Abstract
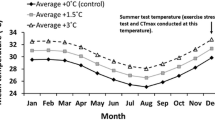
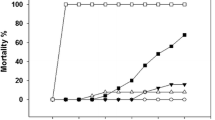
Understanding physiological and molecular compensation mechanisms that shape thermotolerance is crucial for estimating the effects of ocean warming on fish stocks, especially during early life stages, whose tolerance determines recruitment success and population viability. The aims of this study were to assess the sensitivity of fish larvae toward ocean warming and heat wave events in the commercial species, Sparus aurata, whose habitat is likely to be affected by rising water temperatures. We (1) estimated its critical thermal maximum (CTmax) and relative mortality upon warming, (2) quantified stress biomarkers: heat shock protein 70 kDa, total ubiquitin, antioxidant enzymes (superoxide dismutase, catalase, glutathione-S-transferase), lipid peroxidation and protein carbonylation, and (3) analyzed histopathological changes as a result of thermal stress. Larvae showed increasing levels of lethargy with increasing temperature, attaining a cumulative CTmax value of 30 °C. Relative mortality increased upon warming, reaching 80 % at 30 °C. Oxidative damage was higher at moderate temperatures and decreased at 24 °C probably due to a significant increase in superoxide dismutase’s (SODs) activity. Hsp70 chaperone levels also increased at 26 °C, but unfolding persisted at higher temperatures as shown by the increase in total ubiquitin at 26 and 28 °C, indicating protein damage. Skeletal muscle showed disorganization of muscle fibers from 24 °C onwards. Overall, protein denaturation seems to be the major cause of larval mortality, potentially compromising recruitment’s success from 22 °C onwards, since larvae migrate into nursery grounds by spring and summer (i.e., high temperatures), thus hindering the viability of local fish stocks. These data demonstrate that the biochemical homeostasis of fish can be disturbed within an ecologically realistic thermal range and emphasize the risks of rising global temperatures for larval fishes.
Graphical Abstract






Similar content being viewed by others
References
Alamdari DH, Kostidou E, Paletas K, Sarigianni M, Konstas AGP, Karapiperidou A, Koliakos G (2005) High sensitivity enzyme-linked immunosorbent assay (ELISA) method for measuring protein carbonyl in samples with low amounts of protein. Free Radic Biol Med 39:1362–1367
Alami-Durante H, Rouel M, Kentouri M (2006) New insights into temperature-induced white muscle growth plasticity during Dicentrarchus labrax early life: a developmental and allometric study. Mar Biol 149:1551–1565
Andrades JA, Becerra J, Fernández-Llebrez P (1996) Skeletal deformities in larval, juvenile and adult stages of cultures gilthead seabream (S. aurata L.). Aquaculture 141:1–11
Arabaci M, Yilmaz Y, Ceyhun SB et al (2010) A review on population characteristics of gilthead seabream (S. aurata). J Anim Vet Adv 9:976–981
Bartolini F, Barausse A, Pörtner H-O, Giomi F (2013) Climate change reduces offspring fitness in littoral spawners: a study integrating organismic response and long-term time-series. Glob Chang Biol 19:373–386
Basu N, Todgham AE, Ackerman PA et al (2002) Heat shock protein genes and their functional significance in fish. Gene 295:173–183
Blaxter JHS (1992) The effect of temperature on larval fishes. Neth J Zool 42:336–357
Bodinier C, Sucré E, Lecurieux-Belfond L, Blondeau-Bidet E, Charmantier G (2010) Ontogeny of osmoregulation and salinity tolerance in the gilthead sea bream S. aurata. Comp Biochem Physiol A 157:220–228
Cabral HN, Costa MJ, Salgado JP (2001) Does the Tagus estuary fish community reflect environmental changes? Clim Res 18:119–126
Chícharo L, Teodósio MA (1991) Contribuição para o estudo do ictioplâncton do estuário do Guadiana. Rev Biol Univ Aveiro 4:277–286
Damianides P, Chintiroglou CC (2000) Structure and functions of polychaetofauna living in Mytilus galloprovincialis assemblages in Thermaikos Gulf (north Aegean sea). Oceanol Acta 23:323–337
