Abstract
Objective
This study analyzed the frequency of CYP2C9 variant alleles and evaluated the impact of CYP2C9 genotype on diclofenac metabolism in a Spanish population.
Methods
Diclofenac hydroxylation capacity was studied in a population of 102 healthy volunteers. After a single oral dose of 50 mg diclofenac the 0- to 8-h urinary concentrations of diclofenac and its main metabolites, 4'-hydroxy (OH), 3′-OH and 5-OH diclofenac were analyzed by high-performance liquid chromatography. CYP2C9 genotyping for the variant alleles CYP2C9*2 and *3 was carried out with PCR-RFLP.
Results
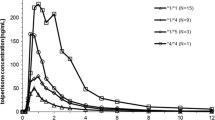
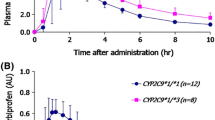
The frequencies of CYP2C9*1, *2, and *3 alleles were 0.74 (95%CI: 0.68–0.80), 0.16 (95%CI: 0.11–0.21) and 0.10 (95%CI: 0.06–0.15), respectively, among the 102 Spaniards studied. The diclofenac/4'-OH diclofenac urinary ratio, but not the diclofenac/3′-OH diclofenac and diclofenac/5-OH diclofenac ratios, was related to CYP2C9 genotype. The diclofenac/4'-OH ratio was significantly higher among subjects with CYP2C9*1/*3 (0.83±0.4, n=14, 95% CI for the difference: 0.02–0.4) and CYP2C9*2/*3 (1.10±0.5, n=4, 95% CI for the difference: 0.16–0.8) genotypes compared to CYP2C9*1/*1 (0.62±0.3, n=59) and approximately threefold higher (1.8) in the only subject homozygous for CYP2C9*3 variant.
Conclusions
The frequencies of CYP2C9*1, *2, and *3 alleles in the Spanish population reported here were similar to those found in the previously studied white European populations, and different of the previously reported in another Spanish population. CYP2C9*3 allele seems to influence the 4'-hydroxylation of diclofenac, although there is a large overlapping in the urinary metabolic ratio between the genotype groups studied


Similar content being viewed by others
References
Miners JO, Birkett DJ (1998) Cytochrome P4502C9: an enzyme of major importance in human drug metabolism. Br J Clin Pharmacol 45:525–538
Stubbins MJ, Harries LW, Smith G, Tarbit MH, Wolf CR (1996) Genetic analysis of the human cytochrome P450 CYP2C9 locus. Pharmacogenetics 6:429–439
Bhasker CR, Miners JO, Coulter S, Birkett DJ (1997) Allelic and functional variability of cytochrome P4502C9. Pharmacogenetics 7:51–58
Yasar Ü, Forslund-Bergengren C, Tybring G, Dorado P, LLerena A, Sjoqvist F, Eliasson E, Dahl ML (2002) Pharmacokinetics of losartan and its metabolite E-3174 in relation to the CYP2C9 genotype. Clin Pharmacol Ther 71:89–98
Veronese ME, Miners JO, Rees DL, Birkett DJ (1993) Tolbutamide hydroxylation in humans: lack of bimodality in 106 healthy subjects. Pharmacogenetics 3:86–93
Caraco Y, Muszkat M, Wood AJ (2001) Phenytoin metabolic ratio: a putative marker of CYP2C9 activity in vivo. Pharmacogenetics 11:587–596
Romkes M, Faletto MB, Blaisdell JA, Raucy JL, Goldstein JA (1993) Cloning and expression of complementary DNAs for multiple members of the human cytochrome P450IIC subfamily. Biochemistry 30:3247–3255 (erratum: 32:1390)
Sullivan-Klose TH, Ghanayem BI, Bell DA, Zhang ZY, Kaminsky LS, Shenfield GM, Miners JO, Birkett DJ, Goldstein JA (1996) The role of the CYP2C9-Leu359 allelic variant in the tolbutamide polymorphism. Pharmacogenetics 6:341–349
Crespi CL, Miller VP (1997) The R144C change in the CYP2C9*2 allele alters interaction of the cytochrome P450 with NADPH: cytochrome P450 oxidoreductase. Pharmacogenetics 7:203–210
Aithal GP, Day CP, Kesteven PJ, Daly AK (1999) Association of polymorphisms in the cytochrome P450 CYP2C9 with warfarin dose requirement and risk of bleeding complications. Lancet 353:717–719
van der Weide J, Steijns LS, van Weelden MJ, de Haan K (2001) The effect of genetic polymorphism of cytochrome P450 CYP2C9 on phenytoin dose requirement. Pharmacogenetics 11:287–291
Nasu K, Kubota T, Ishizaki T (1997) Genetic analysis of CYP2C9 polymorphism in a Japanese population. Pharmacogenetics 7:405–409
