Abstract
Objective
It has been reported that leflunomide and its active metabolite, A771726, are substrates of the ABCG2 (BCRP) transporter in vitro. Recent genome-wide association studies have shown that ABCG2 transporter modulates serum uric acid (UA) levels. We explored the role of ABCG2 genotypes in the pharmacokinetics of A771726 and the relationship between serum UA levels and pharmacokinetics of A771726 in healthy participants.
Methods
Twenty-four healthy individuals were recruited and genotyped for ABCG2. After administration of a single dose of 20 mg leflunomide, plasma concentrations of A771726 were measured. Serum UA levels were measured just before medication, and ABCG2 c.421C>A and c.34G> A polymorphism were genotyped.
Results
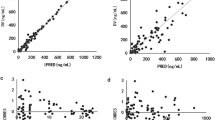
ABCG2 c.421C>A but not c.34G>A substantially influenced the pharmacokinetics of A771726. A771726 Cmax was 30% higher, area under the concentration-time curve (AUC) 83% larger, and oral clearance (CL/F) 41% lower in c.421C>A carriers than in noncarriers. Serum UA levels were also higher in carriers than in noncarriers and exhibited a strong and positive correlation with A771726 AUC (Spearman r = 0.6746, P = 0.0003), but a negative correlation was observed with A771726 CL/F (Spearman r = −0.6616, P = 0.0004).
Conclusion
ABCG2 c.421C>A but not c.34G>A polymorphism appears to be a major determinant of interindividual variability in A771726 disposition. Additionally, serum UA levels exhibited a strong correlation with exposure to A771726.


Similar content being viewed by others
References
Litman T, Druley TE, Stein WD, Bates SE (2001) From MDR to MXR: new understanding of multidrug resistance systems, their properties and clinical significance. Cell Mol Life Sci 58:931–959
Jonker JW, Smit JW, Brinkhuis RF, Maliepaard M, Beijnen JH, Schellens JH et al (2000) Role of breast cancer resistance protein in the bioavailability and fetal penetration of topotecan. J Natl Cancer Inst 92:1651–1656
Robey RW, Medina-Perez WY, Nishiyama K, Lahusen T, Miyake K, Litman T et al (2001) Overexpression of the ATP-binding cassette half-transporter, ABCG2 (Mxr/BCrp/ABCP1), in flavopiridol-resistant human breast cancer cells. Clin Cancer Res 7:145–152
Zamber CP, Lamba JK, Yasuda K, Farnum J, Thummel K, Schuetz JD et al (2003) Natural allelic variants of breast cancer resistance protein (BCRP) and their relationship to BCRP expression in human intestine. Pharmacogenetics 13:19–28
Imai Y, Nakane M, Kage K, Tsukahara S, Ishikawa E, Tsuruo T et al (2002) C421A polymorphism in the human breast cancer resistance protein gene is associated with low expression of Q141K protein and low-level drug resistance. Mol Cancer Ther 1:611–616
Kondo C, Suzuki H, Itoda M, Ozawa S, Sawada J, Kobayashi D et al (2004) Functional analysis of SNPs variants of BCRP/ABCG2. Pharm Res 21:1895–1903
de Jong FA, Marsh S, Mathijssen RH, King C, Verweij J, Sparreboom A et al (2004) ABCG2 pharmacogenetics: ethnic differences in allele frequency and assessment of influence on irinotecan disposition. Clin Cancer Res 10:5889–5894
Breedveld P, Zelcer N, Pluim D, Sonmezer O, Tibben MM, Beijnen JH et al (2004) Mechanism of the pharmacokinetic interaction between methotrexate and benzimidazoles: potential role for breast cancer resistance protein in clinical drug-drug interactions. Cancer Res 64:5804–5811
Cusatis G, Gregorc V, Li J, Spreafico A, Ingersoll RG, Verweij J et al (2006) Pharmacogenetics of ABCG2 and adverse reactions to gefitinib. J Natl Cancer Inst 98:1739–1742
Sparreboom A, Loos WJ, Burger H, Sissung TM, Verweij J, Figg WD et al (2005) Effect of ABCG2 genotype on the oral bioavailability of topotecan. Cancer Biol Ther 4:650–658
Dehghan A, Kottgen A, Yang Q, Hwang SJ, Kao WL, Rivadeneira F et al (2008) Association of three genetic loci with uric acid concentration and risk of gout: a genome-wide association study. Lancet 372:1953–1961
Kolz M, Johnson T, Sanna S, Teumer A, Vitart V, Perola M et al (2009) Meta-analysis of 28, 141 individuals identifies common variants within five new loci that influence uric acid concentrations. PLoS Genet 5:e1000504
Goldenberg MM (1999) Leflunomide, a novel immunomodulator for the treatment of active rheumatoid arthritis. Clin Ther 21:1837–1852, discussion 1821
Kale VP, Bichile LS (2004) Leflunomide: a novel disease modifying anti-rheumatic drug. J Postgrad Med 50:154–157
Kis E, Nagy T, Jani M, Molnar E, Janossy J, Ujhellyi O et al (2009) Leflunomide and its metabolite A771726 are high affinity substrates of BCRP: implications for drug resistance. Ann Rheum Dis 68:1201–1207
Li J, Yao HW, Jin Y, Zhang YF, Li CY, Li YH et al (2002) Pharmacokinetics of leflunomide in Chinese healthy volunteers. Acta Pharmacol Sin 23:551–555
Park JY, Kim KA, Lee YH, Park SW, Lee GH, Ryu JH (2010) Pharmacokinetic comparison and bioequivalence of two leflunomide formulations in humans: a single dose, randomized, open-label, two-way crossover study. Int J Clin Pharmacol Ther 48:291–295
