Abstract
Purpose
The purpose of this study was to compare the impact of initial antihypertensive therapy including angiotensin converting enzyme inhibitors (ACE) or angiotensin II receptor blockers (ARB) on long-term persistence to therapy.
Methods
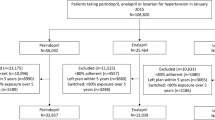
A retrospective cohort study using prescription claims data from the Australian Pharmaceutical Benefit Scheme (PBS). Kaplan-Meier analysis of prescription refills and cox proportional hazard models were used to compare the time on therapy (persistence) in people newly initiated to monotherapy or combination therapy including ACE or ARB, between April 2007 and March 2008. Differences in persistence to initial drug class or any antihypertensive therapy were reported at 4-year follow-up.
Results
About 119,500 persons initiated ACE or ARB: 47 % initiated ACE monotherapy; 32 % ARB monotherapy; 13 % ACE combinations; and 8 % ARB combinations. Persistence (% on treatment at 4 years) to index therapy was lower in people starting ACE and ARB combinations compared to ACE or ARB monotherapies: ACE combination (12 %) versus ACE monotherapy (25 %) and ARB combinations (22 %) versus ARB monotherapy (35 %). Persistence was higher in those initiating fixed dose combinations (FDC) versus separate pill combinations of ACEs (19 vs. 10 %) and ARBs (25 vs. 14 %). Persistence at 4 years to any antihypertensive therapy was similar between initiators to ACE or ARB monotherapy (60 and 61 %, p = 0.08), ACE or ARB combinations (56 %, p = 0.99), and was slightly higher for separate pill combinations (57–59 %) versus FDC (55 %).
Conclusion
Choice of initial antihypertensive may have little impact on long-term persistence to therapy.
Similar content being viewed by others
References
Staessen JA, Wang JG, Thijs L (2003) Cardiovascular prevention and blood pressure reduction: a quantitative overview updated until 1 March 2003. J Hypertens 21(6):1055–1076
Staessen JA, Li Y, Thijs L, Wang JG (2005) Blood pressure reduction and cardiovascular prevention: An update including the 2003-2004 secondary prevention trials. Hypertens Res 28(5):385–407
Cramer JA, Benedict A, Muszbek N, Keskinaslan A, Khan ZM (2008) The significance of compliance and persistence in the treatment of diabetes, hypertension and dyslipidaemia: a review. Int J Clin Pract 62(1):76–87
Cramer JA, Roy A, Burrell A, Fairchild CJ, Fuldeore MJ, Ollendorf DA, et al. (2008) Medication compliance and persistence: terminology and definitions. Value Health 11(1):44–47
McCombs JS, Nichol MB, Newman CM, Sclar DA (1994) The costs of interrupting antihypertensive drug therapy in a Medicaid population. Med Care 32(3):214–226
Degli Esposti E, Sturani A, Di Martino M, Falasca P, Novi MV, Baio G, et al. (2002) Long-term persistence with antihypertensive drugs in new patients. J Hum Hypertens 16(6):439–444
Conlin PR, Gerth WC, Fox J, Roehm JB, Boccuzzi SJ (2001) Four-year persistence patterns among patients initiating therapy with the angiotensin II receptor antagonist losartan versus other antihypertensive drug classes. Clin Ther 23(12):1999–2010
Australian Institute of Health and Welfare. Medicines for cardiovascular health: are they used appropriately? Cardiovascular disease series no 27. AIHW; 2007;Cat no 36.
Simons LA, Ortiz M, Calcino G (2008) Persistence with antihypertensive medication: Australia-wide experience, 2004-2006. Med J Aust 188(4):224–227
Caro JJ, Speckman JL, Salas M, Raggio G, Jackson JD (1999) Effect of initial drug choice on persistence with antihypertensive therapy: the importance of actual practice data. Can Med Assoc J 160(1):41–46
Marentette MA, Gerth WC, Billings DK, Zarnke KB (2002) Antihypertensive persistence and drug class. Can J Cardiol 18(6):649–656
Baggarly SA, Kemp RJ, Wang XJ, Magoun AD (2014) Factors associated with medication adherence and persistence of treatment for hypertension in a Medicaid population. Res Soc Adm Pharm 10(6):E99–E112
Elliott WJ, Plauschinat CA, Skrepnek GH, Gause D (2007) Persistence, adherence, and risk of discontinuation associated with commonly prescribed antihypertensive drug monotherapies. J Am Board Fam Med 20(1):72–80
Grégoire J-P, Moisan J, Guibert R, Ciampi A, Milot A (2006) Predictors of self-reported noncompliance with antihypertensive drug treatment: a prospective cohort study. Can J Cardiol 22(4):323–329
Gupta AK, Arshad S, Poulter NR (2010) Compliance, safety, and effectiveness of fixed-dose combinations of antihypertensive agents: a meta-analysis. Hypertension 55(2):399–407
Simons LA, Ortiz M, Calcino G (2011) Persistence with a single pill versus two pills of amlodipine and atorvastatin: the Australian experience, 2006-2010. Med J Aust 195(3):134–137
Mancia G, De Backer G, Dominiczak A, Cifkova R, Fagard R, Germano G, et al. (2007) 2007 Guidelines for the Management of Arterial Hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens 25(6):1105–1187
Mancia G, Fagard R, Narkiewicz K, Redon J, Zanchetti A, Bohm M, et al. (2013) 2013 ESH/ESC Guidelines for the management of arterial hypertension. Eur Heart J 34(28):2159
Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, et al. (2003) The seventh report of the Joint National Committee on prevention, detection, evaluation, and treatment of high blood pressure: the JNC 7 report. J Am Med Assoc 289(19):2560–2572
National Heart Foundation of Australia (National Blood Pressure and Vascular Disease Advisory Committee) Guide to management of hypertension 2008. Updated 2010. 2010.
