Abstract
Aim
The aim of our study was to study the pattern of prescription of direct-acting oral anticoagulants (DOACs) according to the French recommendations.
Methods
We performed a cross-sectional study using anonymous data of patients covered by the French National Health Insurance information system (SNIIRAM) from 1 January 2010 to 31 December 2013 in the area of Midi-Pyrénées (southwest of France).
Results
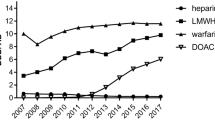
Of the 355,608 patients identified, 325,216 (91.5%) were included, of whom 22,142 received at least one DOAC. About 39.1% (8,652 patients) had DOAC in an orthopedic indication, 46.5% (10,303 patients) in a cardiac indication, and 16.1% (3568 patients) in an indeterminate indication. Overall, guidelines were largely followed as for renal function monitoring, prescribing in orthopedic indications, in cardiac indications in patients aged 80 years and older, and in the case of concomitant use of verapamil. However, inappropriate prescriptions were observed for cardiac indications, and for dosage adjustments in orthopedic indications, with respect to both the age of patients (75 years and older) and those taking verapamil or amiodarone concomitantly. Guidelines were more followed in women and patients aged 80 or more.
Conclusions
Among patients receiving DOACs, 58% were exposed to a prescription falling outside the guidelines. This study on DOAC prescription patterns revealed insufficiencies in the compliance with the French guidelines in certain indications.



Similar content being viewed by others
References
Camm AJ, Lip GYH et al (2012) Focused update of the ESC guidelines for the management of atrial fibrillation: an update of the 2010 ESC guidelines for the management of atrial fibrillation * developed with the special contribution of the European heart rhythm association. Eur Heart J 33:2719–2747. https://doi.org/10.1093/eurheartj/ehs253
Nouveaux anticoagulants oraux Eliquis (apixaban), Pradaxa (dabigatran), Xarelto (rivaroxaban): mise en garde sur les facteurs de risques hémorragiques—Lettre aux professionnels de santé—ANSM: Agence nationale de sécurité du médicament et des produits de santé. http://ansm.sante.fr/S-informer/Informations-de-securite-Lettres-aux-professionnels-de-sante/Nouveaux-anticoagulants-oraux-Eliquis-apixaban-Pradaxa-dabigatran-Xarelto-rivaroxaban-mise-en-garde-sur-les-facteurs-de-risques-hemorragiques-Lettre-aux-professionnels-de-sante. Accessed 22 Jun 2017
Research C for DE and drug safety and availability—FDA Drug Safety Communication Safety review of post-market reports of serious bleeding events with the anticoagulant Pradaxa (dabigatran etexilate mesylate). https://www.fda.gov/Drugs/DrugSafety/ucm282724.htm. Accessed 22 Jun 2017
Research C for DE and Drug Safety and Availability—FDA Drug Safety Communication Update on the risk for serious bleeding events with the anticoagulant Pradaxa (dabigatran). https://www.fda.gov/Drugs/DrugSafety/ucm326580.htm. Accessed 22 Jun 2017
Research C for DE and Drug Safety and Availability—FDA Drug Safety Communication FDA study of Medicare patients finds risks lower for stroke and death but higher for gastrointestinal bleeding with Pradaxa (dabigatran) compared to warfarin. https://www.fda.gov/Drugs/DrugSafety/ucm396470.htm. Accessed 22 Jun 2017
Boehringer Ingelheim. Pradaxa prescribing information. http://docs.boehringer-ingelheim.com/Prescribing%20Information/PIs/Pradaxa/Pradaxa.pdf. Accessed 22 June 2017
HAS Pradaxa 110mg et 150mg_Prévention de l’AVC et de l’embolie systémique chez les patients adultes présentant une fibrillation atriale non valvulaire associée à un ou plusieurs facteurs de risque. https://www.has-sante.fr/portail/upload/docs/application/pdf/2012-03/pradaxa_15022012_avis_ct10749.pdf. Accessed 22 Jun 2017
Adam SS, McDuffie JR, Ortel TL, Williams JW (2012) Comparative effectiveness of warfarin and new oral anticoagulants for the management of atrial fibrillation and venous thromboembolism: a systematic review. Ann Intern Med 157:796–807
Desai NR, Krumme AA, Schneeweiss S, Shrank WH, Brill G, Pezalla EJ, Spettell CM, Brennan TA, Matlin OS, Avorn J, Choudhry NK (2014) Patterns of initiation of oral anticoagulants in? Patients with atrial fibrillation? Quality and cost implications. Am J Med 127:1075–1082.e1. https://doi.org/10.1016/j.amjmed.2014.05.013