Dimitriou E, Katselis G, Moutopoulos DK, Akovitiotis C, Koutsikopoulos C (2007) Possible influence of reared gilthead sea bream (S. aurata, L.) on wild stocks in the area of the Messolonghi lagoon (Ionian Sea, Greece). Aquac Res 38:398–408
Dionísio G, Campos C, Valente LMP, Conceição LEC, Cancela ML, Gavaia PJ (2012) Effect of egg incubation temperature on the occurrence of skeletal deformities in Solea senegalensis. J Appl Ichthyol 28:471–476
Donelson JM, Munday PL, McCormick MI, Pitcher CR (2011) Rapid transgenerational acclimation of a tropical reef fish to climate change. Nat Clim Chang 2:30–32
Dunphy BJ, Ragg NLC, Collings MG (2013) Latitudinal comparison of thermotolerance and Hsp70 production in F2 larvae of the greenshell mussel (Perna canaliculus). J Exp Biol 216:1202–1209
FAO (2015) Cultured Aquatic Species Information Programme. Sparus aurata. In: Colloca F, Cerasi S (eds) Fisheries and Aquaculture Department, Rome. http://www.fao.org/fishery/culturedspecies/Sparus_aurata/en. Accessed 6 Apr 2015
Faria AM, Chícharo MA, Gonçalves EJ (2011) Effects of starvation on swimming performance and body condition of pre-settlement S. aurata larvae. Aquat Biol 12:281–289
Feidantsis K, Pörtner H-O, Lazou A, Kostoglou B, Michaelidis B (2009) Metabolic and molecular stress responses of the gilthead seabream S. aurata during long-term exposure to increasing temperatures. Mar Biol 156:797–809
Feidantsis K, Antonopoulou E, Lazou A, Pörtner H-O, Michaelidis B (2013) Seasonal variations of cellular stress response of the gilthead sea bream (S. aurata). J Comp Physiol B 183:625–639
Fischer EM, Schär C (2010) Consistent geographical patterns of changes in high-impact European heatwaves. Nat Geosci 3(6):398–403
Fisher R, Wilson SK (2004) Maximum sustainable swimming speeds of late-stage larvae of nine species of reef fishes. J Exp Mar Biol Ecol 312:171–186
Froese R, Pauly D (eds) (2006) Fish base. World Wide Web electronic publication. http://www.fishbase.org
Fulda S, Gorman AM, Hori O, Samali A (2010) Cellular stress responses: cell survival and cell death. Int J Cell Biol 2010:23. doi:10.1155/2010/214074
Garrabou J, Coma R, Bensoussan N et al (2009) Mass mortality in Northwestern Mediterranean rocky benthic communities: effects of the 2003 heat wave. Glob Chang Biol 15:1090–1103
Georgakopoulou E, Katharios P, Divanach P, Koumoundouros G (2010) Effect of temperature on the development of skeletal deformities in Gilthead seabream (S. aurata Linnaeus, 1758). Aquaculture 308(1–2):13–19
Godbold JA, Calosi P (2013) Ocean acidification and climate change: advances in ecology and evolution. Philos Trans R Soc B 368(1627):20120448
Green BS, Fisher R (2004) Temperature influences swimming speed, growth and larval duration in coral reef fish larvae. J Exp Mar Biol Ecol 299:115–132
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione-S-transferases. The first enzymatic step in mercapturicacid formation. J Biol Chem 246:7130–7139
Hansen J, Sato M, Ruedy R, Lo K, Lea DW, Medina-Elizade M (2006) Global temperature change. Proc Natl Acad Sci 103(39):14288–14293
Houde ED (1989) Comparative growth, mortality, and energetics of marine fish larvae: temperature and implied latitudinal effects. Fish Bull 87:471–495
Houde ED (2008) Emerging from Hjort’s shadow. J Northwest Atl Fish Sci 41:53–70
Ibarra-Zatarain Z, Duncan N (2015) Mating behaviour and gamete release in gilthead seabream (S. aurata, Linnaeus 1758) held in captivity. Span J Agric Res 13(1):e04-001
IPCC (2001) Third assessment report of the working group I. In: Houghton JT et al (eds) The science of climate change. Cambridge University Press, Cambridge
IPCC (2007) Contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change, chap 3. Observations: surface and atmospheric climate change (section 3.8 Changes in extreme events). In: Solomon S, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Tignor M, Miller HL (eds) Climate change 2007: the physical science basis. Cambridge University Press, Cambridge
