Abstract
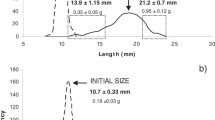
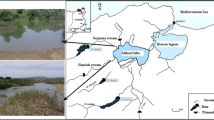
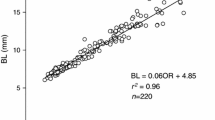
Feeding and growth experiments were conducted to test the hypothesis that physiological attributes responsible for size differentiation among bivalves likely vary with environmental conditions. Juvenile mussels (Mytilus galloprovincialis) were collected from an intertidal population in Biscay, Spain (43°24′42,462″N02°56′43,659″W), in January 2007 and 2009. These mussels were maintained in the laboratory under either restrictive or optimal feeding conditions until fast- and slow-growing individuals on each maintenance regime could be identified. After fast- and slow-growing individuals were identified, the components of energy balance responsible for such growth rate differences were measured. The analysis of physiological traits indicates that under optimal food supply conditions, the capacity to ingest and absorb food and associated costs of growth are the main factors underlying growth rate differences. The set of physiological differences changed when size differentiation took place under restrictive food conditions. Higher rates of absorption coupled with reduced rates of metabolism accounted for faster growth in this case, especially under low food rations.



Similar content being viewed by others
References
Alvarez G, Zapata C, Amaro R, Guerra A (1989) Multilocus heterozygosity at protein loci and fitness in the European oyster, Ostrea edulis L. Heredity 63:359–372
Bayne BL (1999) Physiological components of growth differences between individual oysters (Crassostrea gigas) and a comparison with Saccostrea comercialis. Physiol Biochem Zool 72:705–713
Bayne BL (2000) Relations between variable rates of growth, metabolic costs and growth efficiencies in individual Sydney rock oysters (Saccostrea comercialis). J Exp Mar Biol Ecol 251:185–203
Bayne BL, Hawkins AJS (1997) Protein metabolism, the costs of growth, and genomic heterozygosity: experiments with the mussel Mytilus galloprovincialis Lmk. Phys Zool 70:391–402
Bayne BL, Newell RC (1983) Physiological energetics of marine moluscs. In: Saleudden ASM, Wilbur KM (eds) The Mollusca. Academic Press, New York 4(1): 407–515
Bayne BL, Thompson RJ, Widdows J (1973) Some effects of temperature and food on the rate of oxygen consumption by Mytilus edulis L. In: Wieser W (ed) Effects of temperature on ectothermic organisms. Springer, Berlin, pp 181–193
Bayne BL, Hawkins AJS, Navarro E, Iglesias JIP (1989) Effects of seston concentration on feeding, digestion and growth in the mussel Mytilus edulis. Mar Ecol Prog Ser 55:47–54
Bayne BL, Hedgecock D, McGoldrick D, Rees R (1999a) Feeding behaviour and metabolic efficiency contribute to growth heterosis in Pacific oysters [Crassostrea gigas (Thunberg)]. J Exp Mar Biol Ecol 233:115–130
Bayne BL, Svensson S, Nell JA (1999b) The physiological basis for faster growth in the Sydney rock oyster, Saccostrea commercialis. Biol Bull 197:377–387
Conover RJ (1966) Assimilation of organic matter by zooplankton. Limnol Oceanogr 11:338–354
Crisp DJ (1971) Energy flow measurements. In: Holme NA, Mc Inture AD (eds) Methods for the study of marine benthos, (IBP no 16). Blackwell, Oxford, pp 197–323
David P (1998) Heterozygosity—fitness correlations: new perspectives on old problems. Heredity 80:531–537
David P, Delay B, Berthou P, Jarne P (1995) Alternative models for allozyme-associated heterosis in the marine bivalve Spisula ovalis. Genetics 139:1719–1726
Diehl WJ, Koehn RK (1985) Multiple-locus heterozygosity, mortality and growth in a cohort of Mytilus edulis. Mar Biol 88:256–271
Diehl WJ, Gaffney PM, Koehn RK (1986) Physiological and genetic aspects of growth in the mussel Mytilus edulis. I. Oxygen consumption, growth and weight loss. Physiol Zool 59:201–211
Garton DW (1984) Relationship between multiple locus heterozygosity and physiological energetics of growth in the estuarine gastropod Thais haemastoma. Physiol Zool 57:530–543
Garton DW, Koehn RK, Scott TM (1984) Multiple-locus heterozygosity and the physiological energetics of growth in the coot clam, Munilia lateralis, from a natural population. Genetics 108:445–455
Gentili MR, Beaumont AR (1988) Environmental stress, heterozygosity, and growth rate in Mytilus edulis L. J Exp Mar Biol Ecol 120:145–153
Gnaiger E (1983) Heat dissipation and energetic efficiency in animal anoxibiosis: economy contra power. J Exp Zool 228:471–490