Kjerpeseth LJ, Ellekjær H, Selmer R, Ariansen I, Furu K, Skovlund E (2017) Trends in use of warfarin and direct oral anticoagulants in atrial fibrillation in Norway, 2010 to 2015. Eur J Clin Pharmacol 73:1417–1425
Brown JD, Shewale AR, Dherange P, Talbert JC (2016) A comparison of oral anticoagulant use for atrial fibrillation in the pre- and post-DOAC eras. Drugs Aging 33:427–436
CNAMTS Nouveaux anti-coagulants oraux: une étude de l’Assurance Maladie souligne la dynamique forte de ces nouveaux médicaments et la nécessité d’une vigilance accrue dans leur utilisation. http://ansm.sante.fr/content/download/56115/722405/version/1/file/DP-Etude-Cnam-NACO-2013.pdf. Accessed 22 Jun 2017
Maura G, Blotière P-O, Bouillon K, Billionnet C, Ricordeau P, Alla F, Zureik M (2015) Comparison of the short-term risk of bleeding and arterial thromboembolic events in nonvalvular atrial fibrillation patients newly treated with dabigatran or rivaroxaban versus vitamin K antagonists: a French Nationwide Propensity-Matched Cohort Study. Circulation 132:1252–1260
Maura G, Pariente A, Aalla F, Billionnet C (2017) Adherence with direct oral anticoagulant in non valvular atrial fibrillation new users and associated factors: a French natiowide cohort study. Pharmacoepidemiol Drug Saf 26:1367–1377. https://doi.org/10.1002/pds.4268
Moulis G, Lapeyre-Mestre M, Palmaro A, Pugnet G, Montastruc JL, Sailler L (2014) French health insurance databases: what interest for medical research? Rev Med Interne 36:411–417. https://doi.org/10.1016/j.revmed.2014.11.009
Palmaro A, Moulis G, Despas F, Dupouy J, Lapeyre-Mestre M (2016) Overview of drug data within french health insurance databases and implications for pharmacoepidemiological studies. Fundam Clin Pharmacol 30:616–624
HAS Xarelto: Avis du 17 décembre 2014. http://www.has-sante.fr/portail/upload/docs/evamed/CT-13565_XARELTO_PIC_RI_REEV_Avis3_CT13565.pdf. Accessed 22 Jun 2017
HAS Pradaxa: Avis du 17 décembre 2014. https://www.has-sante.fr/portail/upload/docs/evamed/CT-13190_PRADAXA_PIC_RI_REEV_Avis3_CT13190.pdf. Accessed 22 Jun 2017
HAS Eliquis : Avis du 17 décembre 2014. http://www.has-sante.fr/portail/upload/docs/application/pdf/2015-01/eliquis_pic_reev_avis3_modifiele16012015_ct13857.pdf. Accessed 22 Jun 2017
Kirchhof D, Benussi S, Kotecha D et al (2016) 2016 ESC guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J 37:2893–2962
Suissa S (2008) Immeasurable time bias in observational studies of drug effects on mortality. Am J Epidemiol 168:329–335
Palmaro A, Boucherie Q, Dupouy J, Micallef J, Lapeyre-Mestre M (2017) Immeasurable time bias due to hospitalization in medico-administrative databases: which impact for pharmacoepidemiological studies? Pharmacoepidemiol Drug Saf 26:544–553. https://doi.org/10.1002/pds.4193
HAS (2008) Evaluation de l’autosurveillance de l’INR chez les patients adultes traités par antivitamines K. [cité 18 sept 2014]; Disponible sur: http://www.has-sante.fr/portail/upload/docs/application/pdf/2009-02/rapport_inr_2009-02-11_11-34-20_386.pdf
Komen J, Forslund T, Hjemdahl P, Wettermark B (2017) Factors associated with antithrombotic treatment decisions for stroke prevention in atrial fibrillation in the Stockholm region after the introduction of NOACs. Eur J Clin Pharmacol 73:1315–1322
ANSM Evolution des ventes des anticoagulants oraux en France de janvier 2008 à septembre 2013. http://ansm.sante.fr/content/download/56121/722465/version/1/file/Evolution_ventes_NACO_AVK_2008-2013.pdf. Accessed 22 Jun 2017
Ruiz Ortiz M, Muñiz J, Raña Míguez P, Roldán I, Marín F, Asunción Esteve-Pastor M, Cequier A, Martínez-Sellés M, Bertomeu V, Anguita M (2017) Inappropriate doses of direct oral anticoagulants in real-world clinical practice: prevalence and associated factors. A subanalysis of the FANTASIIA registry. Europace. https://doi.org/10.1093/europace/eux316
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Diaz, H., Bagheri, H., Palmaro, A. et al. Patterns of direct oral anticoagulant drug prescription in France in 2010–2013: a study in the Midi-Pyrénées area. Eur J Clin Pharmacol 74, 945–951 (2018). https://doi.org/10.1007/s00228-018-2449-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-018-2449-x