Abstract
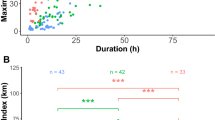
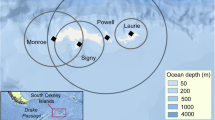
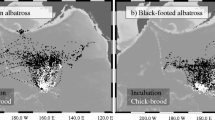
Niche theory predicts that similar species cannot occupy the same geographical space when resources are limited. Sympatric seabirds, such as auks, are ideal models for investigating niche differentiation because they share life history traits and form breeding colonies that rely on common prey items. Auk differentiation may be driven by variations in body mass and wing size, diving capacity, and visual acuity leading each species to forage at different distances, depths, or times of day, respectively. However, previous auk studies have produced diverse results, leaving us with an incomplete understanding of their foraging differentiation across spatial, environmental, and temporal dimensions. In 2021, we tested niche differences at the Mingan Archipelago National Park Reserve, Québec, Canada (50°11′ N, 63°13′ W) by utilizing GPS and time and depth recorders to track the positions of breeding Atlantic puffins (Fratercula arctica), razorbills (Alca torda), and common murres (Uria aalge), which were then paired with environmental data. There was high niche overlap in geographical foraging areas, with auk wing size and mass not appearing to influence their foraging distance. Instead, auk foraging was partitioned over different depths and times of day. Although razorbills and puffins generally exploited shallow foraging areas, puffin foraging activity occurred in deeper waters and at different times of day than razorbills. Murres foraged in the deepest benthic areas and were the only species to forage at night. Our study therefore suggests that auks could be facilitating their coexistence by exhibiting temporal and spatial differences in their foraging behaviours and locations.




Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon request.
References
Abrams PA (1990) Ecological vs evolutionary consequences of competition. Oikos 57:147–215
Ainley DG, Nettleship DN, Carter HR, Storey AE (2020) Common Murre (Uria aalge), version 1.0. Birds of the World
Arnold TB, Emerson JW (2011) Nonparametric goodness-of-fit tests for discrete null distributions. The R Journal 3:34
Barger CP, Kitaysky AS (2012) Isotopic segregation between sympatric seabird species increases with nutritional stress. Biol Let 8:442–445
Barger CP, Young RC, Will A, Ito M, Kitaysky AS (2016) Resource partitioning between sympatric seabird species increases during chick-rearing. Ecosphere 7:e01447
Bates D, Kliegl R, Vasishth S, Baayen H (2018) Parsimonious mixed models. ArXiv 2015
Baylis AMM, Orben RA, Arnould JPY, Peters K, Knox T, Costa DP, Staniland IJ (2015) Diving deeper into individual foraging specializations of a large marine predator, the southern sea lion. Ocealogia 179:1053–1065
Benvenuti S, Dall’Antonia L, Lyngs P (2001) Foraging behaviour and time allocation of chick-rearing razorbills Alca torda at Graesholmen, central Baltic Sea. Ibis 143:402–412
Birt VL, Birt TP, Goulet D, Cairns DK, Montevecchi WA (1987) Ashmole’s halo: direct evidence of prey depletion by a seabird. Mar Ecol Prog Ser 40:205–207
Bonnet-Lebrun AS, Larsen T, Frederiksen M, Fox D, Le Bouard F, Boutet A, Þórarinsson ÞL, Kolbeinsson Y, Deville T, Ratcliffe N (2021) Effects of competitive pressure and habitat heterogeneity on niche partitioning between Arctic and boreal congeners. Sci Rep 11:1–8
Breed GA, Bowen WD, McMillan JI, Leonard ML (2006) Sexual segregation of seasonal foraging habitats in a non-migratory marine mammal. Proc R Soc B Biol Sci 273:2319–2326
