Abstract
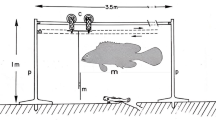
Antipredator behavior of the paralarval stages of merobenthic octopuses is largely unknown. Reported here is an investigation of the behavioral response of Octopus sinensis paralarvae to the sympatric predator fish Girella punctata. Fishes of this species exhibited predatory behavior by engulfing individual paralarvae, but most fishes almost immediately rejected the paralarva from the mouth. No paralarvae died after being engulfed and ejected. The number of such temporary engulfings of a paralarva by a fish during the test period (10 min) decreased with the increase in the number of times a paralarva was engulfed. The frequency of engulfing attacks by the fish also declined. The paralarvae did not eject ink before being engulfed, but 43% of the paralarvae exhibited inking immediately after ejection from the fish’s mouth. The fishes ingested the ink masses released by the paralarvae, and the proportion of fish ingesting the ink masses increased with increasing experience of encountering the ink. When the paralarvae ejected ink, the time interval between first engulfing and subsequent engulfing increased, apparently in relation to distraction of the fish by the ink. These results suggest that paralarvae have an antipredatory mechanism which causes predatory fish to eject them after they have been engulfed. In addition, the paralarvae use the stimulus of being ejected from the mouth of a fish as a cue for ink ejection. The ink mass may be attractive to the fish as a food item because of its potential action as a phagomimetic distraction, enabling the paralarvae to escape.





Similar content being viewed by others
Data availability
All relevant data are within the paper and its Supporting Information files.
References
Agnisola C, Castaldo P, Fiorito G (1996) Octopus vulgaris (Mollusca, Cephalopoda) as a model in behavioral pharmacology: a test of handling effect. Physiol Behev 59:729–733. https://doi.org/10.1016/0031-9384(95)02153-1
Alexander RM (1967) Functional design in fishes. Hutchinson, London
Andrews PLR, Tansey EM (1981) The effects of some anaesthetic agents in Octopus vulgaris. Comp Biochem Physiol C Comp Pharmacol 70:241–247. https://doi.org/10.1016/0306-4492(81)90057-5
Andrews PLR, Darmaillacq A-S, Dennison N, Gleadall IG, Hawkins P, Messenger JB, Osorio D, Smith VJ, Smith JA (2013) The identification and management of pain, suffering and distress in cephalopods, including anaesthesia, analgesia and humane killing. J Exp Mar Biol Ecol 447:46–64. https://doi.org/10.1016/j.jembe.2013.02.010
Bates DM, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects model using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Boletzky SV (1973) Structure and function of the Kölliker organs in young octopods (Mollusca, Cephalopoda). Z Morphol Tiere 75:315–327. https://doi.org/10.1007/bf00288477
Boyle PR, Rodhouse PGK (2005) Cephalopods: ecology and fisheries. Blackwell Science Ltd, Oxford
Crook RJ, Dickson K, Hanlon RT, Walters ET (2014) Nociceptive sensitization reduces predation risk. Curr Biol 24:1121–1125. https://doi.org/10.1016/j.cub.2014.03.043
Dan S, Iwasaki H, Takasugi A, Yamazaki H, Hamasaki K (2018) An upwelling system for culturing common octopus paralarvae and its combined effect with supplying natural zooplankton on paralarval survival and growth. Aquaculture 495:98–105. https://doi.org/10.1016/j.aquaculture.2018.05.036
Dan S, Yamamoto Y, Nishiwaki D, Matsunari H, Kado Y, Yamaki D, Takeshima S, Kamei Y, Hara S, Sakiyama K, Isojima N, Narita A, Hamasaki K (2023) Dietary effects of intensively reared swimming crab Portunus trituberculatus zoeae on survival and growth of East Asian common octopus Octopus sinensis paralarvae. Aquaculture 573:739617. https://doi.org/10.1016/j.aquaculture.2023.739617
Derby CD, Kicklighter CE, Johnson PM, Zhang X (2007) Chemical composition of inks of diverse marine molluscs suggests convergent chemical defenses. J Chem Ecol 33:1105–1113. https://doi.org/10.1007/s10886-007-9279-0
Escánez A, Rubio J, Riera R, Almansa E (2018) Assessment of various anesthetic agents on Octopus vulgaris paralarvae. J World Aquac Soc 49:1019–1025. https://doi.org/10.1111/jwas.12444
Fiorito G, Affuso A, Basil J, Cole A, Girolamo P, D’Angelo L, Dickel L, Gestal C, Grasso F, Kuba M, Mark F, Melillo D, Osorio D, Perkins K, Ponte G, Shasher N, Smith D, Smith J, Andrews PLR (2015) Guidelines for the care and welfare of cephalopods in research -a consensus based on an initiative by CephRes, FELASA and the boyed group. Lab Anim 49:1–90. https://doi.org/10.1177/0023677215580006
Fishelson L, Golani D, Diamant A (2014) SEM study of the oral cavity of members of the Kyphosidae and Girellidae (Pisces, Teleostei), with remarks on Crenidens (Sparidae), focusing on teeth and taste bud numbers and distribution. Zoology 117:122–130. https://doi.org/10.1016/j.zool.2013.10.012
Gerking SD (1994) Mouth and sense organs. In: Gerking SD (ed) Feeding ecology of fish. Academic Press, New York, pp 15–40
