Abstract
Objective: To determine the weight-reducing efficacy of orlistat, a novel gastrointestinal lipase inhibitor, and to define the optimal dosage regimen and establish the tolerability of the drug when used for a 6-month treatment period.
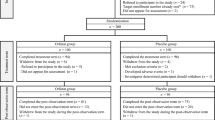
Methods: The study was a multicentre randomised, double-blind, parallel group in design and involved 676 obese male and female subjects aged at least 18 years with a body mass index between 28 and 43 kg · m−2. Following a 5-week placebo run-in period, subjects were randomised to receive orlistat 30 mg, 60 mg, 120 mg, 240 mg or matching placebo three times a day (tid) for 24 weeks during meals. Patients were maintained on a mildly hypocaloric diet throughout the study period. The primary efficacy parameter was body weight change over time.
Results: Orlistat resulted in a significantly greater mean loss of body weight than observed in the placebo group. In absolute terms, mean weight loss was greatest in the 120 mg group (9.8%). More orlistat- than placebo-treated patients lost >10% of initial body weight (37% of the 120 mg group vs 19% of the placebo group). Orlistat was well tolerated. Predictably, in view of its known pharmacological effects, more orlistat-treated patients experienced gastrointestinal events. Mean levels of vitamins A, D and E, and β-carotene remained within the clinical reference ranges in all treatment groups and rarely required supplementation. After 24 weeks, plasma concentrations of orlistat were either non-measurable or detected at the assay's limit of quantitation.
Conclusion: Orlistat treatment results in a dose-dependent reduction in body weight in obese subjects and is well tolerated. Orlistat 120 mg tid represents the optimal dosage regimen.
Similar content being viewed by others
Author information
Authors and Affiliations
Consortia
Additional information
Received: 24 July 1997 / Accepted in revised form: 4 October 1997
Rights and permissions
About this article
Cite this article
Van Gaal, L., Broom, J., Enzi, G. et al. Efficacy and tolerability of orlistat in the treatment of obesity: a 6-month dose-ranging study. E J Clin Pharmacol 54, 125–132 (1998). https://doi.org/10.1007/s002280050433
Issue Date:
DOI: https://doi.org/10.1007/s002280050433