Brisson-Curadeau É, Gilchrist HG, Takahashi A, Dutilleul P, Elliott KH (2018) The formation of foraging aggregations in a highly social seabird, the thick-billed murre (Uria lomvia), at small and large scales. Mar Biol 165:170
Büche BI, Stubbings EM, Boyle D, Perrins CM, Yates L (2013) Seabird monitoring on Skomer Island in 2013. JNCC Contract Report, Joint Nature Conservation Committee, Peterborough
Cairns DK, Bredin KA, Montevecchi WA (1987) Activity budgets and foraging ranges of breeding common murres. Auk 104:218–224
Calado JG, Matos DM, Ramos JA, Moniz F, Ceia FR, Granadeiro JP, Paiva VH (2018) Seasonal and annual differences in the foraging ecology of two gull species breeding in sympatry and their use of fishery discards. J Avian Biol 49:e01463. https://doi.org/10.1111/jav.01463
Calenge C (2006) The package “adehabitat” for the R software: a tool for the analysis of space and habitat use by animals. Ecol Model 197:516–519
Chimienti M, Cornulier T, Owen E, Bolton M, Davies IM, Travis JM, Scott BE (2017) Taking movement data to new depths: Inferring prey availability and patch profitability from seabird foraging behavior. Ecol Evol 7:10252–10265
Clay TA, Manica A, Ryan PG, Silk JR, Croxall JP, Ireland L, Phillips RA (2016) Proximate drivers of spatial segregation in non-breeding albatrosses. Sci Rep 6:1–13
Cody ML (1973) Coexistence, coevoluation and convergent evolution in seabird communities. Ecology 54:31–44
Conners MG, Hazen EL, Costa DP, Shaffer SA (2015) Shadowed by scale: subtle behavioral niche partitioning in two sympatric, tropical breeding albatross species. Mov Ecol 3:1–20
Dall’Antonia L, Gudmundsson GA, Benvenuti S (2001) Time allocation and foraging pattern of chick-rearing razorbills in Northwest Iceland. The Condor 103:469–480
Daunt F, Peters G, Scott B, Grémillet D, Wanless S (2003) Rapid-response recorders reveal interplay between marine physics and seabird behaviour. Mar Ecol Prog Ser 255:283–288
Davoren G, Burger A (1999) Differences in prey selection and behaviour during self-feeding and chick provisioning in rhinoceros auklets. Anim Behav 58:853–863
Dehnhard N, Achurch H, Clarke J, Michel LN, Southwell C, Sumner MD, Eens M, Emmerson L (2020) High inter- and intraspecific niche overlap among three sympatrically breeding, closely related seabird species: generalist foraging as an adaptation to a highly variable environment? J Anim Ecol 89:104–119
Delord K, Barbraud C, Pinaud D, Letournel B, Jaugeon B, Goraguer H, Lazure P, Lormée H (2020) Movements of three alcid species breeding sympatrically in Saint Pierre and Miquelon, northwestern Atlantic Ocean. J Ornithol 161:359–371
Elliott KH, Gaston AJ (2015) Diel vertical migration of prey and light availability constrain foraging in an Arctic seabird. Mar Biol 162:1739–1748
Elliott KH, Woo KJ, Gaston AJ, Benvenuti S, Dall’Antonia L, Davoren GK (2009) Central-place foraging in an Arctic seabird provides evidence for Storer-Ashmole’s halo. Auk 126:613–625
Elliott KH, Ricklefs RE, Gaston AJ, Hatch SA, Speakman JR, Davoren GK (2013) High flight costs, but low dive costs, in auks support the biomechanical hypothesis for flightlessness in penguins. Proc Natl Acad Sci 110:9380–9384
Fayet AL, Clucas GV, Anker-Nilssen T, Syposz M, Hansen ES (2021) Local prey shortages drive foraging costs and breeding success in a declining seabird, the Atlantic puffin. J Anim Ecol 90:1152–1164
Fieberg J, Kochanny CO (2005) Quantifying home-range overlap: the importance of the utilization distribution. J Wildl Manag 69:1346–1359
Ford RG, Krumme DW (1979) The analysis of space use patterns. J Theor Biol 76:125–155
Gaston AJ, Jones IL (1998) The auks: Alcidae. Oxford University Press, New York, NY
Gaston AJ, Ydenberg RC, Smith GEJ (2007) Ashmole’s halo and population regulation in seabirds. Mar Ornithol 35:119–126
