Abstract
Purpose
Idiopathic nephrotic syndrome (INS) is the most frequent form of childhood nephrotic syndrome. Steroids represent the best therapeutic option; however, inter-individual differences in their efficacy and side effects have been reported. To date, there is no way to predict patients’ resistance and/or dependence. Alterations in the cytokine profile of INS patients might contribute to proteinuria and glomerular damage and affect drug sensitivity.
Methods
The cytokine plasma levels were measured in 21 INS children at diagnosis to investigate the association among cytokines pattern and clinical response. Patients were selected on the basis of their clinical response: 7 steroid sensitive (SS), 7 dependent (SD), and 7 resistant (SR). Significant results were then analyzed in 41 additional pediatric INS patients.
Results
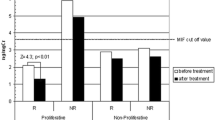
Within the 48 cytokines analyzed, macrophage migration inhibitory factor (MIF) was a good predictor of steroid response. Indeed, SR patients showed significantly higher MIF plasma levels compared with all others (p = 0.022; OR = 4.3, 95%CI = 1.2–25.4): a cutoff concentration of MIF > 501 pg/ml significantly discriminated SR patients (sensitivity = 85.7%, specificity = 71.4%). On the contrary, SD patients showed lower MIF plasma levels compared with others (p = 0.010; OR = 0.12, 95%CI = 9.2 × 10−3–6.7 × 10−1). Significant results were confirmed in the entire cohort.
Conclusions
Our comprehensive cytokine analysis indicates that assessing MIF plasma levels at diagnosis could predict response to glucocorticoids in children with INS.





Similar content being viewed by others
References
Barnes PJ (2010) Mechanisms and resistance in glucocorticoid control of inflammation. J Steroid Biochem Mol Biol 120:76–85. https://doi.org/10.1016/j.jsbmb.2010.02.018
Barnes PJ, Adcock IM (2009) Glucocorticoid resistance in inflammatory diseases. Lancet 373:1905–1917. https://doi.org/10.1016/S0140-6736(09)60326-3
Eddy AA, Symons JM (2003) Nephrotic syndrome in childhood. Lancet 362:629–639. https://doi.org/10.1016/S0140-6736(03)14184-0
Carlotti AP, Franco PB, Elias LL, Facincani I, Costa EL, Foss N et al (2004) Glucocorticoid receptors, in vitro steroid sensitivity, and cytokine secretion in idiopathic nephrotic syndrome. Kidney Int 65:403–408. https://doi.org/10.1111/j.1523-1755.2004.00392.x
Cuzzoni E, De Iudicibus S, Bartoli F, Ventura A, Decorti G (2012) Association between BclI polymorphism in the NR3C1 gene and in vitro individual variations in lymphocyte responses to methylprednisolone. Br J Clin Pharmacol 73:651–655. https://doi.org/10.1111/j.1365-2125.2011.04130.x
Cuzzoni E, De Iudicibus S, Franca R, Stocco G, Lucafò M, Pelin M et al (2015) Glucocorticoid pharmacogenetics in pediatric idiopathic nephrotic syndrome. Pharmacogenomics 16:1631–1648. https://doi.org/10.2217/pgs.15.101
Araya CE, Wasserfall CH, Brusko TM, Mu W, Segal MS, Johnson RJ, Garin EH (2006) A case of unfulfilled expectations. Cytokines in idiopathic minimal lesion nephrotic syndrome. Pediatr Nephrol 21:603–610. https://doi.org/10.1007/s00467-006-0026-5
Garin EH (2000) Circulating mediators of proteinuria in idiopathic minimal lesion nephrotic syndrome. Pediatr Nephrol 14:872–878
Schnaper HW (1989) The immune system in minimal change nephrotic syndrome. Pediatr Nephrol 3:101–110
Farrell RJ, Kelleher D (2003) Glucocorticoid resistance in inflammatory bowel disease. J Endocrinol 178:339–346
Cuzzoni E, De Iudicibus S, Stocco G, Favretto D, Pelin M, Messina G et al (2016) In vitro sensitivity to methyl-prednisolone is associated with clinical response in pediatric idiopathic nephrotic syndrome. Clin Pharmacol Ther 100:268–274. https://doi.org/10.1002/cpt.372
Becker LA. Effect size calculators [home page on the internet] UCCS University of Colorado. 03/021/00 [2018 July 6]. Available from: http://www.uccs.edu/lbecker/effect-size