Yasar Ü, Eliasson E, Dahl ML, Johansson I, Ingelman-Sundberg M, Sjöqvist F (1999) Validation of methods for CYP2C9 genotyping: frequencies of mutant alleles in a Swedish population. Biochem Biophys Res Commun 254:628–631 (erratum: 258:227)
Dickmann LJ, Rettie AE, Kneller MB, Kim RB, Wood AJ, Stein CM, Wilkinson GR, Schwarz UI (2001) Identification and functional characterization of a new CYP2C9 variant (CYP2C9*5) expressed among African-Americans. Mol Pharmacol 60:382–387
García-Martín E, Martínez C, Ladero JM, Gamito FJ, Agúndez JA (2001) High frequency of mutations related to impaired CYP2C9 metabolism in a Caucasian population. Eur J Clin Pharmacol 57:47–49
Scordo MG, Aklillu E, Yasar Ü, Dahl M-L, Spina E, Ingelman-Sundberg M (2001) Genetic polymorphism of Cytochrome P450 2C9 in a Caucasian and a Black-African population. Br J Clin Pharmacol 52:447–451
Aynacioglu AS, Brockmöller J, Bauer S, Sachse C, Guzelbey P, Ongen Z, Nacak M, Roots I (1999) Frequency of cytochrome P450 CYP2C9 variants in a Turkish population and functional relevance for phenytoin. Br J Clin Pharmacol 48:409–415
Hamdy SI, Hiratsuka M, Narahara K, El-Enany M, Moursi N, Ahmed MS, Mizugaki M (2002) Allele and genotype frequencies of polymorphic cytochromes P450 (CYP2C9, CYP2C19, CYP2E1) and dihydropyrimidine dehydrogenase (DPYD) in the Egyptian population. Br J Clin Pharmacol 53:596–603
Wang SL, Huang J, Lai MD, Tsai JJ (1995) Detection of CYP2C9 polymorphism based on the polymerase chain reaction in Chinese. Pharmacogenetics 5:37–42
Yoon YR, Shon JH, Kim MK, Lim YC, Lee HR, Park JY, Cha IJ, Shin JG (2001) Frequency of cytochrome P450 2C9 mutant alleles in a Korean population. Br J Clin Pharmacol 51:277–280
Kimura M, Ieiri I, Mamiya K, Urae A, Higuchi S (1998) Genetic polymorphism of cytochrome P450 s, CYP2C19, and CYP2C9 in a Japanese population. Ther Drug Monit 20:243–247
Gaedigk A, Casley WL, Tyndale RF, Sellers EM, Jurima-Romet M, Leeder JS (2001) Cytochrome P4502C9 (CYP2C9) allele frequencies in Canadian Native Indian and Inuit populations. Can J Physiol Pharmacol 79:841–847
Davies NM, Anderson KE (1997) Clinical pharmacokinetics of diclofenac. Therapeutic insights and pitfalls. Clin Pharmacokinet 33:184–213
Bort R, Mace K, Boobis A, Gomez-Lechon MJ, Pfeifer A, Castell J (1999) Hepatic metabolism of diclofenac: role of human CYP in the minor oxidative pathways. Biochem Pharmacol 58:787–796
Shimamoto J, Ieiri I, Urae A, Kimura M, Irie S, Kubota T, Chiba K, Ishizaki T, Otsubo K, Higuchi S (2000) Lack of differences in diclofenac (a substrate for CYP2C9) pharmacokinetics in healthy volunteers with respect to the single CYP2C9*3 allele. Eur J Clin Pharmacol 56:65–68
Yasar Ü, Eliasson E, Forslund-Bergengren C, Tybring G, Gadd M, Sjoqvist F, Dahl ML (2001) The role of CYP2C9 genotype in the metabolism of diclofenac in vivo and in vitro. Eur J Clin Pharmacol 57:729–735
Morin S, Loriot MA, Poirier JM, Tenneze L, Beaune PH, Funck-Brentano C, Jaillon P, Becquemont L (2001) Is diclofenac a valuable CYP2C9 probe in humans? Eur J Clin Pharmacol 56:793–797
Takanashi K, Tainaka H, Kobayashi K, Yasumori T, Hosakawa M, Chiba K (2000) CYP2C9 Ile359 and Leu359 variants: enzyme kinetic study with seven substrates. Pharmacogenetics 10:95–104
Acknowledgements
This study was supported by a grant from Ministerio de Sanidad y Consumo, Instituto Carlos III, Spain (FIS 01/0699), and partly by the Swedish Society of Medicine, the European Union INCO-Copernicus Project (ERBIC15CT980340) and the Hungarian-Spanish S&T Cooperation Programme (E-45/2001). The authors thank the volunteers for their participation in the study and A. de la Rubia, O Ledea, I. Almirall, M. Martínez, M. Cáceres, J. Cobaleda, and P. Fernández-Salguero for their contribution in developing methods, samples collection, and analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dorado, P., Berecz, R., Norberto, MJ. et al. CYP2C9 genotypes and diclofenac metabolism in Spanish healthy volunteers. Eur J Clin Pharmacol 59, 221–225 (2003). https://doi.org/10.1007/s00228-003-0588-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-003-0588-0