Gleadall IG (2013) The effects of prospective anaesthetic substances on cephalopods: summary of original data and a brief review of studies over the last two decades. J Exp Mar Biol Ecol 447:23–30. https://doi.org/10.1016/j.jembe.2013.02.008
Hanlon RT, Messenger JB (1988) Adaptive coloration in young cuttlefish (Sepia officinalis L.): the morphology and development of body patterns and their relation to behaviour. Philos Trans R Soc Lond B Biol Sci 320:437–487. https://doi.org/10.1098/rstb.1988.0087
Hanlon RT, Messenger JB (2018) Cephalopod behaviour, 2nd edn. Cambridge University Press, Cambridge
Lauder GV (1982) Patterns of evolution in the feeding mechanism of actinopterygian fishes. Am Zool 22:275–285. https://doi.org/10.1093/icb/22.2.275
Messenger JB (2001) Cephalopod chromatophores: neurobiology and natural history. Biol Rev 76:473–528. https://doi.org/10.1017/S1464793101005772
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Sauer WHH, Gleadall IG, Downey-Breedt N, Doubleday Z, Gillespie G, Haimovici M, Ibáñez CM, Katugin ON, Leporati S, Lipinski MR, Markaida U, Ramos JE, Rosa R, Villanueva R, Arguelles J, Briceño FA, Carrasco SA, Che LJ, Chen CS, Cisneros R, Conners E, Crespi-Abril AC, Kulik VV, Drobyazin EN, Emery T, Fernández-Álvarez FA, Furuya H, González LW, Gough C, Krishnan P, Kumar B, Leite T, Lu CC, Mohamed KS, Nabhitabhata J, Noro K, Petchkamnerd J, Putra D, Rocliffe S, Sajikumar KK, Sakaguchi H, Samuel D, Sasikumar G, Wada T, Zheng X, Tian Y, Pang Y, Yamrungrueng A, Pecl G (2021) World octopus fisheries. Rev Fish Sci Aquac 29:279–429. https://doi.org/10.1080/23308249.2019.1680603
Villanueva R, Norman MD (2008) Biology of the planktonic stages of benthic octopuses. Oceanogr Mar Biol 46:105–202. https://doi.org/10.1201/9781420065756.ch4
Villanueva R, Coll-Lladó M, Bonnaud-Ponticelli L, Carrasco SA, Escolar O, Fernández-Álvarez FÁ, Gleadall IG, Nabhitabhata J, Ortiz N, Rosas C, Sánchez P, Voight JR, Swoger J (2021) Born with bristles: new insights on the Kölliker’s organs of octopus skin. Front Mar Sci 8:645738. https://doi.org/10.3389/fmars.2021.645738
Wood JB, Maynard AE, Lawlor AG, Sawyer EK, Simmons DM, Pennoyer KE, Derby CD (2010) Caribbean reef squid, Sepioteuthis sepioidea, use ink as a defense against predatory French grunts, Haemulon flavolineatum. J Mar Biol Ecol 388:20–27. https://doi.org/10.1016/j.jembe.2010.03.010
Yagishita N, Nakabo T (2003) Evolutionary trend in feeding habits of Girella (Perciformes: Girellidae). Ichthyol Res 50:358–366. https://doi.org/10.1007/s10228-003-0180-8
Yasuda F (1960) The types of food habits of fishes assured by stomach contents examination. Bull Japan Soc Sci Fish 26:653–662. https://doi.org/10.2331/suisan.26.653 (in Japanese with English abstract)
York CA, Bartol IK (2016) Anti-predator behavior of squid throughout ontogeny. J Exp Mar Biol 480:26–35. https://doi.org/10.1016/j.jembe.2016.03.011
Young RE, Harman RF (1988) “Larva”, “paralarva” and “subadult” in cephalopod terminology. Malacologia 29(1):201–207
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer publishing, New York
Acknowledgements
We thank Kei Yamamoto, Takanari Iwata, Saki Fujiwara, and Zen Yamada for helping with sample collection, and Ian G. Gleadall (AiCeph LLC, Sendai) for assistance in preparing the manuscript and valuable advice. We are also grateful to the editors and anonymous reviewers for valuable comments and suggestions, which have improved the manuscript.
Funding
This research was supported by the research program on development of innovative technology grants from the Project of the Bio-oriented Technology Research Advancement Institution (BRAIN) under Grant Number JPJ007097.
Author information
Authors and Affiliations
Contributions
Conceptualization: SD, KH, methodology: SD, MY, formal analysis and investigation: SD, MY, KH, writing—original draft preparation: SD, writing—review and editing: KH, YK, KS, resources: YK, KS, SD, MY.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest directly relevant to the content of this article.
Ethical approval
This study was approved by the Committee on the Ethics of Animal Experiments at Tokyo University of Marine Science and Technology (permission number: R2-S1), where the experiments were carried out.
Additional information
Responsible Editor: H.-J. Hoving .
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (MP4 97220 KB)
Supplementary file3 (MP4 75479 KB)
Supplementary file4 (MP4 3800 KB)
Supplementary file5 (MP4 5718 KB)
Supplementary file6 (MP4 1772 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dan, S., Yamaji, M., Kamei, Y. et al. Antipredator strategy of paralarvae of East Asian common octopus Octopus sinensis d’Orbigny, 1841: causing rejection after engulfing by a fish, and subsequent ink release for distraction during escape. Mar Biol 170, 109 (2023). https://doi.org/10.1007/s00227-023-04250-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-023-04250-z