Gipson DS, Massengill SF, Yao L, Nagaraj S, Smoyer WE, Mahan JD, Wigfall D, Miles P, Powell L, Lin JJ, Trachtman H, Greenbaum LA (2009) Management of childhood onset nephrotic syndrome. Pediatrics 124:747–757. https://doi.org/10.1542/peds.2008-1559
Hodson EM, Knight JF, Willis NS, Craig JC (2000) Corticosteroid therapy in nephrotic syndrome: a meta-analysis of randomised controlled trials. Arch Dis Child 83:45–51
Hogg RJ, Portman RJ, Milliner D, Lemley KV, Eddy A, Ingelfinger J (2000) Evaluation and management of proteinuria and nephrotic syndrome in children: recommendations from a pediatric nephrology panel established at the national kidney foundation conference on proteinuria, albuminuria, risk, assessment, detection, and elimination (PARADE). Pediatrics 105:1242–1249
Hahn D, Hodson EM, Willis NS, Craig JC (2007) Corticosteroid therapy for nephrotic syndrome in children. Cochrane Database Syst Rev 3:CD001533. https://doi.org/10.1002/14651858.CD002290.pub3
Wang FF, Zhu LA, Zou YQ, Zheng H, Wilson A, Yang CD, Shen N, Wallace DJ, Weisman MH, Chen SL, Lu LJ (2012) New insights into the role and mechanism of macrophage migration inhibitory factor in steroid-resistant patients with systemic lupus erythematosus. Arthritis Res Ther 14:R103. https://doi.org/10.1186/ar3828
Calandra T, Roger T (2003) Macrophage migration inhibitory factor: a regulator of innate immunity. Nat Rev Immunol 3:791–800. https://doi.org/10.1038/nri1200
Bacher M, Metz CN, Calandra T, Mayer K, Chesney J, Lohoff M, Gemsa D, Donnelly T, Bucala R (1996) An essential regulatory role for macrophage migration inhibitory factor in t-cell activation. Proc Natl Acad Sci U S A 93:7849–7854
Calandra T, Bernhagen J, Metz CN, Spiegel LA, Bacher M, Donnelly T, Cerami A, Bucala R (1995) MIF as a glucocorticoid-induced modulator of cytokine production. Nature 377:68–71. https://doi.org/10.1038/377068a0
Daun JM, Cannon JG (2000) Macrophage migration inhibitory factor antagonizes hydrocortisone-induced increases in cytosolic IkappaBalpha. Am J Physiol Regul Integr Comp Physiol 279:R1043–R1049. https://doi.org/10.1152/ajpregu.2000.279.3.R1043
Mitchell RA, Metz CN, Peng T, Bucala R (1999) Sustained mitogen-activated protein kinase (MAPK) and cytoplasmic phospholipase a2 activation by macrophage migration inhibitory factor (MIF). Regulatory role in cell proliferation and glucocorticoid action. J Biol Chem 274:18100–18106
Bevilacqua A, Ceriani MC, Capaccioli S, Nicolin A (2003) Post-transcriptional regulation of gene expression by degradation of messenger RNAs. J Cell Physiol 195:356–372. https://doi.org/10.1002/jcp.10272
Roger T, Chanson AL, Knaup-Reymond M, Calandra T (2005) Macrophage migration inhibitory factor promotes innate immune responses by suppressing glucocorticoid-induced expression of mitogen-activated protein kinase phosphatase-1. Eur J Immunol 35:3405–3413. https://doi.org/10.1002/eji.200535413
Santos LL, Morand EF (2009) Macrophage migration inhibitory factor: a key cytokine in ra, sle and atherosclerosis. Clin Chim Acta 399:1–7. https://doi.org/10.1016/j.cca.2008.09.014
Leech M, Metz C, Hall P, Hutchinson P, Gianis K, Smith M, Weedon H, Holdsworth SR, Bucala R, Morand EF (1999) Macrophage migration inhibitory factor in rheumatoid arthritis: evidence of proinflammatory function and regulation by glucocorticoids. Arthritis Rheum 42:1601–1608. https://doi.org/10.1002/1529-0131(199908)42:8<1601::AID-ANR6>3.0.CO;2-B
Bruchfeld A, Carrero JJ, Qureshi Lindholm B, Barany P, Heimburger O, Hu M et al (2009) Elevated serum macrophage migration inhibitory factor (mif) concentrations in chronic kidney disease (CKD) are associated with markers of oxidative stress and endothelial activation. Mol Med 15:70–75. https://doi.org/10.2119/molmed.2008.00109
Berdeli A, Mir S, Ozkayin N, Serdaroglu E, Tabel Y, Cura A (2005) Association of macrophage migration inhibitory factor -173C allele polymorphism with steroid resistance in children with nephrotic syndrome. Pediatr Nephrol 20:1566–1571. https://doi.org/10.1007/s00467-005-1930-9
