Abstract
Purpose
This study aimed to determine possible effects of voriconazole and posaconazole on the pharmacokinetics and pharmacological effects of sublingual buprenorphine.
Methods
We used a randomized, placebo-controlled crossover study design with 12 healthy male volunteers. Subjects were given a dose of 0.4 mg (0.6 mg during placebo phase) sublingual buprenorphine after a 5-day oral pretreatment with either (i) placebo, (ii) voriconazole 400 mg twice daily on the first day and 200 mg twice daily thereafter or (iii) posaconazole 400 mg twice daily. Plasma and urine concentrations of buprenorphine and its primary active metabolite norbuprenorphine were monitored over 18 h and pharmacological effects were measured.
Results
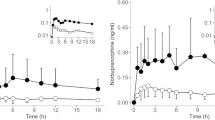
Compared to placebo, voriconazole increased the mean area under the plasma concentration-time curve (AUC0–∞) of buprenorphine 1.80-fold (90 % confidence interval 1.45–2.24; P < 0.001), its peak concentration (Cmax) 1.37-fold (P < 0.013) and half-life (t ½ ) 1.37-fold (P < 0.001). Posaconazole increased the AUC00-∞ of buprenorphine 1.25-fold (P < 0.001). Most of the plasma norbuprenorphine concentrations were below the limit of quantification (0.05 ng/ml). Voriconazole, unlike posaconazole, increased the urinary excretion of norbuprenorphine 1.58-fold (90 % confidence interval 1.18–2.12; P < 0.001) but there was no quantifiable parent buprenorphine in urine. Plasma buprenorphine concentrations correlated with the pharmacological effects, but the effects did not differ significantly between the phases.
Conclusions
Voriconazole, and to a minor extent posaconazole, increase plasma exposure to sublingual buprenorphine, probably via inhibition of cytochrome P450 3 A and/or P-glycoprotein. Care should be exercised in the combined use of buprenorphine with triazole antimycotics, particularly with voriconazole, because their interaction can be of clinical importance.



Similar content being viewed by others
References
Cowan A (1995) Update on the general pharmacology of buprenorphine. In: Cowan A, Lewis J (eds) Buprenorphine combat. Drug Abus. With a unique opioid. Wiley-Liss, New York, pp. 189–211
Mattick RP, Ali R, White JM, et al. (2003) Buprenorphine versus methadone maintenance therapy: a randomized double-blind trial with 405 opioid-dependent patients. Addiction 98:441–452
Petitjean S, Stohler R, Déglon JJ, et al. (2001) Double-blind randomized trial of buprenorphine and methadone in opiate dependence. Drug Alcohol Depend 62:97–104
Yarborough BJH, Stumbo SP, McCarty D, et al. (2016) Methadone, buprenorphine and preferences for opioid agonist treatment: a qualitative analysis. Drug Alcohol Depend 160:112–118. doi:10.1016/j.drugalcdep.2015.12.031
Gryczynski J, Jaffe JH, Schwartz RP, et al. (2013) Patient perspectives on choosing buprenorphine over methadone in an urban, equal-access system. Am J Addict 22:285–291
Cone EJ, Gorodetzky CW, Yousefnejad D, et al. (1984) The metabolism and excretion of buprenorphine in humans. Drug Metab Dispos 12:577–581
Elkader A, Sproule B (2005) Buprenorphine: clinical pharmacokinetics in the treatment of opioid dependence. Clin Pharmacokinet 44:661–680
Mendelson J, Upton RA, Everhart ET, et al. (1997) Bioavailability of sublingual buprenorphine. J Clin Pharmacol 37:31–37
Nath RP, Upton RA, Everhart ET, et al. (1999) Buprenorphine pharmacokinetics: relative bioavailability of sublingual tablet and liquid formulations. J Clin Pharmacol 39:619–623
Alhaddad H, Cisternino S, Declèves X, et al. (2012) Respiratory toxicity of buprenorphine results from the blockage of P-glycoprotein-mediated efflux of norbuprenorphine at the blood-brain barrier in mice. Crit Care Med 40:3215–3223
