Abstract
Purpose
To assess the antiplatelet effect of cilostazol clinically, we compared the effects of cilostazol in combination with clopidogrel on various platelet function tests.
Methods
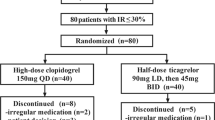
We recruited patients with ischemic stroke at high risk of recurrence who were treated with clopidogrel alone within 180 days after stroke onset. Subjects underwent baseline platelet function tests, and were then randomly assigned to receive dual antiplatelet therapy (DAPT) comprising clopidogrel and cilostazol or clopidogrel monotherapy (SAPT). After 6 months, platelet function was measured again and compared to that at baseline in each group, and the rate of change was compared between groups.
Results
Thirty-four patients were enrolled, but 4 patients were excluded for various reasons. In total, 30 subjects (13 in DAPT and 17 in SAPT group) were analyzed. Adenosine diphosphate- and collagen-induced aggregation, VerifyNow P2Y12 reaction units, vasodilator-stimulated phosphoprotein (platelet reactivity index: PRI) and plasma p-selectin concentration were significantly lower (P = 0.004, 0.042, 0.049, 0.003 and 0.006 respectively), while VerifyNow % inhibition was significantly higher at 6 months compared to baseline (P = 0.003) in the DAPT group only. Comparison of the rate of change in each parameter from baseline to 6 months showed that while PRI decreased at a greater rate (P = 0.012), VerifyNow % inhibition increased at a greater rate (P = 0.003) in the DAPT group than the SAPT group.
Conclusions
The inhibitory effects of adjunctive cilostazol added to clopidogrel on platelet function differed by type of platelet function test. VerifyNow % inhibition and PRI were more inhibited than the other platelet function tests.
Trial registration
CSPS.com substudy in TWMU (UMIN000026672), registered on April 1, 2017. This study was performed as a substudy of CSPS.com (UMIN000012180, registered on October 31, 2013) and was retrospectively registered.


Similar content being viewed by others
Availability of data and materials
The dataset analyzed during the current study are available from the corresponding author on reasonable request.
References
Goto F, Tohgi H, Hirai S, Terashi A, Fukuuchi Y, Otomo E, Shinohara Y, Itoh E, Matsuda T, Sawada T, Yamaguchi T, Nishimaru K, Ohashi Y (2000) Cilostazol stroke prevention study: A placebo controlled double-blind trial for secondary prevention of cerebral infarction. J Stroke Cerebrovasc Dis 9:147–157. https://doi.org/10.1053/jscd.2000.7216
Shinohara Y, Katayama Y, Uchiyama S, Yamaguchi T, Handa S, Matsuoka K, Ohashi Y, Tanahashi N, Yamamoto H, Genka C, Kitagawa Y, Kusuoka H, Nishimaru K, Tsushima M, Koretsune Y, Sawada T, Hamada C, CSPS 2 group (2010) Cilostazol for prevention of secondary stroke (CSPS2): an aspirin-controlled, double-blind, randomised non-inferiority trial. Lancet Neurol 9:959–968. https://doi.org/10.1016/S1474-4422(10)70198-8
Toyoda K, Uchiyama S, Yamaguchi T, Easton JD, Kimura K, Hoshino H, Sakai N, Okada Y, Tanaka K, Origasa H, Naritomi H, Houkin K, Yamaguchi K, Isobe M, Minematsu K, on behalf of the CSPS.com Trial Investigators (2019) Dual antiplatelet therapy using cilostazol for secondary prevention in patients with high-risk ischaemic stroke in Japan: a multicentre, open-label, randomised controlled trial. Lancet Neurol 18:539–548. https://doi.org/10.1016/S1474-4422(19)30148-6
Liu Y, Shakur Y, Yoshitake M, Kambayashi J (2001) Cilostazol (Pletal®): a dual inhibitor of cyclic nucleotide phosphodiesterase type 3 and adenosine uptake. Cardiovasc Drug Rev 19:369–386. https://doi.org/10.1111/j.1527-3466.2001.tb00076.x
Inoue T, Sohma R, Morooka S (1999) Cilostazol inhibits the expression of activation-dependent membrane surface glycoprotein on the surface of platelets stimulated in vitro. Thromb Res 93:137–143. https://doi.org/10.1016/S0049-3848(98)00172-8
