Abstract
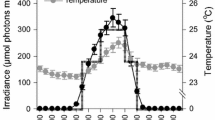
The life-history traits of long-lived benthic littoral invertebrates remain poorly understood. In this study, we analysed patterns of growth in three abundant sublittoral sponges from the western Mediterranean Sea, chosen for their close phylogenetic relatedness, sympatric distribution, and contrasting amounts of photosymbionts: high in Ircinia fasciculata, lower in I. variabilis, and absent in I. oros. Sponge area, perimeter, number of oscula, and epibiont abundance were quantified from in situ digital images taken monthly for 1.5 years and volumetric growth rates were calculated from empirical area–volume relationships. Volumetric growth rates were different among species and coherent with the photosymbiont abundance: high in I. fasciculata (40.03 ± 4.81 % year−1, mean ± SE), low in I. variabilis (5.65 ± 6.11 % year−1), and almost nil in I. oros (−0.04 ± 3.02 % year−1). Furthermore, a marked seasonality was observed in the first two species, with greater growth during the warm season. The high growth rates of I. fasciculata were likely fuelled by symbiont-derived photosynthates and required to compete in the well-lit, algal-dominated habitats this species prefers. In contrast, I. variabilis and I. oros tended to dwell in shaded habitats, where competition from slow-growing invertebrates is intense, and featured lower growth rates. The flattened morphology and lower circularity of I. variabilis indicates a capacity for adaptation to any space that is freed, while I. oros had less oscula and was more massive and circular, suggesting a strategy of passive occupation and minimisation of biological interactions. The results show that even congeneric species living sympatrically can achieve important biomass using different growth and substrate occupation strategies.








Similar content being viewed by others
References
Alfano G, Cimino G, De Stefano S (1979) Palinurin, a new linear sesterpene from a marine sponge. Experientia 35:1136–1137
Amade P, Charroin C, Baby C, Vacelet J (1987) Antimicrobial activities of marine sponges from the Mediterranean Sea. Mar Biol 94:271–275
Arillo A, Bavestrello G, Burlando B, Sarà M (1993) Metabolic integration between symbiotic cyanobacteria and sponges: a possible mechanism. Mar Biol 117:59–162
Ayling AM (1983) Growth and regeneration rates in thinly encrusting demospongiae from temperate waters. Biol Bull 165:343–352
Becerro MA, Uriz MJ, Turon X (1994) Trends in space occupation by the encrusting sponge Crambe crambe: variation in shape as a function of size and environment. Mar Biol 121:301–307
Cebrian E, Uriz MJ, Garrabou J, Ballesteros E (2011) Sponge mass mortalities in a warming mediterranean sea: are cyanobacteria-harboring species worse off? PLoS One 6(6):e20211
Cimino G, De Stefano S, Fattoruso E, Minale L (28) Ircinin-1 and -2 linear sesterterpenes from the marine sponge Ircinia oros. Tetrahedron 28:333–341
Coma R, Ribes M, Gili JM, Zabala M (2000) Seasonality in coastal benthic ecosystems. Trends Ecol Evol 15:448–453
Coma R, Ribes M, Serrano E, Jiménez E, Salat J, Pascual J (2009) Global warming-enhanced stratification and mass mortality events in the Mediterranean. Proc Natl Acad Sci USA 106:6176–6181
De Caralt S, Uriz MJ, Wijffels RH (2008) Grazing, differential size-class dynamics and survival of the Mediterranean sponge Corticium candelabrum. Mar Ecol Prog Ser 360:97–106
Di Camillo CG, Coppari M, Bartolucci I, Bo M, Betti F, Bertolino M, Calcinai B, Cerrano C, De Grandis G, Bavestrello G (2012) Temporal variations in growth and reproduction of Tedania anhelans and Chondrosia reniformis in the North Adriatic Sea. Hydrobiologia 687:299–313
Diaz MC, Rützler K (2001) Sponges: an essential component of Caribbean coral reefs. Bull Mar Sci 69:535–546
Ellison AM, Farnsworth EJ, Twilley RR (1996) Facultative mutualism between red mangroves and root-fouling sponges in Belizean mangal. Ecology 77:2431–2444
Erwin PM, Thacker RW (2008) Phototrophic nutrition and symbiont diversity of two Caribbean sponge–cyanobacteria symbioses. Mar Ecol Prog Ser 362:139–147
Erwin PM, López-Legentil S, González-Pech R, Turon X (2012a) A specific mix of generalists: bacterial symbionts in mediterranean Ircinia spp. FEMS Microbiol Ecol 79:619–637
Erwin PM, López-Legentil S, Turon X (2012b) Ultrastructure molecular phylogenetics and chlorophyll a content of novel cyanobacterial symbionts in temperate sponges. Microb Ecol 64:771–783