Hassan HE, Myers AL, Coop A, Eddington ND (2009) Differential involvement of P-glycoprotein (ABCB1) in permeability, tissue distribution, and antinociceptive activity of methadone, buprenorphine, and Diprenorphine: in vitro and in vivo evaluation. J Pharm Sci 98:4928–4940
Brown SM, Campbell SD, Crafford A, et al. (2012) P-glycoprotein is a major determinant of norbuprenorphine brain exposure and antinociception. J Pharmacol Exp Ther 343:53–61. doi:10.1124/jpet.112.193433
McAleer SD, Mills RJ, Polack T, et al. (2003) Pharmacokinetics of high-dose buprenorphine following single administration of sublingual tablet formulations in opioid naı̈ve healthy male volunteers under a naltrexone block. Drug Alcohol Depend 72:75–83
Ciraulo DA, Hitzemann RJ, Somoza E, et al. (2006) Pharmacokinetics and pharmacodynamics of multiple sublingual buprenorphine tablets in dose-escalation trials. J Clin Pharmacol 46:179–192
Bullingham RE, McQuay HJ, Dwyer D, et al. (1981) Sublingual buprenorphine used postoperatively: clinical observations and preliminary pharmacokinetic analysis. Br J Clin Pharmacol 12:117–122
Brown SM, Holtzman M, Kim T, Kharasch ED (2011) Buprenorphine metabolites, buprenorphine-3-glucuronide and norbuprenorphine-3-glucuronide, are biologically active. Anesthesiol 115:1251–1260
Iribarne C, Picart D, Dréano Y, et al. (1997) Involvement of cytochrome P450 3 A4 in N-dealkylation of buprenorphine in human liver microsomes. Life Sci 60:1953–1964
Kobayashi K, Yamamoto T, Chiba K, et al. (1998) Human buprenorphine N-dealkylation is catalyzed by cytochrome P450 3 A4. Drug Metab Dispos 26:818–821
Moody DE, Slawson MH, Strain EC, et al (2002) A liquid chromatographic-electrospray ionization-tandem mass spectrometric method for determination of buprenorphine, its metabolite, norbuprenorphine, and a coformulant, naloxone, that is suitable for in vivo and in vitro metabolism studies. Anal Biochem 306:31–39
Picard N, Cresteil T, Djebli N, Marquet P (2005) In vitro metabolism study of buprenorphine: evidence for new metabolic pathways. Drug Metab Dispos 33:689–695. doi:10.1124/dmd.105.003681
Brewster D, Humphrey MJ, McLeavy MA (1981) Biliary excretion, metabolism and enterohepatic circulation of buprenorphine. Xenobiotica 11:189–196
Niwa T, Shiraga T, Takagi A (2005) Effect of antifungal drugs on cytochrome P450 (CYP) 2C9, CYP2C19, and CYP3A4 activities in human liver microsomes. Biol Pharm Bull 28:1805–1808
Hagelberg NM, Nieminen TH, Saari TI, et al. (2009) Voriconazole drastically increases exposure to oral oxycodone. Eur J Clin Pharmacol 65:263–271
Saari TI, Laine K, Leino K, et al. (2006) Effect of voriconazole on the pharmacokinetics and pharmacodynamics of intravenous and oral midazolam. Clin Pharmacol Ther 79:362–370
Wexler D, Courtney R, Richards W, et al. (2004) Effect of posaconazole on cytochrome P450 enzymes: a randomized, open-label, two-way crossover study. Eur J Pharm Sci 21:645–653
Moody DE, Liu F, Fang WB (2015) Azole antifungal inhibition of buprenorphine, methadone and oxycodone in vitro metabolism. J Anal Toxicol 39:374–386
Li Y, Theuretzbacher U, Clancy CJ, et al. (2010) Pharmacokinetic/pharmacodynamic profile of posaconazole. Clin Pharmacokinet 49:379–396
Lempers VJC, van den Heuvel JJMW, Russel FGM, et al. (2016) Inhibitory potential of antifungal drugs on ATP-binding cassette transporters P-glycoprotein, MRP1 to MRP5, BCRP, and BSEP. Antimicrob Agents Chemother 60:3372–3379
McCance-Katz EF, Moody DE, Morse GD, et al. (2006) Interactions between buprenorphine and antiretrovirals. I. The nonnucleoside reverse-transcriptase inhibitors efavirenz and delavirdine. Clin Infect Dis 43(Suppl 4):S224–S234