Gause GF (1934) Experimental analysis of Vito Volterra’s mathematical theory of the struggle for existence. Sci New Ser 79:16–17
GEBCO Bathymetric Compilation Group (2020) The GEBCO_2020 grid-A continuous terrain model of the global oceans and land. Liverpool, British Oceanographic Data Centre, National Oceanography Center, NERC
Gochfeld M (1980) Mechanisms and adaptive value of reproductive synchrony in colonial seabirds. Behavior of marine animals. Springer, Boston, MA, pp 207–270
Gulka J, Davoren GK (2019) High individual flexibility in the foraging behavior of a marine predator, the common murre. Mar Biol 166:83
Gulka J, Ronconi RA, Davoren GK (2019) Spatial segregation contrasting dietary overlap: Niche partitioning of two sympatric alcids during shifting resource availability. Mar Biol 166:115
Hardin G (1960) The competitive exclusion principle. Science 131:1292–1297
Harding AM, Piatt JF, Schmutz JA (2007) Seabird behavior as an indicator of food supplies: sensitivity across the breeding season. Mar Ecol Prog Ser 352:269–274
Harris MP, Bogdanova MI, Daunt F, Wanless S (2012) Using GPS technology to assess feeding areas of Atlantic puffins Fratercula arctica. Ringing Migr 27:43–49
Hutchinson GE (1957) Concluding remarks. Cold Spring Harb Symp Quant Biol 22:415–427
Hutchinson GE (1978) An introduction to population ecology. Yale University Press, New Haven
Isaksson N, Evans TJ, Olsson O, Åkesson S (2019) Foraging behaviour of Razorbills Alca torda during chick-rearing at the largest colony in the Baltic Sea. Bird Study 66:11–21
Ito M, Takahashi A, Kokubun N, Kitaysky AS, Watanuki Y (2010) Foraging behavior of incubating and chick-rearing thick-billed murres Uria lomvia. Aquat Biol 8(3):279–287
Jenkins EJ, Davoren GK (2021) Seabird species-and assemblage-level isotopic niche shifts associated with changing prey availability during breeding in coastal Newfoundland. Ibis 163:183–196
Jessopp M, Arneill GE, Nykänen M, Bennison A, Rogan E (2020) Central place foraging drives niche partitioning in seabirds. Oikos 129:1704–1713
Kokubun N, Yamamoto T, Sato N, Watanuki Y, Will A, Kitaysky AS, Takahashi A (2016) Foraging segregation of two congeneric diving seabird species breeding on St. George Island, Bering Sea. Biogeosciences 13:2579–2591
Koutitonsky VG, Bugden GL (1991) The physical oceanography of the Gulf of St. Lawrence: a review with emphasis on the synoptic variability of the motion. Can Spec Publ Fish Aquat Sci 113:57–90
Lavers J, Hipfner JM, Chapdelaine G (2020) Razorbill (Alca torda), version 1.0. Birds of the World
Lenth RV, Buerkner P, Herve M, Love J, Miguez F, Riebl H, Singmann H (2018) emmeans: estimated marginal means, aka least-squares means. R package version 1.3.0. https://CRAN.R-project.org/package=emmeans
Linnebjerg JF, Fort J, Guilford T, Reuleaux A, Mosbech A, Frederiksen M (2013) Sympatric breeding auks shift between dietary and spatial resource partitioning across the annual cycle. PLoS ONE 8:e72987
Linnebjerg JF, Reuleaux A, Mouritsen KN, Frederiksen M (2015) Foraging ecology of three sympatric breeding alcids in a declining colony in southwest Greenland. Waterbirds 38:143–152
Lowther PE, Diamond AW, Kress SW, Robertson GJ, Russell K, Nettleship DN, Kirwan GM, Christie D, Sharpe CJ, Garcia E, Boesman PFD (2020) Atlantic Puffin (Fratercula arctica), version 1.0. Birds of the World
Lynnes A, Reid K, Croxall J, Trathan P (2002) Conflict or co-existence? Foraging distribution and competition for prey between Adélie and Chinstrap Penguins. Mar Biol 141:1165–1174
Marchand C, Simard Y, Gratton Y (1999) Concentration of capelin (Mallotus villosus) in tidal upwelling fronts at the head of the Laurentian Channel in the St. Lawrence estuary. Can J Fish Aquat Sci 56:1832–1848
