Abstract
Purpose
Triflusal is an antiplatelet agent that irreversibly acetylates cyclooxygenase isoform 1 (COX-1) and therefore inhibits thromboxane biosynthesis. It was initially marketed as capsules containing 300 mg of active substance. In 2006 a new 600 mg (10 ml) oral solution form of triflusal was authorized in Spain. The primary aim of this study was to compare the gastrointestinal safety of the new triflusal oral solution with triflusal capsules in healthy volunteers.
Methods
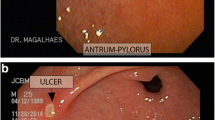
Sixty healthy subjects were randomly assigned, in a 2.5:2.5: 1 ratio, into one of three groups, with 25 subjects receiving one bottle of triflusal oral solution (600 mg) daily, 25 subjects receiving two triflusal capsules (600 mg) once daily, and ten subjects receiving two placebo capsules once daily, respectively, during 7 consecutive days. Gastroscopy was performed at baseline before the administration of study drugs and after 4–8 h of the last dose of study drugs. Effects on the esophagus, stomach, and duodenum were measured in accordance with a modified Lanza scale.
Results
No differences between groups were detected at baseline. After treatment, median global scores in the placebo, triflusal solution, and triflusal capsules groups were, respectively, 0, 1, and 3 (p = 0.003 for comparison between placebo and triflusal capsules and p = 0.042 for comparison between triflusal solution and triflusal capsules). There were no significant differences between the scores on the triflusal solution and placebo groups. All treatments were well tolerated.
Conclusion
In healthy subjects, triflusal solution induced less endoscopically apparent gastrointestinal mucosal damage than triflusal capsules and did not induce more damage than the placebo in healthy volunteers.

Similar content being viewed by others

References
Cruz-Fernández JM (2003) Antiplatelet drugs in the treatment of acute coronary syndromes: Focus on cyclooxigenase inhibitors. Eur Heart J 3[Suppl I]:123–130
Murdoch D, Plosker GL (2006) Triflusal: a review of its use in cerebral infarction and myocardial infarction, and as thromboprohylaxis in atrial fibrillation. Drugs 66:671–692
García-Rafanell J, Ramis J, Gómez L, Forn J (1986) Effect of triflusal and other salicylic acid derivatives on cyclic AMP levels in rat platelets. Arch Int Pharmacodyn Ther 284:155–165
Cruz-Fernández JM, López-Bescos L, García-Dorado D, López García-Aranda V, Cabadés A, Martín-Jadraque L et al (2000) Randomized comparative trial of Triflusal and aspirin following acute myocardial infarction. Eur Heart J 21:457–465
Matias-Guiu J, Ferro JM, Álvarez-Sabin J, Torres F, Dolores Jiménez M, Lago A et al (2003) Comparison of Triflusal and aspirin for prevention of vascular events in patients after cerebral infarction: the TACIP Study: a randomized, double-blind, multicenter trial. Stroke 34:840–848
Culebras A, Rotta-Escalante R, Vila J, Domínguez R, Abiusi G, Famulari A et al (2004) (2004) Triflusal vs aspirin for prevention of cerebral infarction. Neurology 62:1073–1080
Costa J, Ferro JM, Matias-Guiu J, Alvarez-Sabín J, Torres F (2005) Triflusal for preventing serious vascular events in people at high risk. Cochrane Database Syst Rev 3:CD004296
Plaza L, López-Bescós L, Martín-Jadraque L, Alegría E, Cruz-Fernández JM, Velasco J et al (1993) Protective effect of triflusal against acute myocardial infarction in patients with instable angina: results of a Spanish multicenter trial. Cardiology 82:388–398
Guiteras P, Altimiras J, Arís A, Mª AJ, Bassons T, Bonal J et al (1989) Prevention of aortocoronary vein-graft attrition with low-dose aspirin and triflusal, both associated with dipyridamole: a randomized, double-blind, placebo-controlled trial. Eur Heart J 10:159–167
Aramendi JI, Mestres CA, Martínez-León J, Campos V, Muñoz G, Navas C (2005) Triflusal versus oral anticoagulation for primary prevention of thromboembolism after bioprosthetic valve replacement (trac): prospective, randomized, co-operative trial. Eur J Cardio Thorac Surg 27:854–860
Auteri A, Angaroni A, Borgatti E, Catalano M, De Vizzi GB, Forconi S et al (1995) Triflusal in the treatment of patients with chronic peripheral arteriopathy: multicentre double-blind clinical study vs placebo. Int J Clin Pharmacol Res 15:57–63
Pérez-Gómez F, Alegría E, Berjón J, Iriarte JA, Zumalde J, Salvador A et al (2004) Comparative effects of antiplatelet, anticoagulant, or combined therapy in patients with valvular and nonvalvular atrial fibrillation. J Am Coll Cardiol 44:1557–1566
Fraj J, Valero A, Vives R, Pérez I, Borja J, Izquierdo et al (2008) Safety of triflusal (antiplatelet drug) in patients with aspirin-exacerbated respiratory diseases. Allergy 63:112–115