Erwin PM, Pita L, López-Legentil S, Turon X (2012c) Stability of sponge–bacteria symbioses over large seasonal shifts in temperature and irradiance. Appl Environ Microbiol 78:7358–7368
Fan L, Reynolds D, Liu M, Stark M, Kjelleberg S, Webster NS, Thomas T (2012) Functional equivalence and evolutionary convergence in complex communities of microbial sponge symbionts. Proc Natl Acad Sci USA 109:E1878–E1887
Freeman CJ, Thacker RW (2011) Complex interactions between marine sponges and their symbiotic microbial communities. Limnol Oceanogr 56:1577–1586
Garrabou J, Zabala M (2001) Growth dynamics in four mediterranean demosponges. Estuar Coast Shelf Sci 52:293–303
Hoppe WF (1988) Growth, regeneration and predation in three species of large coral reef sponges. Mar Ecol Prog Ser 50:117–125
Koopmans M, Wijffels RH (2008) Seasonal growth rate of the sponge Haliclona oculata (Demospongiae: Haplosclerida). Mar Biotechnol 10:502–510
Liu M, Fan L, Zhong L, Kjelleberg S, Thomas T (2012) Metaproteogenomic analysis of a community of sponge symbionts. ISME J 6:1515–1525
López-Legentil S, Ruchty M, Domenech A, Turon X (2005) Life cycles and growth rates of two morphotypes of Cystodytes (Ascidiacea) in the Western Mediterranean. Mar Ecol Prog Ser 296:219–228
Maldonado M, Sánchez-Tocino L, Navarro C (2010) Recurrent disease outbreaks in corneous demosponges of the genus Ircinia: epidemic incidence and defense mechanisms. Mar Biol 157:1577–1590
Mariani S, Uriz MJ, Turon X (2005) The dynamics of sponge larvae assemblages from northwestern Mediterranean nearshore bottoms. J Plankton Res 27:249–262
Marti R, Uriz MJ, Ballesteros E, Turon X (2004) Benthic assemblages in two Mediterranean caves: species diversity and coverage as a function of abiotic parameters and geographic distance. J Mar Biol Assoc UK 84:557–572
McClintock JB, Amsler CD, Baker BJ, Van Soest RWM (2005) Ecology of Antarctic marine sponges: an overview. Integr Comp Biol 45:359–368
McMurray SE, Blum JE, Pawlik JR (2008) Redwood of the reef: growth and age of the giant barrel sponge Xestospongia muta in the florida keys. Mar Biol 155:159–171
Palacín C, Giribet G, Carner S, Dantart L, Turon X (1998) Low densities of sea urchins influence the structure of algal assemblages in the Western Mediterranean. J Sea Res 39:281–290
Pansini M, Pronzato R (1990) Observations on the dynamics of a mediterranean sponge community. In: Rützler K (ed) New perspectives in sponge biology. 3rd international sponge conference, 1985. Smithsonian Institution Press, Washington DC, pp 404–415
Perez T, Garrabou J, Sartoretto S, Harmelin JG, Francour P, Vacelet J (2000) Mortalité massive d’invertébrés marins: un événement sans precedent en Méditerranée nord-occidentale. Life Sci 323:853–886
Pérez-Porro AR, González J, Uriz MJ (2012) Reproductive traits explain contrasting ecological features in sponges: the sympatric poecilosclerids Hemimycale columella and Crella elegans as examples. Hydrobiologia 687:315–330
Pérez-Portela R, Palacín C, Duran S, Turon X (2007) Biological traits of three closely related species of Pycnoclavella (Ascidiacea) in the Western Mediterranean. Mar Biol 152:1031–1038
Reiswig HM (1974) Water transport, respiration and energetics of three tropical marine sponges. J Exp Mar Biol Ecol 14:231–249
Rohde S, Schupp J (2012) Growth and regeneration of the elephant ear sponge Ianthella basta (Porifera). Hydrobiologia 687:219–226
Rützler K (2012) The role of sponges in the Mesoamerican barrier-reef ecosystem, Belize. Adv Mar Biol 61:211–271
Sebens KP (1987) The ecology of indeterminate growth in animals. Annu Rev Ecol Evol Syst 18:371–407
Stabili L, Cardone F, Alifano P, Tredici SM, Piraino S, Corriero G, Gaino E (2012) Epidemic mortality of the sponge Ircinia variabilis (Schmidt, 1862) associated to proliferation of a Vibrio bacterium. Microb Ecol 64:802–813
Steveli JM, Sweat DE, Bert TM, Sim-Smith C, Kelly M (2011) Sponge mortality at Marathon and Long Key, Florida: patterns of species response and population recovery. Proceedings of the 63rd Gulf and Caribbean Fisheries Institute, pp 384–400
Tanaka K (2002) Growth dynamics and mortality of the intertidal encrusting sponge Halichondria okadai (Demospongiae, Halichondrida). Mar Biol 140:383–389
Taylor MW, Radax R, Steger D, Wagner M (2007) Sponge-associated microorganisms: evolution, ecology and biotechnological potential. Microbiol Mol Biol Rev 71:295–347