Orians GH, Pearson NE (1979) On the theory of central place foraging. In: Horn J, Stairs GR, Mitchell RD (eds) Analysis of ecological systems. Ohio State University Press, Columbus, pp 154–177
Paiva V, Geraldes P, Ramírez I, Meirinho A, Garthe S, Ramos J (2010) Foraging plasticity in a pelagic seabird species along a marine productivity gradient. Mar Ecol Prog Ser 398:259–274
Paredes R, Jones IL, Boness DJ, Tremblay Y, Renner M (2008) Sex-specific differences in diving behaviour of two sympatric Alcini species: thick-billed murres and razorbills. Can J Zool 86(7):610–622
Patterson A, Gilchrist HG, Benjaminsen S, Bolton M, Bonnet-Lebrun AS, Davoren GK, Descamps S, Erikstad KE, Frederiksen M, Gaston AJ et al (2022) Foraging range scales with colony size in high-latitude seabirds. Curr Biol 32:3800–3807
Petalas C, Lazarus T, Lavoie RA, Elliott KH, Guigueno MF (2021) Foraging niche partitioning in sympatric seabird populations. Sci Rep 11:2493
Pianka ER (1973) The structure of lizard communities. Annu Rev Ecol Syst 4:53–74
Piatt JF (1990) The aggregative response of common murres and Atlantic puffins to schools of capelin. Stud Avian Biol 14:36–51
Piatt JF, Nettleship DN (1985) Diving depths of four alcids. Auk 102:293–297
Pratte I, Robertson GJ, Mallory ML (2017) Four sympatrically nesting auks show clear resource segregation in their foraging environment. Mar Ecol Prog Ser 572:243–254
R Core Team (2021) R foundation for statistical computing. https://www.R-project.org/
Rail J-F (2021) Eighteenth census of seabirds breeding in the sanctuaries of the North Shore of the Gulf of St. Lawrence, 2015. The Canadian Field-Naturalist, p 135
Regular P, Davoren G, Hedd A, Montevecchi W (2010) Crepuscular foraging by a pursuit-diving seabird: Tactics of common murres in response to the diel vertical migration of capelin. Mar Ecol Prog Ser 415:295–304
Regular PM, Hedd A, Montevecchi WA (2011) Fishing in the dark: a pursuit-diving seabird modifies foraging behaviour in response to nocturnal light levels. PLoS ONE 6:e26763
Ricklefs RE (1983) Some considerations on the reproductive energetics of pelagic seabirds. Stud Avian Biol 8:84–94
Robertson GS, Bolton M, Grecian WJ, Wilson LJ, Davies W, Monaghan P (2014) Resource partitioning in three congeneric sympatrically breeding seabirds: foraging areas and prey utilization. Auk Ornithol Adv 131:434–446
Robinson JL, Jones IL (2014) An experimental study measuring the effects of a tarsus-mounted tracking device on the behaviour of a small pursuit-diving seabird. Behaviour 151:1799–1826
Senneville S, Schloss IR, Drouin SSO, Bélanger S, Winkler G, Dumont D, Johnston P, St-Onge I (2018) Moderate effect of damming the Romaine River (Quebec, Canada) on coastal plankton dynamics. Estuar Coast Shelf Sci 203:29–43
Shoji A, Elliott K, Fayet A, Boyle D, Perrins C, Guilford T (2015) Foraging behaviour of sympatric razorbills and puffins. Mar Ecol Prog Ser 520:257–267
Shoji A, Aris-Brosou S, Elliott K (2016) Physiological constraints scale with body mass during dives in auks: a comparative analysis. Comp Biochem Physiol A Mol Integr Physiol 196:54–60
Sun A, Whelan S, Hatch SA, Elliott KH (2020) Tags below three percent of body mass increase nest abandonment by rhinoceros auklets, but handling impacts decline as breeding progresses. Mar Ecol Prog Ser 643:173–181
Symons SC, Diamond AW (2019) Short-term tracking tag attachment disrupts chick provisioning by Atlantic Puffins Fratercula arctica and Razorbills Alca torda. Bird Study 66:53–63
