Abstract
Objective
Both sirolimus and cyclosporine are immunosuppressants used in a combined regimen after organ transplantation. When coadministered with the innovator formulation of cyclosporine, sirolimus blood levels increase 3.3-fold due to a pharmacokinetic interaction. We assessed this drug interaction for potential differences when the innovator formulation is replaced by a generic cyclosporine.
Methods
In this randomized single-dose crossover study, 28 healthy subjects received 5 mg sirolimus oral solution with 250 mg cyclosporine soft gelatin capsules given as the innovator formulation (reference treatment) versus a generic formulation (test treatment). Sirolimus peak blood concentration (Cmax) and area under the concentration-time curve (AUC) were compared between test and reference treatments by standard bioequivalence testing.
Results
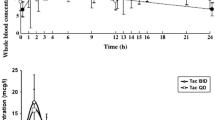
Sirolimus Cmax was significantly lower by 17% in the presence of generic versus innovator cyclosporine (p=0.0003) and failed bioequivalence criteria with a test/reference ratio of 0.83 (90% confidence interval, 0.77–0.90). Nearly half of the subjects (46%) had sirolimus Cmax changes which fell outside the bioequivalence window with individual Cmax decreases up to 52% and increases up to 39%. Sirolimus AUC was significantly lower by 11% in the presence of generic versus innovator cyclosporine (p=0.041) but satisfied average bioequivalence criteria with a test/reference ratio of 0.89 (0.83–0.95). Nonetheless, over a third of the subjects (43%) had sirolimus AUC changes outside the standard bioequivalence window with individual AUC decreases up to 39% and increases up to 42%.
Conclusions
Switching between innovator and generic cyclosporine may have a clinically-relevant impact on coadministered sirolimus pharmacokinetics. If such a switch is initiated by the prescriber, follow-up therapeutic monitoring of both cyclosporine and sirolimus blood levels should be performed to guide dose adjustments as necessary. If the switch is made without consulting the prescriber, potentially significant changes in sirolimus exposure could go unnoticed by the clinician and patient.

Similar content being viewed by others
References
Alloway RR, Isaacs R, Lake K, Hoyer P, First R, Helderman H, Bunnapradist S, Leichtman A, Bennett MW, Tejani A, Takemoto SK (2003) Report of the American Society of Transplantation conference on immunosuppressive drugs and the use of generic immunosuppressants. Am J Transplant 3:1211–1215
Barrett JS, Batra V, Chow A, Cook J, Gould AL, Heller AH, Lo MW, Patterson SD, Smith BP, Stritar JA, Vega JM, Zariffa N (2000) PhRMA perspective on population and individual bioequivalence. J Clin Pharmacol 40:561–570
Ford J, Woolfe J, Florence AT (1999) Nanospheres of cyclosporin A: poor oral absorption in dogs. Int J Pharmaceutics 183:3–6
Pollard S, Nashan B, Johnston A, Hoyer P, Belitsky P, Keown P, Helderman H (2003) Pharmacokinetic and clinical review of the potential clinical impact of using different formulations of cyclosporin A. Clin Ther 25:1654–1668
Rapamune (sirolimus), United States Product Label (2005)
Ritschel WA, Ritschel GB, Sabauni A, Wolochuk D, Schroeder T. Study on the peroral absorption of the endekapeptide cyclosporin A (1989) Method Find Exp Clin Pharmacol 11:281–287
Sabatini S, Ferguson RM, Helderman JH, Hull AR, Kirkpatrick BS, Barr WH (1999) Drug substitution in transplantation: a National Kidney Foundation white paper. Am J Kidney Dis 33:389–397
Shapiro R, Young JB, Milford EL (2005) Immunosuppression: evolution in practice and trends, 1993–2003. Am J Transplant 5:874–886
Zimmerman JJ, Harper D, Getsy J, Jusko WJ (2003) Pharmacokinetic interactions between sirolimus and microemulsion cyclosporine when orally administered jointly and 4 hours apart in healthy volunteers. J Clin Pharmacol 43:1168–1176
MacDonald A, Scarola J, Burke JT, Zimmermann JJ (2000) Clinical pharmacokinetics and therapeutic drug monitoring of sirolimus. Clin Ther 22(Suppl B):B101–B121
Acknowledgements
We thank P Hernandez, L Dillo, E Scheidegger, J Campestrini, and GJ Rivière, for conducting and evaluating the canine experiment and L McMahon and M Hayes for performing the bioanalytics. The experiments reported herein comply with the current laws of the countries in which they were performed inclusive of ethics approval.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kovarik, J.M., Noe, A., Wang, Y. et al. Differentiation of innovator versus generic cyclosporine via a drug interaction on sirolimus. Eur J Clin Pharmacol 62, 361–366 (2006). https://doi.org/10.1007/s00228-006-0109-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-006-0109-z