Kariyazono H, Nakamura K, Shinkawa T, Yamaguchi T, Sakata R, Yamada K (2001) Inhibition of platelet aggregation and the release of p-selectin from platelets by cilostazol. Thromb Res 101:445–453. https://doi.org/10.1016/S0049-3848(00)00415-1
Yamamoto H, Takahashi K, Watanabe H, Yoshikawa Y, Shirakawa R, Higashi T, Kawato M, Ikeda T, Tabuchi A, Morimoto T, Kita T, Horiuchi H (2008) Evaluation of the antiplatelet effects of cilostazol, a phosphodiesterase 3 inhibitor, by VASP phosphorylation and platelet aggregation. Circ J 72:1844–1851. https://doi.org/10.1253/circj.CJ-08-0289
Satoh K, Fukasawa I, Kanemaru K, Yoda S, Kimura Y, Inoue O, Ohta M, Kinouchi H, Ozaki Y (2012) Platelet aggregometry in the presence of PGE1 provides a reliable method for cilostazol monitoring. Thromb Res 130:616–621. https://doi.org/10.1016/j.thromres.2012.05.030
Yamanouchi J, Hato T, Niiya T, Nakagawa K, Kumon Y, Fujiwara H, Yakushijin Y, Yasukawa M (2011) Vasodilator-stimulated phosphoprotein (VASP) phosphorylation assay for platelet response to cilostazol. Platelets 22:135–142. https://doi.org/10.3109/09537104.2010.525976
Geiger J, Brich J, Hönig-Liedl P, Eigenthaler M, Schanzenbächer P, Herbert JM, Walter U (2007) Specific impairment of human platelet P2Y(AC)ADP receptor-mediated signaling by the antiplatelet drug clopidogrel. Arterioscler Vasc Biol 7:249–52. https://doi.org/10.1161/01.atv.19.8.2007
Lee BK, Lee SW, Park SW, Lee SW, Park DW, Kim YH, Lee CW, Hong MK, Kim JJ, Jang S, Chi HS, Park SJ (2007) Effects of triple antiplatelet therapy (aspirin, clopidogrel, and cilostazol) on platelet aggregation and p-selectin expression in patients undergoing coronary artery stent implantation. Am J Cardiol 100:610–614. https://doi.org/10.1016/j.amjcard.2007.03.070
Angiolillo DJ, Capranzano P, Goto S, Aslam M, Desai B, Charlton RK, Suzuki Y, Box LC, Shoemaker SB, Zenni MM, Bass GLA (2008) A randomized study assessing the impact of cilostazol on platelet function profiles in patients with diabetes mellitus and coronary artery disease on dual antiplatelet therapy: results of the OPTIMUS-2 study. Eur Heart J 29:2202–2211. https://doi.org/10.1093/eurheartj/ehn287
Jeong YH, Lee SW, Choi BR, Kim IS, Seo MK, Kwak CH, Hwang JY, Park SW (2009) Randomized comparison of adjunctive cilostazol versus high maintenance dose clopidogrel in patients with high post-treatment platelet reactivity. Results of the ACCEL-RESISTANCE (Adjunctive Cilostazol Versus High Maintenance Dose Clopidogrel in Patients With Clopidogrel Resistance) Randomized Study. J Am Coll Cardiol 53:1101–1109. https://doi.org/10.1016/j.jacc.2008.12.025
Toyoda K, Uchiyama S, Hoshino H, Kimura K, Origasa H, Naritomi H, Minematsu K, Yamaguchi T, for the CSPS.com Study Investigators (2015) Protocol for Cilostazol Stroke Prevention Study for Antiplatelet Combination (CSPS.com): a randomized, open-label, parallel-group trial. Int J Stroke 10:253–258. https://doi.org/10.1111/ijs.12420
Yamazaki M, Uchiyama S, Iwata M (2001) Measurement of platelet fibrinogen binding and p-selectin expression by flow cytometry in patients with cerebral infarction. Thromb Res 104:197–205. https://doi.org/10.1016/S0049-3848(01)00350-4
Yamazaki M, Ohnishi T, Hosokawa K, Yamaguchi K, Yoneyama T, Kawashima A, Okada Y, Kitagawa K, Uchiyama S (2016) Measurement of residual platelet thrombogenicity under arterial shear conditions in cerebrovascular disease patients receiving antiplatelet therapy. J Thromb Haemost 14:1788–1797. https://doi.org/10.1111/jth.13391
Fisher RG, Teitel ER, Yee AT, Chuang AZ (2013) Evaluation of the effectiveness of the AggreGuide A-100 in measuring platelet aggregation in individuals taking a high or low dose of aspirin. Lab Medicine 44:e83–e91. https://doi.org/10.1309/LMJF9WK85YAAAEYV
