Abstract
Objective
To evaluate the main characteristics of case reports of arterial hypertension (AH) related to COX-2 inhibitor (coxib) use in real-life practice.
Methods
This study was based on spontaneous reports of adverse drug reactions (ADRs) submitted to the French Pharmacovigilance system. Associations between AH and the different groups of those using non-steroidal anti-inflammatory drugs (NSAIDs: rofecoxib, celecoxib and non-selective NSAIDs) were compared using calculation of the odds ratio (OR) with 95% confidence intervals (CIs).
Results
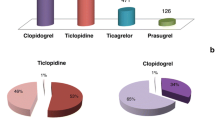
In France, between 1 April 2000 and 30 November 2003, 34 AH cases related to coxibs were reported. Case reports include predominantly patients older than 65 years, with a previous story of essential AH. Most AH (60%) occurred during the first 15 days of treatment. The AH was reported significantly more frequently for rofecoxib than celecoxib. The OR for development of AH with rofecoxib versus celecoxib was 3.3 (1.6–6.9). The AH was also reported more frequently with coxib (2.8%) than with non-selective NSAID (0.5%) use, OR = 5.9 (3.8–9.0).
Conclusion
This study shows that coxibs are associated with a risk of AH in real-life practice. More spontaneous reports of AH to the French Pharmacovigilance system concern rofecoxib than celecoxib (and coxibs than non-selective NSAIDs). This ADR is of special epidemiological importance due to both the risks of AH and the large use of coxibs.
Similar content being viewed by others
References
Pope JE, Anderson JJ, Felson DT (1993) A meta-analysis of the effects of non-steroidal anti-inflammatory drugs on blood pressure. Arch Intern Med 153:477–484
Jonhson AG, Nguyen TV, Day RO (1994) Do nonsteroidal anti-inflammatory drugs affect blood pressure? A meta-analysis. Ann Intern Med 121:289–300
Frishman W (2002) Effects of nonsteroidal anti-inflammatory drug therapy on blood pressure and peripheral edema. Am J Cardiol 89(Suppl):18D–25D
Amstrong E, Malone D (2003) The impact of non-steroidal anti-inflammatory drugs on blood pressure, with emphasis on newer agents. Clin Ther 25:1–18
Edwards I, Aronson J (2000) Adverse drug reactions: definitions, diagnosis, and management. Lancet 356:1255–1260
Bégaud B, Evreux JC, Jouglard J, Lagier G (1985) Imputation of the unexpected or toxic effects of drugs. Actualization of the method used in France. Thérapie 40:111–118
Whelton A, White W, Bello A, Puma J, Fort J, *the SUCCESS-VII Investigators (2002) Effects of celecoxib and rofecoxib on blood pressure and edema in patients ≥65 years of age with systemic hypertension and osteoarthritis. Am J Cardiol 90:959–963
Zhao S, Reynolds M, Lefkowitz J, Whelthon A, Arellano F (2001) A comparison of renal-related adverse drug reactions between rofecoxib and celecoxib, based on the World health Organisation/Uppsala Monitoring Centre Safety Database. Clin Ther 23:1478–1491
Solomon D, Schneeweiss S, Levin R, Avorn J (2004) Relationship between COX-2 specific inhibitors and hypertension. Hypertension 44:140–145
Wolfe F, Zhao S, Reynolds M, Petitt D (2004) Blood pressure destabilization and edema among 8538 users of celecoxib, rofecoxib, and nonselective nonsteroidal antiinflammatory drugs (NSAID) and nonusers of NSAID receiving ordinary clinical care. J Rheumatol 31:1143–11151
Brinker A, Goldkind L, Bonnel R, Beitz J (2004) Spontaneous reports of hypertension leading to hospitalisation in association with rofecoxib, celecoxib, nabumetone and oxaprozin. Drugs Aging 21:479–484
Mandani M, Juurlink D, Lee D, Rochon P, Koop A, Naglie G, Austin P (2004) Cyclo-oxygenase-2 inhibitors versus non-selective non-steroidal anti-inflammatory drugs and congestive heart failure outcomes in elderly patients: a population-based cohort study. Lancet 363:1751–1756
Griffin MR, Stein CM, Graham DJ, Daugherty JR, Arbogast PG, Ray WA (2004) High frequency of use of rofecoxib at greater than recommended doses: cause for concern. Pharmacoepidemiol Drug Saf 13:339–343
Bombardier C, Laine L, Reicin A et al (2000) Comparison of upper gastro-intestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis; VIGOR Study Group. N Engl J Med 343:1520–1528
Silverstein FE, Faich G, Golstein JL et al (2000) for the Celecoxib Long-term Arthritis Safety Study. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the class study: a randomized controlled trial. JAMA 284:1247–1255
Whelton A, Singh G, White W et al (2001) Celecoxib does not increase the risk of cardiac failure, edema, or hypertension compared to NSAIDs: results from SUCCESS 1, a double blind, randomized trial in 13 274 OA patients. In: Program and abstracts of the American College of Rheumatology 65th Annual Scientific Meeting; November 10–15, San Francisco, CA, Abstract
Riendeau D, Percival MD, Brideau C et al (2001) Etoricoxib (MK-0663): preclinical profile and comparison with other agents that selectively inhibit cyclooxygenase-2. J Pharmacol Exp Ther 296:558–566
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Durrieu, G., Olivier, P., Montastruc, J.L. et al. COX-2 inhibitors and arterial hypertension: an analysis of spontaneous case reports in the Pharmacovigilance database. Eur J Clin Pharmacol 61, 611–614 (2005). https://doi.org/10.1007/s00228-005-0964-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-005-0964-z