Abstract
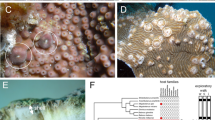
Animal camouflage in mollusks in the Family Ovulidae involves imitating the color and pattern of their host coral to evade predators. However, the extent to which they adapt to host color across regions and coral species remains unquantified. The camouflage efficiency of Phenacovolva rosea across various coral hosts along the coast of Vietnam was assessed. A total of 57 Ph. rosea specimens collected from five coastal sites in Vietnam were photographed with their hosts in the field. The colors of mollusk and host were comparatively analyzed to study contrasts between them. Generalized Linear Model (GLM) analysis investigated factors influencing camouflage efficiency, including host species and region. This revealed a significant effect of coral species identity on mollusk camouflage efficiency, with the lowest contrast recorded for an Astrogorgia sp. and the highest for Menella sp. Region did not significantly influence camouflage efficiency. Linear regression analysis demonstrated that the coral host color explained 30% of the total variance in mollusk color. This study further revealed that Ph. rosea effectively adapts its color to match the coral hosts. The mechanism behind this adaptation remains unknown, but the presumed phenotypic plasticity allowed by nutritional homochromy is the most likely candidate. Poor color adaptation to Menella sp. possibly arises from recent individual migration, maladaptive traits, or alternative color strategies. Overall, phenotypic plasticity in Ph. rosea may play a significant role in survival on different host species in different environments.




Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Barnett JB, Cuthill IC (2014) Distance-dependent defensive coloration. Curr Biol 24:R1157–R1158. https://doi.org/10.1016/j.cub.2014.11.015
Burkepile DE, Hay ME (2007) Predator release of the gastropod Cyphoma gibbosum increases predation on gorgonian corals. Oecologia 154:167–173. https://doi.org/10.1007/s00442-007-0801-4
Caro T, Stankowich T, Kiffner C, Hunter J (2013) Are spotted skunks conspicuous or cryptic? Ethol Ecol Evol 25:144–160. https://doi.org/10.1080/03949370.2012.744359
Cuthill IC (2019) Camouflage. J Zool 308:75–92. https://doi.org/10.1111/jzo.12682
Dewitt TJ, Sih A, Wilson DS (1998) Costs and limits of phenotypic plasticity. Trends Ecol Evol 13:77–81. https://doi.org/10.1016/s0169-5347(97)01274-3
Diamond SE, Martin RA (2021) Buying time: plasticity and population. In: Pfennig DW (ed.) Phenotypic plasticity and evolution. Causes, consequences, controversies, 1st edn. CRC Press, Boca Raton, pp 185–209. https://doi.org/10.1201/9780429343001-11
Draper R, Churchill S (2014) The invisibility cloak: Ovulids and Octocorals of Melanesia and Australia and the magic mantle that links them. Self-Published
Edmunds M (1991) Does warning coloration occur in nudibranchs? Malacologia 32:241–255
Endler JA (1978) A predator’s view of animal coloration. Evol Biol 11:319–364
Fabricius K, Alderslade PN (2001) Soft corals and sea fans: a comprehensive guide to the shallow-water genera of the central-west Pacific, the Indian Ocean and the Red Sea. Australian Institute of Marine Science, Townsville
Grasshoff M (1999) The shallow water gorgonians of New Caledonia and adjacent islands (Coelenterata: Octocorallia). Senck Biol 78:1–121
Grenier S, Barre P, Litrico I (2016) Phenotypic plasticity and selection: nonexclusive mechanisms of adaptation. Scientifica (cairo) 2016:1–9. https://doi.org/10.1155/2016/7021701
Guthrie DM, Muntz WRA (1993) Role of vision in fish behaviour. In: Pitcher TP (ed) Behaviour of teleost fishes, 2nd edn. Chapman & Hall, London, pp 89–121
Harrison RG (1989) Animal mitochondrial DNA as a genetic marker in population and evolutionary biology. Trends Ecol Evol 4:6–11. https://doi.org/10.1016/0169-5347(89)90006-2
Hurst GDD, Jiggins FM (2005) Problems with mitochondrial DNA as a marker in population, phylogeographic and phylogenetic studies: the effects of inherited symbionts. Proc R Soc B 272:1525–1534. https://doi.org/10.1098/RSPB.2005.3056