McCance-Katz EF, Moody DE, Smith PF, et al. (2006) Interactions between buprenorphine and antiretrovirals. II. The protease inhibitors nelfinavir, lopinavir/ritonavir, and ritonavir. Clin Infect Dis 43(Suppl 4):S235–S246
McCance-Katz EF, Moody DE, Morse GD, et al. (2007) Interaction between buprenorphine and atazanavir or atazanavir/ritonavir. Drug Alcohol Depend 91:269–278
Hulskotte EGJ, Bruce RD, Feng H-P, et al. (2015) Pharmacokinetic interaction between HCV protease inhibitor boceprevir and methadone or buprenorphine in subjects on stable maintenance therapy. Eur J Clin Pharmacol 71:303–311
Nieminen TH, Hagelberg NM, Saari TI, et al. (2009) Rifampin greatly reduces the plasma concentrations of intravenous and oral oxycodone. Anesthesiol 110:1371–1378
Saari TI, Grönlund J, Hagelberg NM, et al. (2010) Effects of itraconazole on the pharmacokinetics and pharmacodynamics of intravenously and orally administered oxycodone. Eur J Clin Pharmacol 66:387–397
Michna E, Ross EL, Hynes WL, et al. (2004) Predicting aberrant drug behavior in patients treated for chronic pain: importance of abuse history. J Pain Symptom Manag 28:250–258
Hannington-Kiff JG (1970) Measurement of recovery from outpatient general anaesthesia with a simple ocular test. Br Med J 3:132–135
Cogan D (1941) A simplified entoptic pupillometer. Am J Ophthalmol 24:1431–1433
Stone BM (1984) Pencil and paper tests--sensitivity to psychotropic drugs. Br J Clin Pharmacol 18(Suppl 1):15S–20S
R Foundation for Statistical Computing (2015) R: a language and environment for statistical computing
Tournier N, Chevillard L, Megarbane B, et al. (2010) Interaction of drugs of abuse and maintenance treatments with human P-glycoprotein (ABCB1) and breast cancer resistance protein (ABCG2). Int J Neuropsychopharmacol 13:905–915
Krishna G, Moton A, Ma L, et al. (2009) Effects of oral posaconazole on the pharmacokinetic properties of oral and intravenous midazolam: a phase I, randomized, open-label, crossover study in healthy volunteers. Clin Ther 31:286–298
Bruce RD, Altice FL, Moody DE, et al. (2010) Pharmacokinetic interactions between buprenorphine/naloxone and once-daily lopinavir/ritonavir. J Acquir Immune Defic Syndr 54:511–514
DuBuske LM (2005) The role of P-glycoprotein and organic anion-transporting polypeptides in drug interactions. Drug Saf 28:789–801
Kharasch ED, Hoffer C, Whittington D, Sheffels P (2003) Role of P-glycoprotein in the intestinal absorption and clinical effects of morphine. Clin Pharmacol Ther 74:543–554
Kharasch ED, Hoffer C, Altuntas TG, Whittington D (2004) Quinidine as a probe for the role of p-glycoprotein in the intestinal absorption and clinical effects of fentanyl. J Clin Pharmacol 44:224–233
Kalvass JC, Olson ER, Pollack GM (2007) Pharmacokinetics and pharmacodynamics of alfentanil in P-glycoprotein-competent and P-glycoprotein-deficient mice: P-glycoprotein efflux alters alfentanil brain disposition and antinociception. Drug Metab Dispos 35:455–459
Sansone-Parsons A, Krishna G, Simon J, et al. (2007) Effects of age, gender, and race/ethnicity on the pharmacokinetics of posaconazole in healthy volunteers. Antimicrob Agents Chemother 51:495–502
Ashbee HR, Gilleece MH (2012) Has the era of individualised medicine arrived for antifungals? A review of antifungal pharmacogenomics. Bone Marrow Transplant 47:881–894
Saari TI, Laine K, Leino K, et al. (2006) Effect of voriconazole on the pharmacokinetics and pharmacodynamics of zolpidem in healthy subjects. Br J Clin Pharmacol 63:116–120
Saari TI, Laine K, Bertilsson L, et al. (2007) Voriconazole and fluconazole increase the exposure to oral diazepam. Eur J Clin Pharmacol 63:941–949
Saari TI, Laine K, Leino K, et al. (2006) Voriconazole, but not terbinafine, markedly reduces alfentanil clearance and prolongs its half-life. Clin Pharmacol Ther 80:502–508
