Heading
Abstract
Objective. The aim of this study was to investigate the effect of hormone-replacement therapy (HRT) on the pharmacokinetics of the selective monoamine oxidase B inhibitor selegiline and its primary metabolites desmethylselegiline and l-metamphetamine.
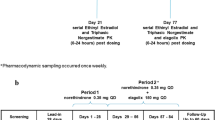
Methods. In this randomised, double-blind, cross-over trial, 12 healthy female subjects received once daily for 10 days either HRT containing 2 mg estradiol valerate and 250 µg levonorgestrel or matched placebo. On day 10, they took a single 10-mg oral dose of selegiline. The serum concentrations of selegiline, desmethylselegiline and metamphetamine were measured for 32 h.
Results. There was a 59% difference (P=0.14) in the area under the serum concentration–time curve (AUC) of selegiline during the HRT compared with the placebo phase, but only a little or no concomitant reduction in the AUC of desmethylselegiline (–7%, P=0.071) or metamphetamine (2%, P=0.614) was observed. Maximum plasma concentration (Cmax) of selegiline was not changed, but a small, statistically significant, reduction in the Cmax of desmethylselegiline (–17%, P=0.03) was seen during the HRT phase. The Cmax of methamphetamine was slightly but not significantly reduced (–5%, P=0.06). The unchanged AUC ratios of desmethylselegiline/selegiline and metamphetamine/selegiline indicate that the primary metabolism of selegiline was not affected by HRT. All study treatments were well tolerated.
Conclusion. Unlike oral contraceptives, HRT is not likely to have clinically significant pharmacokinetic interaction with selegiline.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Accepted in revised form: 7 March 2002
Electronic Publication
Rights and permissions
About this article
Cite this article
Palovaara, S., Anttila, M., Nyman, L. et al. Effect of concomitant hormone replacement therapy containing estradiol and levonorgestrel on the pharmacokinetics of selegiline. Eur J Clin Pharmacol 58, 259–263 (2002). https://doi.org/10.1007/s00228-002-0469-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-002-0469-y