Department of Health. About the Pharmaceutical Benefits Scheme Canberra, Australia Commonwealth Government of Australia; 2015 [cited 2015 12 December 2015] Available from: http://www.pbs.gov.au/info/about-the-pbs.
Paige E, Kemp-Casey A, Korda R, Banks E. Using Australian Pharmaceutical Benefits Scheme data for pharmacoepidemiological research: challenges and approaches. Public Health Res Pract. 25(4):e2541546.
Hall KB, Tett SE, Nissen LM (2006) Perceptions of the influence of prescription medicine samples on prescribing by family physicians. Med Care 44(4):383–387
Department of Health. Pharmaceutical Benefits Scheme Information For Prescribers Canberra, Australia: Commonwealth Government of Australia; 2015 [cited 2015 12 December] Available from: http://www.pbs.gov.au/info/healthpro/explanatory-notes/section1/Section_1_2_Explanatory_Notes#Preparing-general-PBS-prescriptions.
Vitry A, Wong SA, Roughead EE, Ramsay E, Barratt J (2009) Validity of medication-based co-morbidity indices in the Australian elderly population. Aust N Z J Public Health 33(2):126–130
Grimmsmann T, Himmel W (2014) Persistence of antihypertensive drug use in German primary care: a follow-up study based on pharmacy claims data. Eur J Clin Pharmacol 70(3):295–301
Powers BG, L.M LB (2011) A summary of AHRQs comparative effectiveness review of ACEs, ARBs and direct renin inhibitors. J Manag Care Pharm 17(8):3–14
Messerli FH, Williams B, Ritz E (2007) Essential hypertension. Lancet 370(9587):591–603
Kamaruzzaman S, Watt H, Carson C, Ebrahim S (2010) The association between orthostatic hypotension and medication use in the British Women's Heart and Health Study. Age Ageing 39(1):51–56
Poon IO, Braun U (2005) High prevalence of orthostatic hypotension and its correlation with potentially causative medications among elderly veterans. J Clin Pharm Ther 30(2):173–178
National Prescribing Service (2007) Managing hypertension as a cardiovascular risk factor. National Prescribing Service Limited, Strawberry Hills
National Prescribing Service. Olmesartan medoxomil (Olmetec) monotherapy or in combination with hydrochlorothiazide (Olmetec Plus) for hypertension: National Prescribing Service Limited; 2007.
National Prescribing Service. Be aware of dose ranges for new ramipril with felodipine combination (Triasyn): National Prescribing Service Limited; 2007.
National Prescribing Service. Valsartan (Diovan) and combinations with hydrochlorothiazide (Co-Diovan) or amlodipine (Exforge): National Prescribing Service Limited; 2009.
Rosenthal T, Gavras I (2006) Fixed-drug combinations as first-line treatment for hypertension. Prog Cardiovasc Dis 48(6):416–425
Gadzhanova S, Ilomaki J, Roughead EE (2013) Antihypertensive use before and after initiation of fixed-dose combination products in Australia: a retrospective study. Int J Clin Pharm 35(4):613–620
World Health Organization Collaborating Centre for Drug Statistics Methodology. Anatomical Therapeutic Chemical Code Classification index with Defined Daily Doses.
Department of Health. The Pharmaceutical Benefits Schedule Canberra, Australia: Commonwealth Governent; [cited 2015]. Available from: http://www.pbs.gov.au/pbs/home.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare there is no conflict of interest.
Funding
S.G. had funding support from the Australian Government Department of Health. L.B. is a part-time employee of the Commonwealth Department of Health. There are no other financial relationships with any organisations that might have an interest in the submitted work in the previous 3 years and no other relationships or activities that could appear to have influenced the submitted work.
Electronic supplementary material
ESM 1
(DOCX 47.9 kb)
Rights and permissions
About this article
Cite this article
Gadzhanova, S., Roughead, E.E. & Bartlett, L.E. Long-term persistence to mono and combination therapies with angiotensin converting enzymes and angiotensin II receptor blockers in Australia. Eur J Clin Pharmacol 72, 765–771 (2016). https://doi.org/10.1007/s00228-016-2037-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-016-2037-x