Madhavi R, Rajasekhar D, Vanajakshamma V, Reddy GO, Kapil C, Sowjenya G (2018) Variability in antiplatelet drugs’ response in Indian patients undergoing percutaneous coronary intervention and outcome modification by tailored antiplatelet therapy. J Cardiol Cardiovasc Ther 9:555762. https://doi.org/10.19080/JOCCT.2018.09.555762
Hosokawa K, Ohnishi T, Fukasawa M, Kondo T, Sameshima H, Koide T, Tanaka K, Maruyama I (2012) A microchip flow-chamber system for quantitative assessment of the platelet thrombus formation process. Microvasc Res 83:154–161. https://doi.org/10.1016/j.mvr.2011.11.007
Hosokawa K, Ohnishi T, Sameshima H, Miura N, Ito T, Koide T, Maruyama I (2013) Analysing responses to aspirin and clopidogrel by measuring platelet thrombus formation under arterial flow conditions. Thromb Haemost 109:102–111. https://doi.org/10.1160/TH12-06-0441
Maruyama H, Takeda H, Dembo T, Nagoya H, Kato Y, Fukuoka T, Deguchi I, Horiuchi Y, Tanahashi N (2011) Clopidogrel resistance and the effect of combination cilostazol in patients with ischemic atroke or carotid artery stenting using the VerifyNow P2Y12 assay. Intern Med 50:695–698. https://doi.org/10.2169/internalmedicine.50.4623
KhannaV HA, Mikael R, Sambu N, Englyst N, Curzen N (2014) Does the VerifyNow P2Y12 assay overestimate “therapeutic response” to clopidogrel? Insights using short thrombelastography Thromb Haemost 111:1150–1159. https://doi.org/10.1160/TH13-10-0856
Bieber M, Schuhmann MK, Volz J, Kumar GJ, Vaidya JR, Nieswandt B, Pham M, Stoll G, Kleinschnitz C, Kraft P (2019) Description of a novel phosphodiesterase (PDE)-3 Inhibitor protecting mice from ischemic stroke independent from platelet function. Stroke 50:478–486. https://doi.org/10.1161/STROKEAHA.118
Acknowledgements
We thank Miss Yamamoto for her assistance with the platelet function tests and Heidi Tran, PhD, of DMC Corp. for English language editing.
Funding
CSPS.com was supported by the Japan Cardiovascular Research Foundation, which was sponsored by Otsuka Pharmaceutical Co. Ltd. This platelet function study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
MY contributed to the study conception and design, performance of platelet function tests, analysis and interpretation of the data, and was responsible for the drafting, editing, and submitting the manuscript for publication. YS contributed to the recruitment of subjects. TO contributed to the T-TAS measurements (platelet thrombus formation). KH contributed to revising the manuscript for intellectual content. JD contributed to the AggreGuide measurements, writing of the manuscript and subsequent revision for intellectual content. KK contributed to the study conception and design, and critically appraised and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures were performed in accordance with the ethics standards of Tokyo Women’s Medical University and the Declaration of Helsinki. This study was approved by the Tokyo Women’s Medical University Ethics Committee (approval number 160808). Written informed consent was obtained from all subjects prior to participation in the study.
Consent for publication
Not applicable.
Competing interests
TO and KH are employees of Fujimori Kogyo Co., Ltd. JD has received consulting fees from Fujimori Kogyo Co., Ltd. and Aggredyne, Inc. KK received lecture fees and a research grant from Otsuka Pharmaceutical Co. Ltd. and Sanofi Co. The other authors state that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Masako Yamazaki is currently at the Department of Artificial Intelligence Medicine Graduate School of Medicine, Chiba University.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yamazaki, M., Shirai, Y., Ohnishi, T. et al. Differential inhibition of platelet function by cilostazol in combination with clopidogrel. Eur J Clin Pharmacol 79, 1623–1630 (2023). https://doi.org/10.1007/s00228-023-03553-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-023-03553-w