IPCC (2013) Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. In: Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate change 2013: the physical science basis. Cambridge University Press, Cambridge
IPCC (2014) Contribution of working groups I, II and III to the fifth assessment report of the intergovernmental panel on climate change. In: Pachauri RK, Meyer LA (eds) Climate change 2014: synthesis report. IPCC, Geneva
Johansson LH, Borg LAH (1988) A spectrophotometric method for determination of catalase activity in small tissue samples. Anal Biochem 174:331–336
José R (2012) Sparus aurata larvae production in mesocosm: evaluation of abiotic and biotic parameters. Dissertation, University of Porto, Portugal
Kissil GW, Lupatsch I, Elizur A, Zohar Y (2001) Long photoperiod delayed spawning and increased somatic growth in gilthead seabream (S. aurata). Aquaculture 200:363–379
Koumoundouros G, Ashton C, Xenikoudakis G, Giopanou I, Georgakopoulou E, Stickland N (2009) Ontogenetic differentiation of swimming performance in Gilthead seabream (S. aurata, Linnaeus 1758) during metamorphosis. J Exp Mar Biol Ecol 370:75–81
Kristiansen T, Stock C, Drinkwater KF, Curchitser EN (2014) Mechanistic insights into the effects of climate change on larval cod. Glob Chang Biol 20:1559–1584
Landsman SJ, Gingerich AJ, Philipp DP, Suski CD (2011) The effects of temperature change on the hatching success and larval survival of largemouth bass Micropterus salmoides and smallmouth bass Micropterus dolomieu. J Fish Biol 78:1200–1212
Leis J, McCormick M (2002) The biology, behaviour, and ecology of the pelagic, larval stage of coral reef fishes. In: Sale P (ed) The ecology of fishes on coral reefs. Academic Press, San Diego, pp 171–200
Lesser MP (2006) Oxidative stress in marine environments: biochemistry and physiological ecology. Annu Rev Physiol 68:253–278
Linares-Casenave J, Werner I, Van Eenennaam JP, Doroshov SI (2013) Temperature stress induces notochord abnormalities and heat shock proteins expression in larval green sturgeon (Acipenser medirostris Ayres 1854). J Appl Ichthyol 29:958–967
Logan CA, Somero GN (2011) Effects of thermal acclimation on transcriptional responses to acute heat stress in the eurythermal fish Gillichthys mirabilis (Cooper). Am J Physiol Regul Integr Comp Physiol 300:R1373–R1383
Madeira D, Narciso L, Cabral HN, Vinagre C (2012a) Thermal tolerance and potential impacts of climate change on coastal and estuarine organisms. J Sea Res 70:32–41
Madeira D, Narciso L, Cabral HN, Vinagre C, Diniz MS (2012b) HSP70 production patterns in coastal and estuarine organisms facing increasing temperatures. J Sea Res 73:137–147
Madeira D, Narciso L, Cabral HN, Vinagre C, Diniz MS (2013) Influence of temperature in thermal and oxidative stress responses in estuarine fish. Comp Biochem Physiol A 166:237–243
Madeira D, Vinagre C, Costa PM, Diniz MS (2014) Histopathological alterations, physiological limits, and molecular changes of juvenile S. aurata in response to thermal stress. Mar Ecol Prog Ser 505:253–266
McLeod IM, Rummer JL, Clark TD, Jones GP, McCormick MI, Wenger AS, Munday PL (2013) Climate change and the performance of larval coral reef fishes: the interaction between temperature and food availability. Conserv Physiol 1(1):cot024
Meyer E, Aglyamova GV, Matz MV (2011) Profiling gene expression responses of coral larvae (Acropora milepora) to elevated temperature and settlement inducers using a novel RNA-Seq procedure. Mol Ecol 20(17):3599–3616
Miranda PMA, Coelho FES, Tomé AR, Valente MA, Carvalho A, Pires C, Pires HO, Pires VC, Ramalho C (2002) 20th century portuguese climate and climate scenarios, in climate change in Portugal. In: Santos FD, Forbes K, Moita R (eds) Scenarios, impacts and adaptation measures—SIAM project. Gradiva, Lisboa, pp 23–83
Mora C, Maya MF (2006) Effect of the rate of temperature increase of the dynamic method on the heat tolerance of fishes. J Therm Biol 31:337–341