Lanas A, Serrano P, Bajador E, Fuentes J, Sáinz R (2003) Risk of upper gastrointestinal bleeding associated with non-aspirin cardiovascular drugs, analgesics and nonsteroidal anti-inflammatory drugs. Eur J Gastroenterol Hepatol 15:173–178
Ibáñez L, Vidal X, Vendrell L, Moretti U, Laporte JR (2006) Upper gastrointestinal bleeding associated with antiplatelet drugs. Aliment Pharmacol Ther 23:235–242
Izquierdo I, Borja J, Rovira R, Pelagio P, Torres F, Cebrecos J et al (2010) Comparative bioavailability study of triflusal oral solution vs. triflusal capsules in healthy subjects. Arzneimittelforschung 60:36–41
Lanza FL, Roger GL, Nelson RS (1980) Endoscopic evaluation of the effects of aspirin, buffered aspirin, and enteric-coated aspirin on gastric and duodenal mucusa. N Engl J Med 303:136–138
Meyboom RHB, Hekster YA, Egberts ACG, Gribnau FWJ, Edwards IR (1997) Causal or casual? The role of causality assessment in pharmacovigilance. Drug Saf 17:374–389
Fiorucci S, Santucci L, Gresele P (2003) Faccino R M, Del Soldato P, Morelli A (2003) Gastrointestinal safety of NO-aspirin (NCX-4016) in healthy human volunteers: A proof of concept endoscopic study. Gastroenterology 124:600–607
Riambau-Alonso V, Fernández S, Lara M, Caballero MD, Rubio E, Martín A (2009) Estudio epidemiológico observacional para evaluar el manejo de los trastornos gastrointestinales en el paciente con tratamiento antiagregante. Angiologia 61:185–194
Patrono C, Baigent C, Hirsh J, Roth G (2008) Antiplatelet drugs. American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th edn). Chest 133:199S–233S
CAPRIE Steering Committee (1996) A randomised, blinded, trial of clopiodgrel versus aspirin in patients at risk of ischaemic events (CAPRIE). Lancet 348:1329–1339
Anderson JL, Adams CD, Antman EM, Bridges ChR, Califf RM, Casey DE et al (2007) ACC/AHA 2007 Guidelines for the Management of Patients With Unstable Angina/Non ST-Elevation Myocardial Infarction. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients With Unstable Angina/Non ST-Elevation Myocardial Infarction). Circulation 116:e148–e304
Bhatt DL, Scheiman J, Abraham NS, Antman EM, Chan FKL, Furberg CD (2008) ACCF/ACG/AHA 2008 Expert Consensus Document on Reducing the Gastrointestinal Risks of Antiplatelet Therapy and NSAID Use. A Report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents. Circulation 118:1894–1909
Gilard M, Arnaud B, Le Gal G, Abgrall JF, Boschat J (2006) Influence of omeprazol on the antiplatelet action of clopidogrel associated to aspirin. J Thromb Haemost 4:2508–2509
Ho PM, Maddox TM, Wang L, Fihn SD, Jesse RL, Peterson ED et al (2009) Risk of adverse outcomes associated with concomitant use of clopidogrel and proton pump inhibibitors following acute coronary syndrome. JAMA 301:937–944
Adams HP Jr, del Zoppo G, Alberts MJ, Bhatt DL, Brass L, Furlan A et al (2007) Guidelines for the early management of adults with ischemic stroke. Stroke 38:1655–1711
Finiels H, Struble D, Jaquot JM (2001) Les troubles de la déglutition du sujet âgé. Presse Méd 30:1623–1634
Hotfiezer JW, Silvoso GR, Burks M, Ivey KJ (1980) Comparison of the effects of regular and enteric-coated aspirin on gastroduodenal mucosa on man. Lancet 2:609–612
Lanza F (1980) L, Royer GL Jr, Nelson R S. Endoscopic evaluation of the effects of aspirin, buffered aspirin, and enteric-coated aspirin on gastric and duodenal mucosa. N Engl J Med 303:136–138
Hawthorne AB, Mahida YR, Cole AT, Hawkey CJ (1991) Aspirin-induced gastric mucosa damage: prevention by enteric-coating and relation to prostaglandin synthesis. Br J Clin Pharmacol 32:77–83
Walker J, Robinson J, Stewart J, Jacob S (2007) Does enteric-coated aspirin result in a lower incidence of gastrointestinal complications compared to normal aspirin? Interact Cardiovasc Thorac Surg 6:519–522
Acknowledgments
The authors thank J. Uriach y Compañía, S.A. (Palau-solità I Plegamans, Barcelona, Spain) for financial support to this study, and to Montserrat Puntes, Joan Martínez, and Salvador Rico for their assistance during data collection. This study was partially supported by the National Scientific Research Program of the Spanish Ministry of Science and Technology.
Conflict of interest
Iñaki Izquierdo, Javier Borja, and Iris Blanch are employees of J. Uriach y Compañía, S.A. Esther Donado was an employee of J. Uriach y Compañía, S.A. when the study was performed. Manel Barbanoj has been an external advisor of J. Uriach y Compañía, S.A.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Antonijoan, R.M., Gich, I., Azaro, A. et al. Gastrointestinal safety of triflusal solution in healthy volunteers: a proof of concept endoscopic study. Eur J Clin Pharmacol 67, 663–669 (2011). https://doi.org/10.1007/s00228-011-1004-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-011-1004-9