Hawkins AJS (1995) Effects of temperature change on ectotherm metabolism and evolution: metabolic and physiological interrelations underlying the superiority of multi-locus heterozygotes in heterogeneous environments. J Therm Biol 2:23–33
Hawkins AJS, Day AJ (1996) The metabolic basis of genetic differences in growth efficiency among marine animals. J Exp Mar Biol Ecol 203:93–115
Hawkins AJS, Bayne BL, Day AJ (1986) Protein turnover, physiological energetics and heterozygosity in the blue mussel Mytilus edulis: the basis of variable age-specific growth. Proc R Soc Lond 229B:161–176
Hawkins AJS, Widdows J, Bayne BL (1989) The relevance of whole-body protein metabolism to measured costs of maintenance and growth in Mytilus edulis. Physiol Zool 62:745–763
Holey ME, Foltz DW (1987) Effect of multiple-locus heterozygosity and salinity on clearance rate in a brackish water clam, Rangia cuneata (sowerby). J Exp Mar Biol Ecol 111:121–131
Hulbert AJ, Else PL (2005) Membranes and the setting of energy demand. J Exp Biol 208:1593–1599
Koehn RK, Gaffney PM (1984) Genetic heterozygosity and growth rate in Mytilus edulis. Mar Biol 82:1–7
Koehn RK, Diehl WJ, Scott TM (1988) The differential contribution by individual enzymes of glycolysis and protein catabolism to the relationship between heterozygosity and growth rate in the coot clam, Mulinia lateralis. Genetics 118:121–130
Launey S, Hedgecock D (2001) High genetic load in the Pacific oyster Crassostrea gigas. Genetics 159:255–265
LeBlanc N, Tremblay R, Davidson J, Landry T, McNiven M (2008) The effect of selection treatments on Mytilus edulis, modifications of genetic and physiological characteristics. Mar Biol 153:1141–1152
Meyer E, Manahan DT (2010) Gene expression profiling of genetically determined growth variation in bivalve larvae (Crassostrea gigas). J Exp Biol 213:749–758
Pace DA, Marsh AG, Leong Patrick K, Green AJ, Hedgecock D, Manahan DT (2006) Physiological bases of genetically determined variation in growth of marine invertebrate larvae: a study of growth heterosis in the bivalve Crassostrea gigas. J Exp Mar Biol Ecol 335:188–209
Parry GD (1983) The influence of the costs of growth on ectotherm metabolism. J Theor Biol 101:453–477
Pernet F, Tremblay R, Redjah I, Sévigny JM, Gionet C (2008) Physiological and biochemical traits correlate with differences in growth rate and temperature adaptation among groups of the eastern oyster Crassostrea virginica. J Exp Biol 211:969–977
Tamayo D, Ibarrola I, Urrutia MB, Navarro E (2011) The physiological basis for inter-individual growth variability in the spat of clams (Ruditapes philippinarum). Aquaculture 321:113–120
Tamayo D, Ibarrola I, Navarro E (2013) Thermal dependence of clearance and metabolic rates in slow- and fast-growing spats of manila clam Ruditapes philippinarum. J Comp Physiol B 183:893–904
Tamayo D, Ibarrola I, Urrutxurtu I, Navarro E (2014) Physiological basis of extreme growth rate differences in the spat of oyster (Crassostrea gigas). Mar Biol 161:1627–1637
Tamayo D, Ibarrola I, Navarro E (2015) The effects of food conditioning on feeding and growth responses to variable rations in fast and slow growing spat of the Manila clam (Ruditapes philippinarum). J Exp Mar Biol Ecol 471:92–103
Thompson RJ, Bayne BL (1974) Some relationships between growth, metabolism and food in the mussel Mytilus edulis. Mar Biol 27:317–326
Toro JE, Vergara AM (1998) Growth and heterozygosity in a 12-month-old cohort of Ostrea chilensis obtained by mass spawning in laboratory. P S Z N Mar Ecol 19:311–323
Whyte J (1987) Biochemical composition and energy content of six species of phytoplankton used in mariculture of bivalves. Aquaculture 60:231–241
Acknowledgments
This study was funded through the project AGL2009-09981 of the Spanish Ministry of Science and Innovation. D.T. was funded by a FPI grant from the Basque Government. Authors are indebted to two referees for valuable comments and suggestions. Finally, Tamayo D. wants to thank, in particular, the invaluable support received from Muguruza C. over the years. Will you marry me?
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: J. Grassle.
Reviewed by H. Guderley and an undisclosed expert.
Rights and permissions
About this article
Cite this article
Tamayo, D., Azpeitia, K., Markaide, P. et al. Food regime modulates physiological processes underlying size differentiation in juvenile intertidal mussels Mytilus galloprovincialis . Mar Biol 163, 131 (2016). https://doi.org/10.1007/s00227-016-2905-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-016-2905-z