Mora C, Ospina AF (2001) Tolerance to high temperatures and potential impact of sea warming on reef fishes of Gorgona Island (tropical eastern Pacific). Mar Biol 139:765–769
Moretti A, Fernandez-Criado MP, Cittolin G, Guidastri R (1999) Manual on hatchery production of seabass and gilthead seabream, vol 1. Food and Agriculture Organization of the United Nations, Rome
Munday PL (2014) Transgenerational acclimation of fishes to climate change and ocean acidification. F1000 Prime Rep 6:99
Mylonas C, Zohar Y, Pankhurst N, Kagawa H (2011) Reproduction and broodstock management. In: Pavlidis MA, Mylonas CC (eds) Sparidae: biology and aquaculture of gilthead seabream and others species. Wiley, Oxford, pp 95–121
O’Connor MI, Bruno JF, Gaines SD, Halpern BS, Lester SE, Kinlan BP, Weiss JM (2007) Temperature control of larval dispersal and the implications for marine ecology, evolution, and conservation. Proc Natl Acad Sci USA 104:1266–1271
Pearce AF, Feng M (2013) The rise and fall of the “marine heat wave” off Western Australia during the summer of 2010/2011. J Mar Syst 111–112:139–156
Peck MA, Huebert KB, Llopiz JK (2012) Intrinsic and extrinsic factors driving match–mismatch dynamics during the early life history of marine fishes. Adv Ecol Res 47:178–278
Pepin P (1991) Effect of temperature and size on development, mortality, and survival rates of the pelagic early life history stages of marine fish. Can J Fish Aquat Sci 48(3):503–518
Perry AL, Low PJ, Ellis JR, Reynolds JD (2005) Climate change and distribution shifts in marine fishes. Science 308:1912–1915
Pimentel M, Faleiro F, Dionísio G, Repolho T, Pousão-Ferreira P, Machado J, Rosa R (2014) Defective skeletogenesis and oversized otoliths in fish early stages in a changing ocean. J Exp Biol 217:2062–2070
Plaut I (2001) Critical swimming speed: its ecological relevance. Comp Biochem Physiol A 131:41–50
Polo A, Yafera M, Pascual E (1991) Effects of temperature on egg and larval development of Spartus aurata L. Aquaculture 92:367–375
Pörtner HO, Farrell AP (2008) Physiology and climate change. Science 322:690–692
Rhee JS, Kim BM, Kim RO, Seo JS, Kim IC, Lee YM, Lee JS (2013) Co-expression of antioxidant enzymes with expression of p53, DNA repair, and heat shock protein genes in the gamma ray-irradiated hermaphroditic fish Kryptolebias marmoratus larvae. Aquat Toxicol 140C–141C:58–67
Rosa R, Baptista M, Lopes VM, Pegado MR, Paula R, Trübenbach K, Leal MC, Calado R, Repolho T (2014) Early-life exposure to climate change impairs tropical shark survival. Proc R Soc B 281:1793
Rose TH, Smale DA, Botting G (2012) The 2011 marine heat wave in Cockburn Sound, southwest Australia. Ocean Sci 8:545–550
Salinas S, Munch SB (2012) Thermal legacies: transgenerational effects of temperature on growth in a vertebrate. Ecol Lett 15:159–163
Santos FD, Miranda P (eds) (2006) Climate change in Portugal: scenarios, impacts and adaptation measures—SIAM II project. Gradiva, Lisboa
Shama LNS, Strobel A, Mark FC, Wegner KM (2014) Transgenerational plasticity in marine sticklebacks: maternal effects mediate impacts of a warming ocean. Funct Ecol 28(6):1482–1493
Skjærven KH, Penglase S, Olsvik PA, Hamre K (2013) Redox regulation in Atlantic cod (Gadus morhua) embryos developing under normal and heat-stressed conditions. Free Radic Biol Med 57:29–38
Sola L, Moretti A, Crosetti D, Karaiskou N, Magoulas A, Rossi AR, Rye M, Triantafyllidis A, Tsigenopoulos CS (2007) Genetic effects of domestication, culture and breeding of fish and shellfish, and their impacts on wild populations: Gilthead seabream S. aurata. In: Svåsand T, Crosetti D, García-Vázquez E, Verspoor E (eds) Genetic impact of aquaculture activities on native populations, a European network (EU contract no. RICA-CT-2005-022802). Final scientific report, p 176. http://genimpact.imr.no/
Somero GN (2010) The physiology of climate change: how potentials for acclimatization and genetic adaptation will determine ‘winners’ and ‘losers’. J Exp Biol 213:912–920