Teixidó N, Pineda MC, Garrabou J (2009) Decadal demographic trends of a long-lived temperate encrusting sponge. Mar Ecol Prog Ser 375:113–124
Teixidó N, Garrabou J, Harmelin JG (2011) Low dynamics, high longevity and persistence of sessile structural species dwelling on mediterranean coralligenous outcrops. PLoS One 6(8):e23744
Thacker RW (2005) Impacts of shading on sponge–cyanobacteria symbioses: a comparison between host-specific and generalist associations. Integr Comp Biol 45:369–376
Thacker RW, Freeman CJ (2012) Sponge–microbe symbioses: recent advances and new directions. Adv Mar Biol 62:58–111
Trussell GC, Lesser MP, Patterson MR, Genovese SJ (2006) Depth-specific differences in growth of the reef sponge Callyspongia vaginalis: role of bottom-up effects. Mar Ecol Prog Ser 323:149–158
Tsoukatou M, Hellio C, Vagias C, Harvala C, Roussis V (2002) Chemical defense and antifouling activity of three mediterranean sponges of the genus Ircinia. Z Naturforsch 57:161–171
Turon X (1990) Distribution and abundance of ascidians from a locality on the northeast coast of Spain. Mar Ecol PSZNI 11:291–308
Turon X, Becerro MA (1992) Growth and survival of several ascidian species from the northwestern Mediterranean. Mar Ecol Prog Ser 82:235–247
Turon X, Becerro MA, Uriz MJ, Llopis J (1996) Small-scale association measures in epibenthic communities as a clue for allelochemical interactions. Oecologia 108:351–360
Turon X, Tarjuelo I, Uriz MJ (1998) Growth dynamics and mortality of the encrusting sponge Crambe crambe (Poecilosclerida) in contrasting habitats: correlation with population structure and investment in defence. Funct Ecol 12:631–639
Uriz MJ, Rosell D, Martín D (1992) The sponge population of the Cabrera Archipelago (Balearic Islands): characteristics, distribution, and abundance of the most representative species. Mar Ecol PSZNI 13:101–117
Vacelet J, Gaino E, Gallissian MF, Vacelet E (1994) Bacterial attack of spongin skeleton during the 1990 Mediterranean sponge disease. In: van Soest RWM, van Kempen TMG, Braekman JC (eds) Sponges in time and space; biology, chemistry, paleontology. Balkema, Rotterdam, pp 355–362
Van Soest RWM, Boury-Esnault N, Vacelet J et al (2012) Global diversity of sponges (Porifera). PLoS One 7:e35105
Van Treeck P, Eisinger M, Müller J, Paster M, Schuhmacher H (2003) Mariculture trials with mediterranean sponge species. The exploitation of an old natural resource with sustainable and novel methods. Aquaculture 218:439–455
Webster NS, Taylor MW (2011) Marine sponges and their microbial symbionts: love and other relationships. Environ Microbiol 14:335–346
Wulff JL (1985) Patterns and processes of size change in Caribbean demosponges of branching morphology. In: Rützler K (ed) New perspectives in sponge biology. Smithsonian Institution Press, Washington, pp 425–435
Wulff JL (1991) Asexual fragmentation, genotype success, and population dynamics of erect branching sponges. J Exp Mar Biol Ecol 149:227–247
Wulff JL (1997) Mutualisms among species of coral reef sponges. Ecology 78:146–159
Wulff JL (2005) Trade-offs in resistance to competitors and predators, and their effects on the diversity of tropical marine sponges. J Anim Ecol 74:313–321
Wulff JL (2006) Rapid diversity and abundance decline in a Caribbean coral reef sponge community. Biol Conserv 127:167–176
Wulff JL (2008) Life-history differences among coral reef sponges promote mutualism or exploitation of mutualism by influencing partner fidelity feedback. Am Nat 171:597–609
Wulff JL (2012) Ecological interactions and the distribution, abundance and diversity of sponges. Adv Mar Biol 61:273–344
Acknowledgments
We thank Dr. S López-Legentil (UB) for critically reading previous versions of the manuscript and F. Crespo (CEAB) for logistic support in the field. This research was supported by the US National Science Foundation under grant 0853089, the Spanish Government projects CTM2010-22218 and CTM2010-17755, and the Catalan Government Grant 2009SGR-484 for Consolidated Groups.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kühl.
Rights and permissions
About this article
Cite this article
Turon, X., Garriga, A. & Erwin, P.M. Lights and shadows: growth patterns in three sympatric and congeneric sponges (Ircinia spp.) with contrasting abundances of photosymbionts. Mar Biol 160, 2743–2754 (2013). https://doi.org/10.1007/s00227-013-2267-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-013-2267-8