Abstract
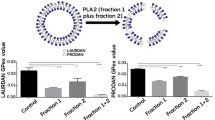
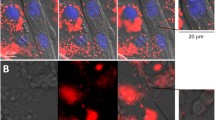
As a rule, zootoxins are complex and biologically active, and therefore the greater part of zootoxins is subjected to biotransformation and interacts with biological membranes. In this case, the interaction of different venom components with the membranes is not always the same. The present study shows how the giant unilamellar vesicles (GUV) from bovine brain proteolipids interact with Macrovipera lebetina obtusa venom. GUV (mean diameter 30 μm) were formed by the electroformation method. We used 8-anilino-1-naphthalenesulfonic acid and pyrene as fluorescence probes, which allowed us to quantify the fluidity changes in the membrane by measuring the fluorescence intensity.





Similar content being viewed by others
References
Angelova MI, Soleau S, Meleard P, Faucon JF, Bothorel P (1992) Preparation of giant vesicles by external fields. Kinetics and application. Progr Colloid Polym Sci 89:127–131
Ayvazian NM, Ghazaryan NA (2012) Lipid bilayer condition abnormalities following Macroviperalebetinaobtusa snake envenomation. Toxicon 60:607–613
Bagatolli LA, Gratton E (1999) Two-photon fluorescence microscopy observations of shape changes at the phase transition in phospholipid giant unilamellar vesicles. Biophys J 77:2090–2101
Bagatolli LA, Gratton EA (2000) Correlation between lipid domain shape and binary phospholipid mixture composition in free standing bilayers: a two-photon fluorescence microscopy study. Biophys J 79:434–447
Bordushkov Yu, Goroshinskaya I, Frantzyantz E (1993) Effects of electromagnetic field exposure on some structural and functional properties of blood lymphocyte and erythrocyte membranes. Biofizika 38:1098–1103
Burack WR, Dibble AR, Allietta MM, Biltonen RL (1997) Changes in vesicle morphology induced by lateral phase separation modulate phospholipase A2 activity. Biochemistry 36:10551–10557
Eble JA, Bruckner P, Mayer U (2003) Viperalebetina venom contains two disintegrins inhibiting laminin-binding beta1 integrins. J Biol Chem 278:26488–26496
Galla HJ, Sackman E (1974) Lateral diffusion in the hydrophobic region of membranes: use of pyreneexcimers as optical probes. Biochim Biophys Acta 339:103–115
Lakin GF (1992) Biometry. Vishaya Shcola, Moscow [in Russian]
Mueller P, Rudin D, Tien H (1962) Reconstruction of cell membranes structure in vitro and its transformation into an excitable system. Nature 194:979–980
Sanz L, Ayvazyan N, Calvete JJ (2008) Snake venomics of the Armenian mountain vipers Macroviperalebetinaobtusa and Viperaraddei. J Proteomics 71:198–209
Verstraeten SV, Hammerstone JF, Keen CL, Fraga CG, Oteiza PI (2005) Antioxidant and membrane effects of procyanidin dimmers and trimers isolated from peanut and cocoa. J Agric Food Chem 53:5041–5048
Zakharyan AE, Ayvazian NM (2005) Modeling of BLMs in aspect of phylogenetic development of vertebrates. In: Ottova-Leitmannova A (ed) Advances in planar lipid bilayers and liposomes, 2nd edn. Elsevier, Amsterdam, pp 238–259
Zobelinsky CA, Pomazansky LF, Chirkovskaya EB (1984) Brain proteolirids of representatives of different vertebratae classes. J Evol Biochem Physiol 20:240–246
Acknowledgments
This work was supported by grant N11-1f231 from the State Committee of Science, Ministry of Education and Science RA.
Conflict of interest
The authors report that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghazaryan, N.A., Ghulikyan, L.A. & Ayvazyan, N.M. Morphological Changes of Proteolipid Giant Unilamellar Vesicles Affected by Macrovipera lebetina obtusa Venom Visualized with Fluorescence Microscope. J Membrane Biol 246, 627–632 (2013). https://doi.org/10.1007/s00232-013-9576-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00232-013-9576-1