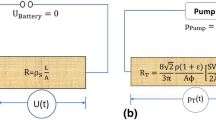
Abstract
We present experimental and theoretical results of electroporation of small patches of planar lipid bilayers by means of linearly rising current. The experiments were conducted on ~120-μm-diameter patches of planar phospholipid bilayers. The steadily increasing voltage across the bilayer imposed by linearly increasing current led to electroporation of the membrane for voltages above a few hundred millivolts. This method shows new molecular mechanisms of electroporation. We recorded small voltage drops preceding the breakdown of the bilayer due to irreversible electroporation. These voltage drops were often followed by a voltage re-rise within a fraction of a second. Modeling the observed phenomenon by equivalent electric circuits showed that these events relate to opening and closing of conducting pores through the bilayer. Molecular dynamics simulations performed under similar conditions indicate that each event is likely to correspond to the opening and closing of a single pore of about 5 nm in diameter, the conductance of which ranges in the 100-nS scale. This combined experimental and theoretical investigation provides a better quantitative characterization of the size, conductance and lifetime of pores created during lipid bilayer electroporation. Such a molecular insight should enable better control and tuning of electroporation parameters for a wide range of biomedical and biotechnological applications.








Similar content being viewed by others
References
Benz R, Janko K (1976) Voltage-induced capacitance relaxation of lipid bilayer membranes: effects of membrane composition. Biochim Biophys Acta 455:721–738
Bockmann RA, de Groot BL, Kakorin S et al (2008) Kinetics, statistics, and energetics of lipid membrane electroporation studied by molecular dynamics simulations. Biophys J 95:1837–1850
Darden T, York D, Pedersen L (1993) Particle mesh Ewald—an n.log(n) method for Ewald sums in large systems. J Chem Phys 98:10089–10092
Delemotte L, Dehez F, Treptow W, Tarek M (2008) Modeling membranes under a transmembrane potential. J Phys Chem B 112:5547–5550
Essmann U, Perera L, Berkowitz M et al (1995) A smooth particle mesh Ewald method. J Chem Phys 103:8577–8593
Genco I, Gliozzi A, Relini A et al (1993) Electroporation in symmetric and asymmetric membranes. Biochim Biophys Acta 1149:10–18
Glaser R, Leikin S, Chernomordik L et al (1988) Reversible electrical breakdown of lipid bilayers—formation and evolution of pores. Biochim Biophys Acta 940:275–287
Golzio M, Teissie J, Rols M (2002) Direct visualization at the single-cell level of electrically mediated gene delivery. Proc Natl Acad Sci USA 99:1292–1297
Gurtovenko A, Vattulainen I (2005) Pore formation coupled to ion transport through lipid membranes as induced by transmembrane ionic charge imbalance: atomistic molecular dynamics study. J Am Chem Soc 127:17570–17571
Gurtovenko A, Anwar J, Vattulainen I (2010) Defect-mediated trafficking across cell membranes: insights from in silico modeling. Chem Rev 110:6077–6103
Heller R, Gilbert R, Jaroszeski M (1999) Clinical applications of electrochemotherapy. Adv Drug Deliv Rev 35:119–129
Henin J, Shinoda W, Klein ML (2008) United-atom acyl chains for CHARMM phospholipids. J Phys Chem B 112:7008–7015
Jorgensen W, Chandrasekhar J, Madura J et al (1983) Comparison of simple potential functions for simulating liquid water. J Chem Phys 79:926–935
Kale L, Skeel R, Bhandarkar M et al (1999) NAMD2: greater scalability for parallel molecular dynamics. J Comput Phys 151:283–312
Kalinowski S, Figaszewski Z (1995a) A 4-electrode system for measurement of bilayer lipid membrane capacitence. Meas Sci Technol 6:1043–1049
Kalinowski S, Figaszewski Z (1995b) A 4-electrode potentiostat-galvanostat for studies of bilayer lipid membranes. Meas Sci Technol 6:1050–1055
Kalinowski S, Ibron G, Bryl K, Figaszewski Z (1998) Chronopotentiometric studies of electroporation of bilayer lipid membranes. Biochim Biophys Acta 1369:204–212
Kandasamy SK, Larson RG (2006) Cation and anion transport through hydrophilic pores in lipid bilayers. J Chem Phys 125:074901
Koronkiewicz S, Kalinowski S (2004) Influence of cholesterol on electroporation of bilayer lipid membranes: chronopotentiometric studies. Biochim Biophys Acta 1661:196–203
Koronkiewicz S, Kalinowski S, Bryl K (2002) Programmable chronopotentiometry as a tool for the study of electroporation and resealing of pores in bilayer lipid membranes. Biochim Biophys Acta 1561:222–229
Kotnik T, Bobanovic F, Miklavcic D (1997) Sensitivity of transmembrane voltage induced by applied electric fields—a theoretical analysis. Bioelectrochem Bioenerg 43:285–291
Kotnik T, Macek-Lebar A, Miklavcic D, Mir L (2000) Evaluation of cell membrane electropermeabilization by means of a nonpermeant cytotoxic agent. Biotechniques 28:921–926
Kotulska M, Koronkiewicz S, Kalinowski S (2004) Self-similar processes and flicker noise from a fluctuating nanopore in a lipid membrane. Phys Rev E 69:031920
