneurysms treated with detachable coils: comparison of plain radiographs, 3D time-of-flight MRA and digital subtraction angiography. Neuroradiology 45:818–824
Derdeyn CP, Graves VB, Turski PA, Masaryk AM, Strother CM (1997) MR angiography of saccular aneurysms after treatment with Guglielmi detachable coils: preliminary experience. AJNR Am J Neuroradiol 18:279–286
Gonner F, Heid O, Remonda L, Nicoli G, Baumgartner RW, Godoy N, Schroth G (1998) MR angiography with ultrashort echo time in cerebral aneurysms treated with Guglielmi detachable coils. AJNR Am J Neuroradiol 19:1324–1328
Guglielmi G, Vinuela F, Dion J, Duckwiler G (1991) Electrothrombosis of saccular aneurysms via endovascular approach. Part 2: Preliminary clinical experience. J Neurosurg 75:8–14
Guglielmi G, Vinuela F, Sepetka I, Macellari V (1991) Electrothrombosis of saccular aneurysms via endovascular approach. Part 1: Electrochemical basis, technique, and experimental results. J Neurosurg 75:1–7
Hartman J, Nguyen T, Larsen D, Teitelbaum GP (1997) MR artifacts, heat production, and ferromagnetism of Guglielmi detachable coils. AJNR Am J Neuroradiol 18:497–501
Hennemeyer CT, Wicklow K, Feinberg DA, Derdeyn CP (2001) In vitro evaluation of platinum Guglielmi detachable coils at 3 T with a porcine model: safety issues and artifacts. Radiology 219:732–737
Jennett B, Bond M (1975) Assessment of outcome after severe brain damage: a practical scale. Lancet 1:480–484
Jupp B, Williams JP, Tesiram YA, Vosmansky M, O’Brien TJ (2006) MRI compatible electrodes for the induction of amygdala kindling in rats. J Neurosci Methods 155:72–76
Kang HS, Han MH, Kwon BJ, Kwon OK, Kim SH, Choi SH, Chang KH (2005) Short-term outcome of intracranial aneurysms treated with polyglycolic acid/lactide copolymer-coated coils compared to historical controls treated with bare platinum coils: a single-center experience. AJNR Am J Neuroradiol 26:1921–1928
Kang HS, Han MH, Kwon BJ, Kwon OK, Kim SH (2006) Repeat endovascular treatment in post-embolization recurrent intracranial aneurysms. Neurosurgery 58:60–70
Kang HS, Han MH, Lee TH, Shin YS, Roh HG, Kwon OK, Kwon BJ, Kim SY, Kim SH, Byun HS (2007) Embolization of intracranial aneurysms with hydrogel-coated coils: result of Korean Multicenter Trial. Neurosurgery 61:51–59
Kwon BJ, Han MH, Oh CW, Kim KH, Chang KH (2002) Anatomical and clinical outcomes after endovascular treatment for unruptured cerebral aneurysms: a single center experience. Intervent Neuroradiol 8:367–376
Kwon BJ, Han MH, Oh CW, Kim KH, Chang KH (2003) Procedure-related haemorrhage in embolisation of intracranial aneurysms with Guglielmi detachable coils. Neuroradiology 45:562–569
Lovblad KO, Yilmaz H, Chouiter A, San Millan Ruiz D, Abdo G, Bijlenga P, de Tribolet N, Ruefenacht DA (2006) Intracranial aneurysm stenting: follow-up with MR angiography. J Magn Reson Imaging 24:418–422
Marshall MW, Teitelbaum GP, Kim HS, Deveikis J (1991) Ferromagnetism and magnetic resonance artifacts of platinum embolization microcoils. Cardiovasc Intervent Radiol 14:163–166
Martinez-Santiesteban FM, Swanson SD, Noll DC, Anderson DJ (2007) Magnetic field perturbation of neural recording and stimulating microelectrodes. Phys Med Biol 52:2073–2088
Schmalbrock P, Yuan C, Chakeres DW, Kohli J, Pelc NJ (1990) Volume MR angiography: methods to achieve very short echo times. Radiology 175:861–865
Shellock FG, Detrick MS, Brant-Zawadski MN (1997) MR compatibility of Guglielmi detachable coils. Radiology 203:568–570
Walker MT, Tsai J, Parish T, Tzung B, Shaibani A, Krupinski E, Russell EJ (2005) MR angiographic evaluation of platinum coil packs at 1.5 T and 3 T: an in vitro assessment of artifact production: technical note. AJNR Am J Neuroradiol 26:848–853
Wall A, Kugel H, Bachman R, Matuszewski L, Kramer S, Heindel W, Maintz D (2005) 3.0 T vs. 1.5 T MR angiography: in vitro comparison of intravascular stent artifacts. J Magn Reson Imaging 22:772–779
Yamada N, Hayashi K, Murao K, Higashi M, Iihara K (2004) Time-of-flight MR angiography targeted to coiled intracranial aneurysms is more sensitive to residual flow than is digital subtraction angiography. AJNR Am J Neuroradiol 25:1154–1157
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, HS., Moon, WJ., Roh, H.G. et al. MR angiographic evaluation is limited in intracranial aneurysms embolized with Nexus coils. Neuroradiology 50, 171–178 (2008). https://doi.org/10.1007/s00234-007-0320-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-007-0320-3