Kim KA, Park PW, Hong SJ, Park JY (2008) The effect of CYP2C19 polymorphism on the pharmacokinetics and pharmacodynamics of clopidogrel: a possible mechanism for clopidogrel resistance. Clin Pharmacol Ther 84:236–242
Park JY, Kim KA, Joo HJ (2009) Multiplex pyrosequencing analysis of the most common ABCG2 polymorphisms, 34G>A and 421C>A in a Korean population. J Clin Pharm Ther (Submitted)
Herrmann ML, Schleyerbach R, Kirschbaum BJ (2000) Leflunomide: an immunomodulatory drug for the treatment of rheumatoid arthritis and other autoimmune diseases. Immunopharmacology 47:273–289
Silva Junior HT, Morris RE (1997) Leflunomide and malononitrilamides. Am J Med Sci 313:289–301
James DR, Price CP (1984) Problems associated with the measurement of uric acid using two enzyme-mediated reaction systems. Ann Clin Biochem 21(Pt 5):405–410
Rozman B (2002) Clinical pharmacokinetics of leflunomide. Clin Pharmacokinet 41:421–430
Kalgutkar AS, Nguyen HT, Vaz AD, Doan A, Dalvie DK, McLeod DG et al (2003) In vitro metabolism studies on the isoxazole ring scission in the anti-inflammatory agent lefluonomide to its active alpha-cyanoenol metabolite A771726: mechanistic similarities with the cytochrome P450-catalyzed dehydration of aldoximes. Drug Metab Dispos 31:1240–1250
Chan V, Charles BG, Tett SE (2005) Population pharmacokinetics and association between A77 1726 plasma concentrations and disease activity measures following administration of leflunomide to people with rheumatoid arthritis. Br J Clin Pharmacol 60:257–264
Honjo Y, Hrycyna CA, Yan QW, Medina-Perez WY, Robey RW, van de Laar A et al (2001) Acquired mutations in the MXR/BCRP/ABCP gene alter substrate specificity in MXR/BCRP/ABCP-overexpressing cells. Cancer Res 61:6635–6639
Mitomo H, Kato R, Ito A, Kasamatsu S, Ikegami Y, Kii I et al (2003) A functional study on polymorphism of the ATP-binding cassette transporter ABCG2: critical role of arginine-482 in methotrexate transport. Biochem J 373:767–774
Lee SS, Jeong HE, Yi JM, Jung HJ, Jang JE, Kim EY et al (2007) Identification and functional assessment of BCRP polymorphisms in a Korean population. Drug Metab Dispos 35:623–632
Sparreboom A, Gelderblom H, Marsh S, Ahluwalia R, Obach R, Principe P et al (2004) Diflomotecan pharmacokinetics in relation to ABCG2 421C>A genotype. Clin Pharmacol Ther 76:38–44
Yamasaki Y, Ieiri I, Kusuhara H, Sasaki T, Kimura M, Tabuchi H et al (2008) Pharmacogenetic characterization of sulfasalazine disposition based on NAT2 and ABCG2 (BCRP) gene polymorphisms in humans. Clin Pharmacol Ther 84:95–103
Mizuarai S, Aozasa N, Kotani H (2004) Single nucleotide polymorphisms result in impaired membrane localization and reduced atpase activity in multidrug transporter ABCG2. Int J Cancer 109:238–246
Tamura A, Onishi Y, An R, Koshiba S, Wakabayashi K, Hoshijima K et al (2007) In vitro evaluation of photosensitivity risk related to genetic polymorphisms of human ABC transporter ABCG2 and inhibition by drugs. Drug Metab Pharmacokinet 22:428–440
Woodward OM, Kottgen A, Coresh J, Boerwinkle E, Guggino WB, Kottgen M (2009) Identification of a urate transporter, ABCG2, with a common functional polymorphism causing gout. Proc Natl Acad Sci USA 106:10338–10342
Kunkel G, Cannon GW (2006) Leflunomide in the treatment of rheumatoid arthritis. Expert Rev Clin Immunol 2
Cannon GW, Holden WL, Juhaeri J, Dai W, Scarazzini L, Stang P (2004) Adverse events with disease modifying antirheumatic drugs (DMARD): a cohort study of leflunomide compared with other DMARD. J Rheumatol 31:1906–1911
Merino G, van Herwaarden AE, Wagenaar E, Jonker JW, Schinkel AH (2005) Sex-dependent expression and activity of the ATP-binding cassette transporter breast cancer resistance protein (BCRP/ABCG2) in liver. Mol Pharmacol 67:1765–1771
Bohanec Grabar P, Rozman B, Tomsic M, Suput D, Logar D, Dolzan V (2008) Genetic polymorphism of CYP1A2 and the toxicity of leflunomide treatment in rheumatoid arthritis patients. Eur J Clin Pharmacol 64:871–876
Bohanec Grabar P, Grabnar I, Rozman B, Logar D, Tomsic M, Suput D et al (2009) Investigation of the influence of CYP1A2 and CYP2C19 genetic polymorphism on 2-Cyano-3-hydroxy-N-[4-(trifluoromethyl)phenyl]-2-butenamide (A77 1726) pharmacokinetics in leflunomide-treated patients with rheumatoid arthritis. Drug Metab Dispos 37:2061–2068
Acknowledgements
This study was supported by a grant from the Korea Health 21 R & D Project, Ministry of Health & Welfare, Republic of Korea (AO30001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, KA., Joo, HJ. & Park, JY. Effect of ABCG2 genotypes on the pharmacokinetics of A771726, an active metabolite of prodrug leflunomide, and association of A771726 exposure with serum uric acid level. Eur J Clin Pharmacol 67, 129–134 (2011). https://doi.org/10.1007/s00228-010-0916-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-010-0916-0