Somero GN, Hofmann GE (1997) Temperature thresholds for protein adaptation: when does temperature change start to ‘hurt’? In: Wood CM, McDonald DG (eds) Global warming: implications for freshwater and marine fish. Cambridge University Press, Cambridge, pp 1–24
Storch D, Fernández M, Navarrete SA, Pörtner HO (2001) Thermal tolerance of larval stages of the Chilean kelp crab Taliepus dentatus. Mar Ecol Prog Ser 429:157–167
Suau P, Lopez J (1976) Contribution to knowledge of biology of Gilt-Head (S. aurata L.). Investig Pesq 40:169–199
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxide dismutase. Clin Chem 34:497–500
Sung YY, Pineda C, MacRae TH, Sorgeloos P, Bossier P (2008) Exposure of gnotobiotic Artemia franciscana larvae to abiotic stress promotes heat shock protein 70 synthesis and enhances resistance to pathogenic Vibrio campbellii. Cell Stress Chaperones 13(1):59–66
Uchiyama M, Mihara M (1978) Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 86:271–278
Valente LMP, Moutou KA, Conceição L, Engrola S, Fernandes JMO, Johnston IA (2013) What determines growth potential and juvenile quality of farmed fish species? Rev Aquac 5:1–26
Verdiell-Cubedo D, Oliva-Paterna FJ, Ruiz-Navarro A, Torralva M (2013) Assessing the nursery role for marine fish species in a hypersaline coastal lagoon (Mar Menor, Mediterranean Sea). Mar Biol Res 9:739–748
Vinagre C, Santos FD, Cabral HN, Costa MJ (2009) Impact of climate and hydrology on juvenile fish recruitment towards estuarine nursery grounds in the context of climate change. Estuar Coast Shelf Sci 85:479–486
Vinagre C, Narciso L, Pimentel M, Cabral HN, Costa MJ, Rosa R (2013) Contrasting impacts of climate change across seasons: effects on flatfish cohort. Reg Environ Chang 13:853–859
Vinagre C, Narciso L, Cabral HN, Costa MJ, Rosa R (2014) Thermal sensitivity of native and invasive seabreams. Mar Ecol 35:292–297
Walther G-R, Post E, Convey P, Menzel A, Parmesan C, Beebee TJC, Fromentin J-M, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395
Wernberg T, Smale DA, Tuya F, Thomsen MS, Langlois TJ, de Bettignies T, Bennett S, Rousseaux CS (2013) An extreme climatic event alters marine ecosystem structure in a global biodiversity hotspot. Nat Clim Chang 3:78–82
Werner I, Linares-Casenave J, Van Eenennaam JP, Doroshov SI (2007) The effect of temperature stress on development and heat-shock protein expression in larval green sturgeon (Acipenser medirostris). Environ Biol Fish 79:191–200
Williams BR, van Heerwaarden B, Dowling DK, Sgrò CM (2012) A multivariate test of evolutionary constraints for thermal tolerance in Drosophila melanogaster. J Evol Biol 25:1415–1426
Woodin SA, Hilbish TJ, Helmuth B, Jones SJ, Wethey DS (2013) Climate change, species distribution models, and physiological performance metrics: predicting when biogeographic models are likely to fail. Ecol Evol 3(10):3334–3346
Acknowledgments
The authors would like to thank Marta Martins, Ana Patrícia and Carolina Madeira for the help given in the maintenance of experimental systems and feeding of the organisms. Authors would like to thank MARESA for providing not only S. aurata larvae but also microalgae, rotifers and Artemia salina nauplii.
Funding
This study had the support of the Portuguese Fundação para a Ciência e a Tecnologia (FCT) (individual grants: senior researcher position to CV, SFRH/BPD/72564/2010 to PMC, SFRH/BD/80613/2011 to DM; Project Grants PTDC/MAR/119068/2010 and PTDC/MAR-EST/2141/2012; strategic Project Grants UID/Multi/04378/2013 and UID/MAR/04292/2013).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
We have no competing interests.
Additional information
Responsible Editor: H.-O. Pörtner.
Reviewed by Undisclosed experts.
Rights and permissions
About this article
Cite this article
Madeira, D., Costa, P.M., Vinagre, C. et al. When warming hits harder: survival, cellular stress and thermal limits of Sparus aurata larvae under global change. Mar Biol 163, 91 (2016). https://doi.org/10.1007/s00227-016-2856-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-016-2856-4