Kasumyan A, Isaeva O, Zvonareva S (2021) Coloration type of two allied cowries (Ovulidae: Gastropoda) tested through palatability evaluation in feeding experiments with fish. J Exp Mar Biol Ecol 538:151529. https://doi.org/10.1016/j.jembe.2021.151529
Krinsky NI (1989) Antioxidant functions of carotenoids. Free Radical Biol Med 7:617–635. https://doi.org/10.1016/0891-5849(89)90143-3
Lei H (2016) Diterpenoids of gorgonian corals: chemistry and bioactivity. Chem Biodivers 13:345–365
Levis NA, Pfennig DW (2021) Innovation and diversification via plasticity-led evolution. In: Pfennig DW (ed) Phenotypic plasticity and evolution, CRC Press, Boca-Raton, pp 211–240. https://doi.org/10.1201/9780429343001-12
Lorenz F, Fehse D (2009) The living Ovulidae. A manual of the families of allied cowries: Ovulidae, Pediculariidae and Eocypraeidae. Conchbooks, Hackenheim
Lucas MQ, Rodríguez LR, Sanabria DJ, Weil E (2014) Natural prey preferences and spatial variability of predation pressure by Cyphoma gibbosum (Mollusca: Gastropoda) on Octocoral communities off La Parguera. Puerto Rico Int Sch Res Notices 2014:742387. https://doi.org/10.1155/2014/742387
Maia LF, Oliveira VE, Oliveira MER, Reis FD, Fleury BG, Edwards HGM, Oliveira LFC (2012) Colour diversification in octocorals based on conjugated polyenes: a Raman spectroscopic view. J Raman Spectrosc 44:560–566. https://doi.org/10.1002/jrs.4226
O’Neal W, Pawlik JR (2002) A reappraisal of the chemical and physical defenses of Caribbean gorgonian corals against predatory fishes. Mar Ecol Prog Ser 240:117–126
Pawlik JR (2012) Antipredatory defensive roles of natural products from marine invertebrates. In: Fattorusso E, Gerwick WH, Taglialatela-Scafati O (eds) Handbook of marine natural products. Springer Science, New York, pp 677–710. https://doi.org/10.1007/978-90-481-3834-0_12
Pfennig DW (2021) Phenotypic plasticity and evolution: causes, consequences, controversies. CRC Press, Boca Raton. https://doi.org/10.1201/9780429343001
Pfennig DW, Wund MA, Snell-Rood EC, Cruickshank T, Schlichting CD, Moczek AP (2010) Phenotypic plasticity’s impacts on diversification and speciation. Trends Ecol Evol 25:459–467. https://doi.org/10.1016/j.tree.2010.05.006
Rosenberg G (1992) An introduction to the Ovulidae (Gastropoda:Cypraeacea). Am Conchol 20:4–7
Sammarco PW, Coll JC (1992) Chemical adaptations in the Octocorallia: evolutionary considerations. Mar Ecol Prog Ser 88:93–104
Stevens M, Ruxton GD (2019) The key role of behaviour in animal camouflage. Biol Rev 94:116–134. https://doi.org/10.1111/brv.12438
Wesolkowski S, Jernigan ME, Dony RD (1999) Global color image segmentation strategies: euclidean distance vs. vector angle. In: Hu Y-E, Larsen J, Wilson E, Douglas S (eds) Neural networks for signal processing IX. Piscataway, IEEE Press, pp 419–428
Zvonareva SS, Mekhova ES, Hoang DT, Nguyen THT, Vo HT, Fedosov AE (2020) Diversity and relationship of shallow-water Ovulidae (Mollusca: Gastropoda) of Vietnam. Arch Moll 149:113–146. https://doi.org/10.1127/arch.moll/149/113-146
Acknowledgements
The authors thank the staff of Russian-Vietnamese tropical center for providing facilities and organizational assistance during field work. We thank the reviewers for their valuable comments. The work was supported by Russian Science Foundation (Grant number [22-74-00113]).
Funding
This work was supported by Russian Science Foundation (Grant number [22-74-00113]).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by SSZ, MSE and ZSA. The first draft of the manuscript was written by SZ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethic declaration
No approval of research ethics committees was required to accomplish the goals of this study because experimental work was conducted with an unregulated invertebrate species.
Additional information
Responsible Editor: J. Grassle.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zvonareva, S.S., Mekhova, S.E. & Zaitsev, S.A. Phenotypic plasticity of Phenacovolva rosea results in various camouflage efficiencies on different coral host species. Mar Biol 171, 71 (2024). https://doi.org/10.1007/s00227-024-04393-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-024-04393-7