Kotulska M, Kubica K, Koronkiewicz S, Kalinowski S (2007) Modeling the induction of lipid membrane electropermeabilization. Bioelectrochemistry 70:64–70
Kotulska M, Basalyga J, Derylo M, Sadowski P (2010) Metastable pores at the onset of constant-current electroporation. J Membr Biol 236:37–41
Kramar P, Miklavcic D, Lebar AM (2007) Determination of the lipid bilayer breakdown voltage by means of linear rising signal. Bioelectrochemistry 70:23–27
Kramar P, Miklavcic D, Lebar AM (2009) A system for the determination of planar lipid bilayer breakdown voltage and its applications. IEEE Trans Nanobiosci 8:132–138
Marty M, Sersa G, Garbay JR et al (2006) Electrochemotherapy—an easy, highly effective and safe treatment of cutaneous and subcutaneous metastases: results of ESOPE (European Standard Operating Procedures of Electrochemotherapy) study. EJC Suppl 4:3–13
Melikov K, Frolov V, Shcherbakov A et al (2001) Voltage-induced nonconductive pre-pores and metastable single pores in unmodified planar lipid bilayer. Biophys J 80:1829–1836
Mir LM, Banoun H, Paoletti C (1988) Introduction of definite amounts of nonpermeant molecules into living cells after electropermeabilization—direct access to the cytosol. Exp Cell Res 175:15–25
Mir L, Orlowski S, Belehradek J et al (1995) Biomedical applications of electric pulses with special emphasis on antitumor electrochemotherapy. Bioelectrochem Bioenerg 38:203–207
Montal M, Mueller P (1972) Formation of bimolecular membranes from lipid monolayers and a study of their electrical properties. Proc Natl Acad Sci USA 69:3561–3566
Neumann E, Rosenheck K (1972) Permeability changes induced by electric impulses in vesicular membranes. J Membr Biol 10:279–290
Phillips J, Braun R, Wang W et al (2005) Scalable molecular dynamics with NAMD. J Comput Chem 26:1781–1802
Prausnitz MR, Bose VG, Langer R, Weaver JC (1993) Electroporation of mammalian skin—a mechanism to enhance transdermal drug-delivery. Proc Natl Acad Sci USA 90:10504–10508
Pucihar G, Mir L, Miklavcic D (2002) The effect of pulse repetition frequency on the uptake into electropermeabilized cells in vitro with possible applications in electrochemotherapy. Bioelectrochemistry 57:167–172
Rols M, Teissie J (1998) Electropermeabilization of mammalian cells to macromolecules: control by pulse duration. Biophys J 75:1415–1423
Sersa G, Cemazar M, Miklavcic D (1995) Antitumor effectiveness of electrochemotherapy with cis-diamminedichloroplatinum(II) in mice. Cancer Res 55:3450–3455
Tarek M (2005) Membrane electroporation: a molecular dynamics simulation. Biophys J 88:4045–4053
Tarek M, Delemotte L (2010) Electroporation of lipid membranes. In: Pakhomov A, Miklavčič D, Markov M (eds) Advanced electroporation techniques in biology and medicine. Taylor and Francis/CRC Press, Boca Raton, FL
Teissie J, Rols M (1993) An experimental evaluation of the critical potential difference inducing cell-membrane electropermeabilization. Biophys J 65:409–413
Teissie J, Eynard N, Gabriel B, Rols M (1999) Electropermeabilization of cell membranes. Adv Drug Deliv Rev 35:3–19
Teissie J, Escoffre JM, Rols MP, Golzio M (2008) Time dependence of electric field effects on cell membranes. A review for a critical selection of pulse duration for therapeutical applications. Radiol Oncol 42:196–206
Tieleman D (2004) The molecular basis of electroporation. Biophys J 86:371A–372A
Tieleman D, Leontiadou H, Mark A, Marrink S (2003) Simulation of pore formation in lipid bilayers by mechanical stress and electric fields. J Am Chem Soc 125:6382–6383
Tien HT (1974) Bilayer lipid membranes. Marcel Dekker, New York
Tuma T, Buermen Á (2009) Circuit simulation with SPICE OPUS: theory and practice. Birkhäuser, Boston
Villemejane J, Mir LM (2009) Physical methods of nucleic acid transfer: general concepts and applications. Br J Pharmacol 157:207–219
Ziegler MJ, Vernier PT (2008) Interface water dynamics and porating electric fields for phospholipid bilayers. J Phys Chem B 112:17003
Zimmermann U, Pilwat G, Beckers F, Riemann F (1976) Effects of external electrical fields on cell-membranes. Bioelectrochem Bioenerg 3:58–83
Acknowledgements
This work was in part supported by various grants from the Slovenian Research Agency and bilateral cooperation programs between Poland and Slovenia and between France and Slovenia (PROTEUS). The research was conducted in the scope of the EBAM European Associated Laboratory. Simulations were performed using HPC resources from GENCI-CINES (Grant 2010-075137). We thank A. Burmen for valuable discussion regarding SPICE modeling. M. T. acknowledges the support of the French Agence Nationale de la Recherche (Grant ANR-10_BLAN-916-03-INTCELL).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kramar, P., Delemotte, L., Maček Lebar, A. et al. Molecular-Level Characterization of Lipid Membrane Electroporation using Linearly Rising Current. J Membrane Biol 245, 651–659 (2012). https://doi.org/10.1007/s00232-012-9487-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00232-012-9487-6