Takahashi A, Thiebot JB, Will A, Tsukamoto S, Merkel B, Kitaysky A (2021) Breeding together, wintering an ocean apart: foraging ecology of the northern Bering Sea thick-billed and common murres in years of contrasting sea-ice conditions. Polar Sci 27:100552
Thaxter CB, Wanless S, Daunt F, Harris MP, Benvenuti S, Watanuki Y, Grémillet D, Hamer KC (2010) Influence of wing loading on the trade-off between pursuit-diving and flight in common guillemots and razorbills. J Exp Biol 213:1018–1025
Thieurmel G, Elmarhraoui A (2019) ‘suncalc’: compute sun position, sunlight phases, moon position and lunar phase. R Package Version 0.5. https://cran.r-project.org/web/packages/suncalc/suncalc.pdf
Venables B, Dichmont C (2004) GLMs, GAMs and GLMMs: an overview of theory for applications in fisheries research. Fish Res 70:315–333
Wakefield ED, Owen E, Baer J, Carroll MJ, Daunt F, Dodd SG, Green JA, Guilford T, Mavor RA, Miller PI, Newell MA, Newton SF, Robertson GS, Shoji A, Soanes LM, Votier SC, Wanless S, Bolton M (2017) Breeding density, fine-scale tracking, and large-scale modeling reveal the regional distribution of four seabird species. Ecol Appl 27:2074–2091
Watanuki Y, Wanless S, Harris M, Lovvorn JR, Miyazaki M, Tanaka H, Sato K (2006) Swim speeds and stroke patterns in wing-propelled dives: a comparison between alcids and a penguin. J Exp Biol 209:1217–1230
Weimerskirch H, Bertrand S, Silva J, Bost C, Peraltilla S (2012) Foraging in Guanay cormorant and Peruvian booby, the major guano-producing seabirds in the Humboldt Current System. Mar Ecol Prog Ser 458:231–245
Worton BJ (1989) Kernel methods for estimating the utilization distribution in home-range studies. Ecology 70:164–168
Acknowledgements
The authors thank Francis St-Pierre and Manon Sorais for contributing to data collection in the field. We want to thank Mingan Archipelago National Park Reserve, Parks Canada staff, Aurore Perot, Marie-Claude Roy, Adam Desjardins, and Marie-Bé Leduc who facilitated fieldwork. We also thank Elliott lab members, A. Patterson, and F. Van Oordt, for help with data cleaning. This project was supported by the Fonds de Recherche Québec Nature et Technologies Master’s scholarship to C. Petalas at McGill University. Data collection was supported by Ocean Protection Plan (ECCC).
Funding
Funds and support were provided by the Fonds de Recherche Québec Nature et Technologies Master’s scholarship to C. P. at McGill University.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. CP conducted fieldwork and experiments. Data analysis was conducted by CP with feedback from RAL and KHE throughout. The writing and figures were written and made by CP with feedback from RAL and KHE throughout. All authors contributed and commented on manuscript drafts and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
All applicable institutional guidelines for the care and use of animals were followed. This work was carried out with permits to capture, band, and handle birds from #10711 bird banding permit. All animal experimentation met the Canadian Council for Animal Care wildlife guidelines for the ethical treatment of animals (authorization from the Easter Wildlife Animal Care Committee, Environment and Climate Change Canada, #21RL01). Access to the site was permitted by Parks Canada (MIN-2021–39198) and Canadian Wildlife Service (access to Migratory Bird Sanctuary, #RE-70).
Additional information
Responsible Editor: T.A. Clay.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Petalas, C., Lavoie, R.A. & Elliott, K.H. Multidimensional niche differentiation of chick-rearing sympatric auks in the Gulf of St. Lawrence. Mar Biol 171, 103 (2024). https://doi.org/10.1007/s00227-024-04422-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-024-04422-5