Vivarelli M, D’Urbano LE, Stringini G, Ghiggeri GM, Caridi G, Donn R et al (2008) Association of the macrophage migration inhibitory factor -173*C allele with childhood nephrotic syndrome. Pediatr Nephrol 23:743–748. https://doi.org/10.1007/s00467-007-0729-2
Choi HJ, Cho HY, Ro H, Lee SH, Han KH, Lee H, Kang HG, Ha IS, Choi Y, Cheong HI (2011) Polymorphisms of the MDR1 and MIF genes in children with nephrotic syndrome. Pediatr Nephrol 26:1981–1988. https://doi.org/10.1007/s00467-011-1903-0
Ramayani OR, Sekarwana N, Trihono PP, Sadewa AH, Lelo A (2016) A genetic study of steroid-resistant nephrotic syndrome: relationship between polymorphism −173 G to C in the MIF gene and serum level MIF in children. J Dev Orig Health Dis 7:102–107. https://doi.org/10.1017/S2040174415007850
Acknowledgments
We thank all the members of Nefrokid Study Group, whose collaboration was essential in order to perform the study. The following institutions participated in the study: Ospedale della Gruccia Montevarchi (Arezzo); Ospedale San Donato Arezzo; Ospedale di Asola - Asola (MN); Policlinico-ospedale Giovanni XXIII, Bari; AUO di Bologna, Policlinico S.Orsola-Malpighi; Ospedale dei Bambini di Brescia; AUSL Modena Presidio Ospedaliero Carpi, Ospedale Ramazzini; AUSL di Cesena Ospedale Maurizio Bufalini; Ospedale di Circolo di Desio (MI); Ospedale di Esine (BS); A.O.Universitaria di Ferrara, Arcispedale S. Anna; Azienda Ospedaliera Universitaria Meyer Firenze; Ospedale Gaslini, Genova; Ospedale Santa Maria della Scaletta, AUSL di Imola; Ospedale “C. Poma” - Mantova; Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico - Milano; Ospedale dei Bambini “Vittore Buzzi” - Milano; Azienda Ospedaliero-Universitaria Policlinico di Modena; Fondazione MBBM, S. Gerardo - Monza; ARNAS Civico, Di Cristina e Fatebenefratelli, Palermo; AO San Salvatore Pesaro; Ospedale Guglielmo da Saliceto, Piacenza; Ospedale Santa Maria degli Angeli, Pordenone; AO. Santa Maria Nuova di Reggio Emilia; Ospedale degli Infermi Rimini; Ospedale Generale Provinciale di Saronno (VA); Nuovo Ospedale Civile Sassuolo; Ospedale di Circolo Galmarini - Tradate (VA); Ospedale Pediatrico “Burlo Garofalo,” Trieste; Ospedale “Filippo Del Ponte” - Varese; Ospedale di Vimercate (MI).
Funding
EC was supported by the Italian Society of Pharmacology grant SIF-MSD Italy 2015. We would also thank the charity organization “Il Sogno di Stefano” and The Nando Peretti Foundation for their generous financial support.
Author information
Authors and Affiliations
Contributions
Eva Cuzzoni designed the research, wrote the manuscript, planned and performed experiments, and analyzed data; Raffaella Franca wrote and revised the manuscript; Sara De Iudicibus designed the research and analyzed data; Annalisa Marcuzzi performed experiments and analyzed data; Marianna Lucafò performed experiments and analyzed data; Marco Pelin performed experiments; Diego Favretto performed experiments; William Morello enrolled patients and revised the manuscript, Luciana Ghio enrolled patients and revised the manuscript, Claudio La Scola enrolled patients and revised the manuscript, Francesca Mencarelli enrolled patients and revised the manuscript, Andrea Pasini designed the research, enrolled patients and revised the manuscript; Giovanni Montini designed the research, enrolled patients and revised the manuscript; Giuliana Decorti analyzed data and revised the manuscript; Gabriele Stocco analyzed data, wrote and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cuzzoni, E., Franca, R., De Iudicibus, S. et al. MIF plasma level as a possible tool to predict steroid responsiveness in children with idiopathic nephrotic syndrome. Eur J Clin Pharmacol 75, 1675–1683 (2019). https://doi.org/10.1007/s00228-019-02749-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-019-02749-3