Saari TI, Laine K, Neuvonen M, et al. (2008) Effect of voriconazole and fluconazole on the pharmacokinetics of intravenous fentanyl. Eur J Clin Pharmacol 64:25–30
Liu P, Foster G, Labadie R, et al. (2007) Pharmacokinetic interaction between voriconazole and methadone at steady state in patients on methadone therapy. Antimicrob Agents Chemother 51:110–118
Olkkola KT, Backman JT, Neuvonen PJ (1994) Midazolam should be avoided in patients receiving the systemic antimycotics ketoconazole or itraconazole. Clin Pharmacol Ther 55:481–485
Olkkola KT, Ahonen J, Neuvonen PJ (1996) The effects of the systemic antimycotics, itraconazole and fluconazole, on the pharmacokinetics and pharmacodynamics of intravenous and oral midazolam. Anesth Analg 82:511–516
Bullingham RE, McQuay HJ, Moore A, Bennett MR (1980) Buprenorphine kinetics. Clin Pharmacol Ther 28:667–672
Kuhlman JJ, Lalani S, Magluilo J, et al. (1996) Human pharmacokinetics of intravenous, sublingual, and buccal buprenorphine. J Anal Toxicol 20:369–378
Kuhlman JJ, Levine B, Johnson RE, et al. (1998) Relationship of plasma buprenorphine and norbuprenorphine to withdrawal symptoms during dose induction, maintenance and withdrawal from sublingual buprenorphine. Addiction 93:549–559
Kaukonen KM, Olkkola KT, Neuvonen PJ (1997) Itraconazole increases plasma concentrations of quinidine. Clin Pharmacol Ther 62:510–517
Varhe A, Olkkola KT, Neuvonen PJ (1994) Oral triazolam is potentially hazardous to patients receiving systemic antimycotics ketoconazole or itraconazole. Clin Pharmacol Ther 56:601–607
Backman JT, Kivistö KT, Olkkola KT, Neuvonen PJ (1998) The area under the plasma concentration-time curve for oral midazolam is 400-fold larger during treatment with itraconazole than with rifampicin. Eur J Clin Pharmacol 54:53–58
Granfors MT, Backman JT, Neuvonen M, et al. (2004) Fluvoxamine drastically increases concentrations and effects of tizanidine: a potentially hazardous interaction. Clin Pharmacol Ther 75:331–341
Kapil RP, Cipriano A, Michels GH, et al. (2012) Effect of ketoconazole on the pharmacokinetic profile of buprenorphine following administration of a once-weekly buprenorphine transdermal system. Clin Drug Investig 32:583–592
Acknowledgments
We thank Mrs. Elina Kahra (medical laboratory technologist, Clinical Pharmacology, TYKSLAB, Hospital District of Southwest Finland, Turku, Finland) for her skillful technical assistance. This study was supported financially by Turku University Hospital research fund (EVO 13821), Turku, Finland.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Authors’ contributions
Mari Fihlman took care of the clinical phase of the study and data collection, participated in data analysis and statistical analysis and wrote the manuscript. Klaus Olkkola and Kari Laine designed the study, wrote the protocol, supervised and coordinated the clinical implementation of the study, and participated in data analysis. Tuija Hemmilä and Kristiina Kuusniemi participated the clinical phase and data collection. Janne T. Backman, Jouko Laitila and Pertti J Neuvonen performed the analytical assays. Teijo Saari designed the study, analyzed the data, performed statistical analysis, and wrote the manuscript. All authors materially participated in the research and/or manuscript preparation. All authors have contributed to and approved the final manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Fihlman, M., Hemmilä, T., Hagelberg, N.M. et al. Voriconazole more likely than posaconazole increases plasma exposure to sublingual buprenorphine causing a risk of a clinically important interaction. Eur J Clin Pharmacol 72, 1363–1371 (2016). https://doi.org/10.1007/s00228-016-2109-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-016-2109-y